Clear Sky Science · en
DNAM-1 mediates NK-cell activation and host-pathogen interaction via direct binding to fungal cell wall proteases
How our immune sentinels spot silent fungal invaders
Invasive fungal infections caused by Aspergillus and Candida can be deadly for people with weakened immune systems, yet the body often stops these microbes before they take hold. This study uncovers a previously unknown way in which natural killer (NK) cells – fast‑acting immune sentinels – recognize fungal threads and switch on their weapons. By pinpointing a direct contact between an NK‑cell surface molecule called DNAM‑1 and a fungal enzyme named Sap10, the work sheds light on how our immune system senses and responds to dangerous fungi.
Fungi that threaten fragile patients
For most healthy people, inhaled fungal spores are harmlessly cleared. But in patients with damaged or rebuilding immune systems, such as those receiving stem‑cell transplants, fungi like Aspergillus fumigatus in the lungs and Candida albicans in the bloodstream can spread and become life‑threatening. NK cells help defend against these invaders by releasing toxic molecules that damage fungal filaments, known as hyphae, and by secreting chemical signals that recruit and activate other immune cells. To do this effectively, NK cells must recognize that a fungal cell, not a human cell, is in front of them – a task handled by specialized receptors on their surface.
A watchful receptor with a steady stance
DNAM‑1 is best known for helping NK cells detect and attack cancer cells, but its role in fungal infections had not been explored. Using super‑resolution microscopy, the researchers mapped where DNAM‑1 sits on human NK cells. They found that in resting cells, DNAM‑1 is spread evenly over the cell surface, and – unlike some other NK receptors – it stays broadly distributed even when the NK cell makes contact with fungal hyphae. Another receptor, NCAM‑1, clearly clusters at the tight contact zone, or immunological synapse, with the fungus, but DNAM‑1 does not. Despite this calm appearance, when the team applied purified DNAM‑1 protein to fungal hyphae, it bound directly to their cell walls, hinting that the receptor can sense specific fungal components even without visibly clustering.
Hunting for the fungal handshake partner
To identify what DNAM‑1 grabs on the fungal surface, the authors turned to computational analysis of protein domains – recurring building blocks within proteins. Scanning the surface proteomes of Candida albicans and Aspergillus fumigatus, they predicted dozens of candidate fungal proteins that might interact with DNAM‑1. Among the top hits were several secreted aspartic proteases, enzymes that help fungi remodel their cell walls and interact with the host. One, called Sap10 in Candida, stood out because it is anchored at the fungal surface, linked to virulence, and experimentally accessible as a purified protein. Structural modeling suggested that Sap10 could form a tight, extensive contact with the outer portion of DNAM‑1, and that sugar chains attached to Sap10 would further stabilize this interface.
A direct link that flips on NK‑cell attack programs
Experiments confirmed this predicted handshake. In a modified pull‑down assay, fluorescently tagged Sap10 was efficiently captured only when DNAM‑1 was present, indicating high‑affinity binding. Fluorescence correlation spectroscopy – a technique that tracks the movement of single molecules – showed that Sap10 and DNAM‑1 moved together in solution in a ratio consistent with strong, specific pairing. On intact cells, fluorescent Sap10 coated the surface of normal human NK cells but not NK cells engineered to lack DNAM‑1, nor unrelated control cells, demonstrating that DNAM‑1 is required for Sap10 to latch onto the NK‑cell membrane.
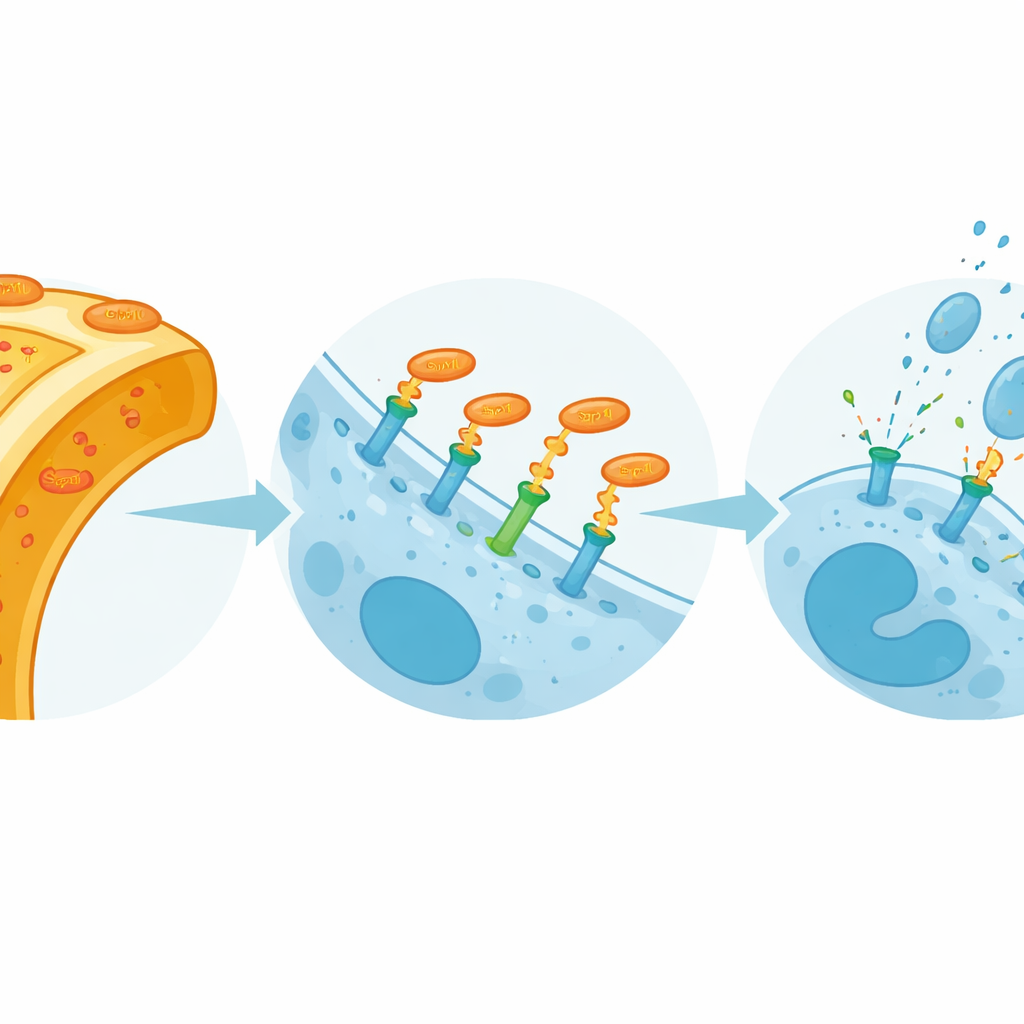
From binding to boosted antifungal defense
Binding alone would be of limited interest if it did not change NK‑cell behavior. The team therefore exposed NK cells to increasing amounts of purified Sap10 and measured classic signs of activation. With higher Sap10 doses and longer exposure, NK cells upregulated CD69, an early activation marker, and released more perforin, a pore‑forming molecule that damages target cells, as well as the chemokine CCL3, which attracts additional immune cells. When DNAM‑1 was blocked with a specific antibody, Sap10‑induced activation and secretion were substantially reduced, showing that this response depends largely on the Sap10–DNAM‑1 interaction. In parallel assays using live fungi, blocking DNAM‑1 partially protected Candida hyphae from NK‑cell damage and increased their survival, confirming that DNAM‑1 contributes meaningfully to antifungal killing.
What this means for fighting fungal disease
This work reveals that DNAM‑1 is not just a cancer‑sensing receptor but also a key fungal sensor on NK cells. By directly binding Sap10 and related fungal surface proteases, DNAM‑1 helps NK cells recognize fungal hyphae and switch on both direct killing mechanisms and chemical alarms that summon reinforcements. For patients at high risk of invasive fungal infections, understanding this molecular handshake could guide future strategies to boost NK‑cell function or design therapies that mimic or enhance DNAM‑1 signaling. In simple terms, the study shows how a specific "lock‑and‑key" contact between our immune cells and fungal enzymes helps the body notice dangerous fungi early and mount a more effective defense.
Citation: Natasha, F., Heilig, L., Helmerich, D.A. et al. DNAM-1 mediates NK-cell activation and host-pathogen interaction via direct binding to fungal cell wall proteases. Commun Biol 9, 537 (2026). https://doi.org/10.1038/s42003-026-10056-8
Keywords: natural killer cells, fungal infections, CD226 DNAM-1, Candida albicans Sap10, innate immunity