Clear Sky Science · en
Exosome-mediated cholesterol flow drives scoliosis progression via promoting the spinal cartilage-bone positive feedback
Why a crooked spine matters
Most people think of scoliosis as a simple spine curve that braces or surgery can fix. But for many teens, we still do not know why a mild bend in the back quietly worsens into a serious deformity. This study looks inside the tiny tissues of the spine to uncover a surprising driver of curve progression: a self-reinforcing loop of aging cartilage cells, weakened bone, and intense local cholesterol traffic between them.
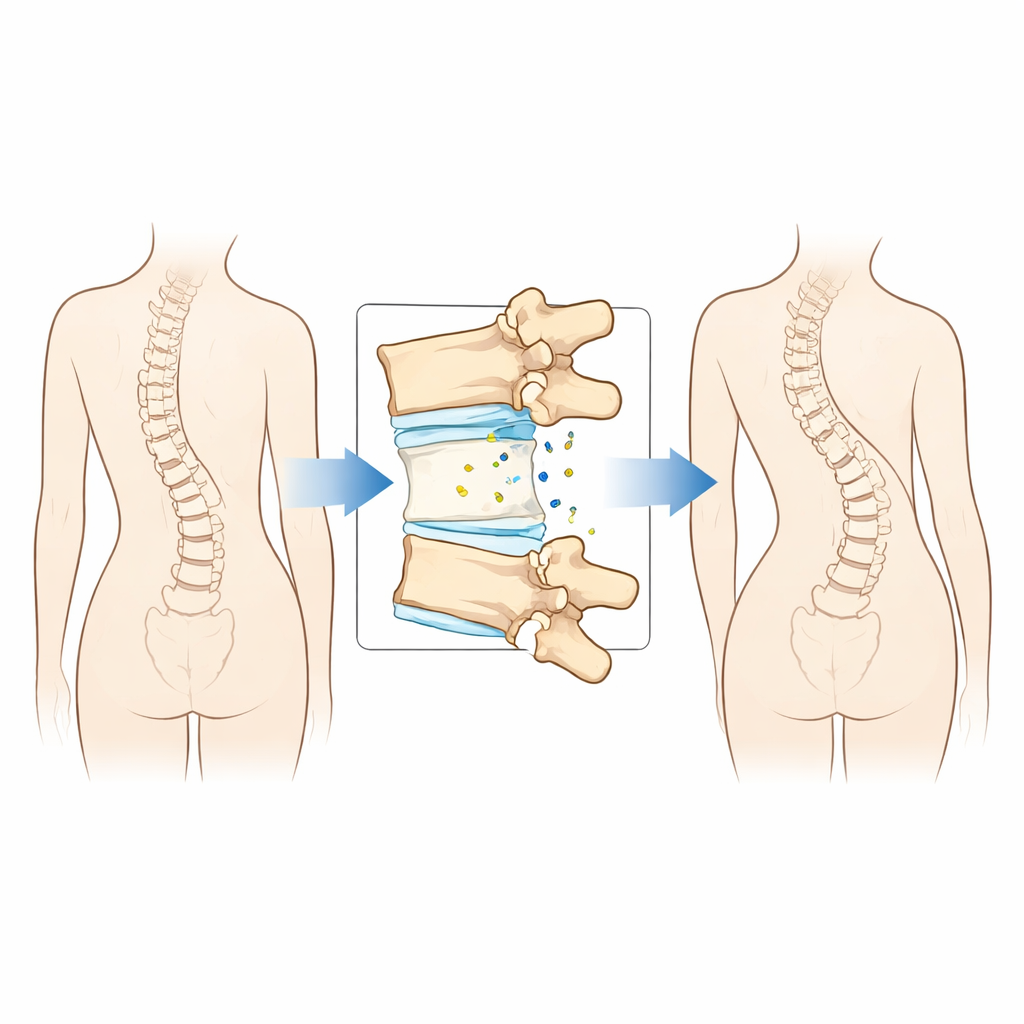
Stressed joints and tired cells
In adolescent idiopathic scoliosis, the spine twists and bends in three dimensions. As this happens, the small facet joints that guide spinal motion are unevenly loaded: some regions bear extra twisting and compression, while others are relatively spared. The researchers examined cartilage from these joints in teens with severe scoliosis and found clear signs that many cartilage cells were prematurely “tired.” These cells showed hallmarks of cellular aging, including damaged DNA, stressed mitochondria, high levels of reactive oxygen molecules, and increased release of inflammatory substances. The worst damage appeared in regions of the curve that experience the greatest mechanical strain, suggesting that abnormal forces on the spine help push cartilage into an aged, breakdown-prone state.
Weak bone beside worn cartilage
Right next to this damaged cartilage sit the vertebral bones. When the team isolated bone-forming cells from the same spinal segments, they found that these osteoblasts were less able to mature and lay down mineral, meaning the bone in these areas was weaker. Importantly, the severity of cartilage aging and bone weakness matched at each location along the curve. Where cartilage was most worn and senescent, bone-building was most impaired. This parallel pattern hinted that cartilage and bone are not suffering in isolation, but rather are communicating in a way that amplifies damage on both sides of the joint.
Tiny message carriers and a cholesterol storm
To understand that communication, the researchers focused on exosomes, microscopic bubbles that cells release to ferry proteins and fats to neighbors. They showed that exosomes from aged cartilage cells could be absorbed by bone cells and slow their development, while exosomes from weakened bone cells could speed up aging and matrix loss in otherwise healthy cartilage. A deeper protein survey revealed two key cargo molecules. Cartilage exosomes were enriched in HSP90β, which boosts cholesterol production and export in bone cells. In response, these bone cells pumped out more cholesterol together with carrier proteins such as APOA1, sending a cholesterol-rich stream into the surrounding fluid. Bone exosomes, in turn, carried APOB, a protein that helps drive cholesterol into cartilage cells. Cartilage cells exposed to this influx accumulated cholesterol, which further fueled cell aging and breakdown through known cholesterol-sensitive molecular pathways.
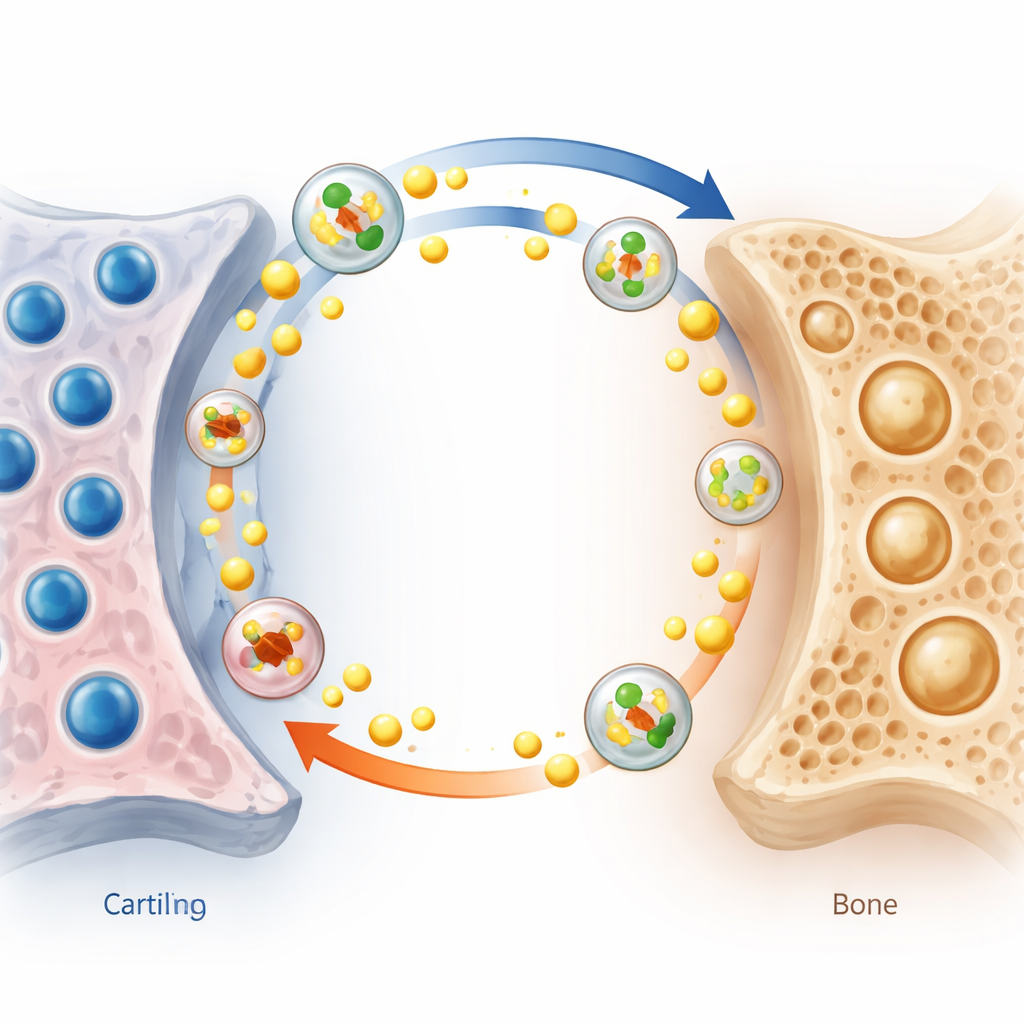
A vicious loop that bends the spine
These findings point to a local “cholesterol loop” between cartilage and bone in the curved spine. Under uneven mechanical stress, cartilage cells age and release HSP90β-packed exosomes that push neighboring bone cells to make and expel cholesterol while losing their bone-building capacity. Bone cells then send APOB-rich exosomes back to cartilage, increasing cholesterol entry and accelerating cartilage wear and tear. This back-and-forth exchange forms a positive feedback loop: more stress leads to more cholesterol flow, more tissue damage, weaker bone, and progressively worsening spinal curvature. Blood tests from patients suggested that this disturbance is confined to the spine’s microenvironment rather than reflecting a whole-body cholesterol problem.
Breaking the loop without surgery
To test whether cutting this loop could slow scoliosis, the team engineered a mouse model in which bone cells overproduced a key cholesterol-making enzyme while the animals stood upright on their hind legs, increasing spinal load. These mice developed clear spinal curves, mimicking human disease. When treated with two compounds — Corylin, which dampens HSP90β activity, and Avasimibe, which promotes the breakdown of APOB — the mice showed smaller curves, healthier cartilage, and stronger bone, without signs of major side effects. In plain terms, the study proposes that some cases of scoliosis worsen not just because of bone shape or growth patterns, but because cartilage and bone in the stressed spine talk to each other in a harmful, cholesterol-fueled conversation. Interrupting this dialogue by targeting exosomal proteins like HSP90β and APOB could one day offer new, non-surgical ways to keep mild scoliosis from becoming disabling.
Citation: Zuo, M., Xu, H., Yang, Y. et al. Exosome-mediated cholesterol flow drives scoliosis progression via promoting the spinal cartilage-bone positive feedback. Commun Biol 9, 547 (2026). https://doi.org/10.1038/s42003-026-09960-w
Keywords: adolescent idiopathic scoliosis, spinal cartilage, bone remodeling, exosomes, cholesterol metabolism