Clear Sky Science · en
Rod photoreceptors control the ON vs OFF polarity of cone-signaling neurons
How the Eye Tells Light from Dark
When you walk from a sunny sidewalk into a dim room, your eyes somehow keep track of which parts of the scene are brighter and which are darker. This study explores how the retina, the light-sensing tissue at the back of the eye, separates signals for light spots and dark spots, and reveals that these signals are much more flexible than previously thought.
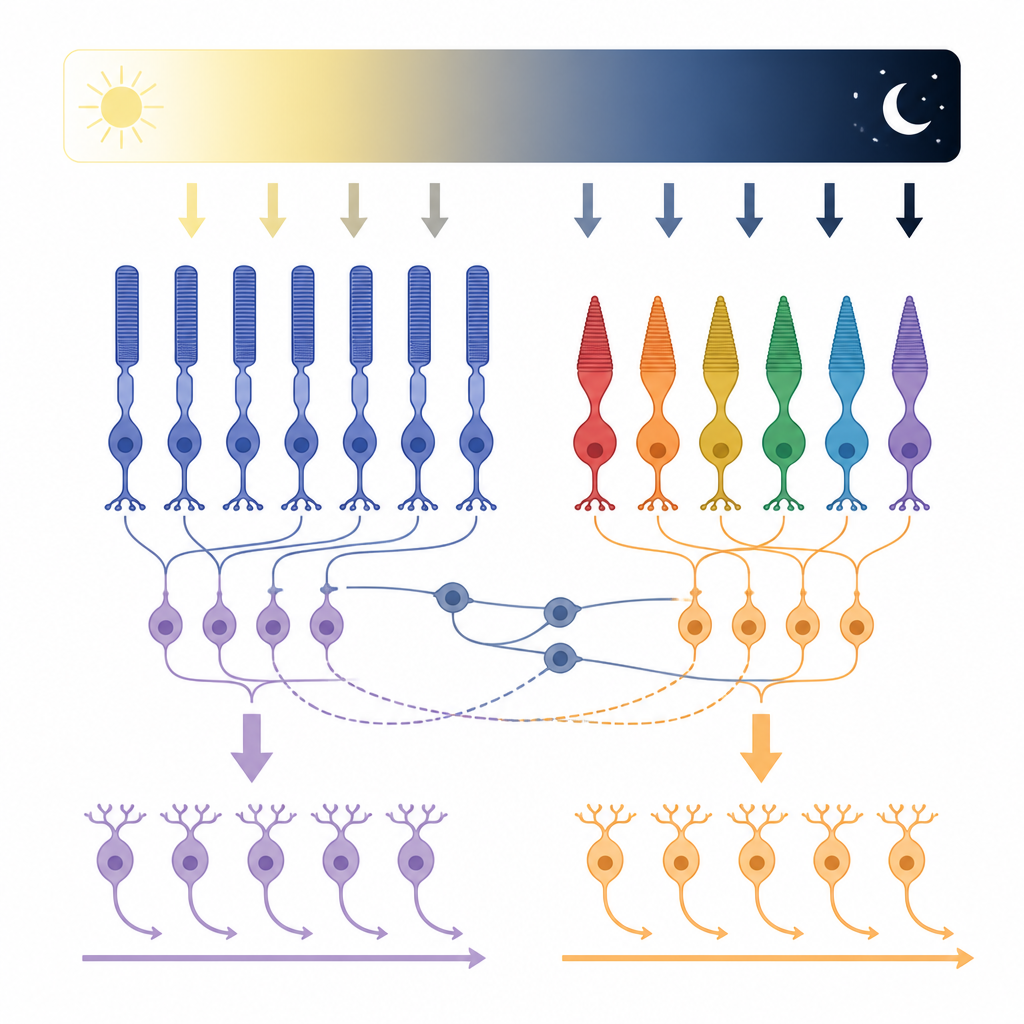
The Usual Story of Light and Dark Channels
Classically, vision scientists describe two main information streams in the retina: one that turns “on” when light appears in a spot, and another that turns “on” when light disappears or a region becomes darker. These so‑called ON and OFF pathways begin when rod and cone cells detect light and pass signals to the next layer of cells called bipolar cells, and then on to additional cells that feed the brain. For decades, the rule seemed simple: ON cells respond to brighter areas, OFF cells to darker areas, and each type connects to matching circuits laid out in distinct layers inside the retina.
A Surprising Flip in Signal Direction
The researchers revisited this rule by recording electrical activity from identified retinal cells in whole, intact mouse retinas. They focused on cone bipolar cells, which carry cone-driven signals, and starburst amacrine cells, which help compute the direction of motion. To their surprise, many cells that are anatomically ON behaved like OFF cells under certain lighting conditions, and vice versa. Instead of only depolarizing to light increments, some ON cells hyperpolarized at light onset, as if their sign had flipped. This polarity switch could occur within minutes and was not simply caused by strong light bleaching the rods, nor by surrounding inhibitory circuits that normally sharpen contrast.
Rods Quietly Rewire Cone Pathways
To find the source of this flip, the team systematically changed background light from very dim (rod‑dominated) to bright (cone‑dominated) levels. The polarity switch was most prominent at intermediate, twilight‑like intensities and faded at very low or very high light levels. Using genetically altered mice lacking either functional rods or cones, they showed that rods are essential for both the appearance and disappearance of the polarity switch. When rods did not work, bipolar cells kept their expected ON or OFF responses. When cones did not work, rod‑based circuits still produced both correct and flipped signals, depending on the light level. These experiments pointed to rods as the hidden driver that can redirect how cone pathways signal light and dark.
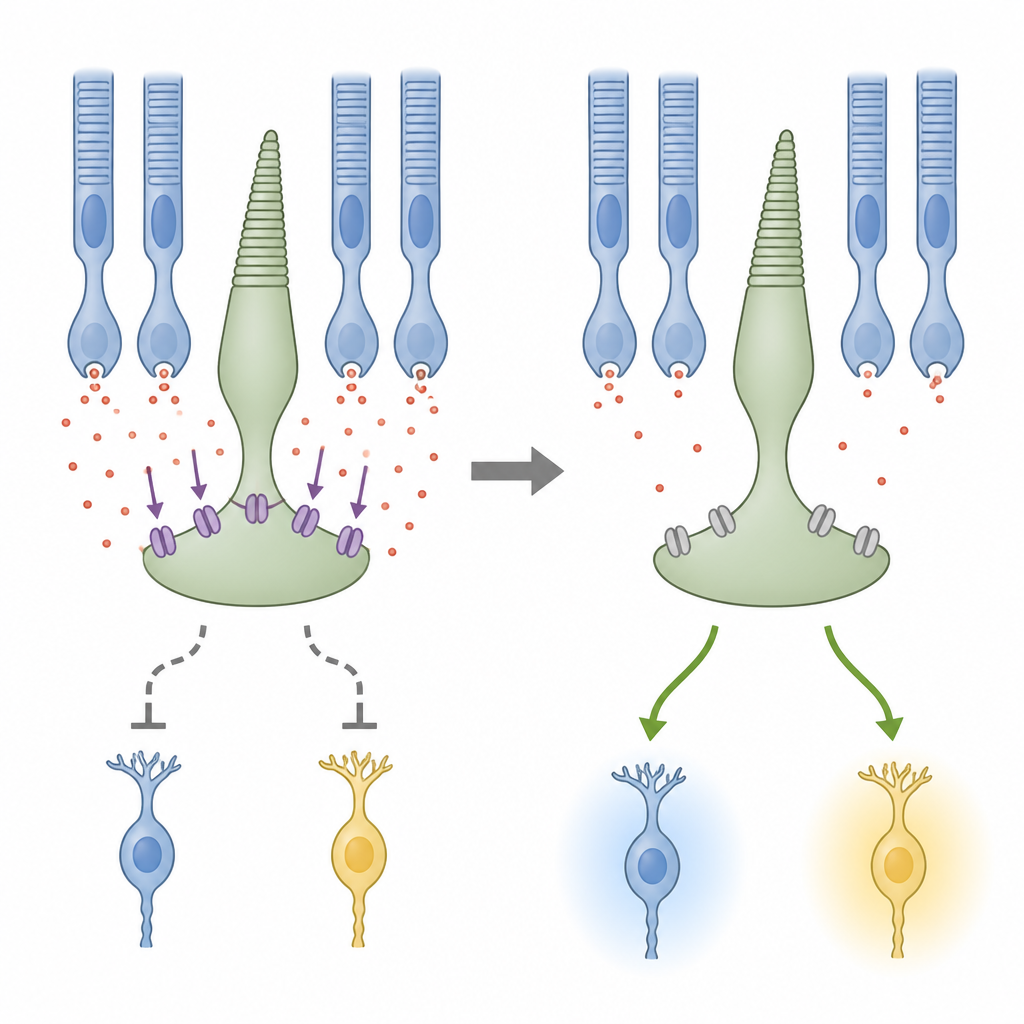
A Chemical Gatekeeper at the First Synapse
The study then probed which retinal structures carry these rod‑driven effects. Blocking electrical junctions between rods and cones, and disrupting feedback from horizontal cells, weakened the normal, “correct” responses at bright light but did not remove the polarity switch at mid‑range light. This suggested that the conventional rod and horizontal cell routes help preserve standard ON and OFF behavior, while another mechanism flips polarity. The key suspect turned out to be a glutamate transporter protein called EAAT5, located at the terminals of photoreceptors. This transporter not only clears chemical signals but also opens a chloride channel that can inhibit cone terminals. When the team applied a drug that blocks these transporters, the polarity switch in bipolar cells disappeared, yet their normal responses remained, implying that EAAT5‑driven chloride currents in cones are responsible for generating the flipped signals.
Why a Flexible Switch Matters
These findings show that the retina’s division into ON and OFF channels is not rigid. Instead, rod activity, working through a transporter‑linked chloride current in cone terminals, can dynamically reverse whether downstream cells treat a change in light as brighter or darker. For everyday vision, especially at dawn, dusk, or in shifting indoor light, this flexibility may help the eye maintain a useful contrast signal across a wide range of brightness. In simple terms, rods not only help you see in the dark; they also act as subtle knobs that can flip and rebalance how cone circuits report light and shadow to the brain.
Citation: Beaudoin, D.L., Hassan, A.R., Shehu, A. et al. Rod photoreceptors control the ON vs OFF polarity of cone-signaling neurons. Commun Biol 9, 637 (2026). https://doi.org/10.1038/s42003-026-09885-4
Keywords: retina, photoreceptors, contrast processing, ON OFF pathways, glutamate transporters