Clear Sky Science · en
Modulating microbial intake helps to maintain the gut microbiome diversity
Why the microbes in our meals matter
Our intestines are teeming with trillions of microbes that help digest food, train the immune system, and may influence everything from metabolism to mood. Doctors often look at how many different kinds of microbes we carry—a measure called diversity—because a less diverse gut community is frequently linked with poorer health. While much attention has gone to the nutrients in what we eat, this study asks a simpler but overlooked question: how does the number and timing of live microbes that arrive with food or probiotics shape the diversity of the gut community over time?
Looking at meals as waves of incoming microbes
The authors built a mathematical model of a gut community where many microbial types compete for space and resources. In this model, each type grows, is washed out of the gut, and is limited by an overall carrying capacity—the maximum total number of microbes the gut can hold. On top of these internal processes, feeding acts as a series of sudden bursts of incoming microbes, such as those swallowed with food or probiotic capsules. Each feeding event adds a fixed number of cells, divided among the different types according to how common they are in the food. Between meals, the community evolves on its own; at mealtimes, immigration briefly dominates as new microbes arrive.
Finding a sweet spot for keeping many species
Without any incoming microbes, the model predicts that one type eventually outcompetes all others, leaving diversity near zero. Introducing periodic feeding changes this picture. When meals are very rare, arrivals cannot fully rescue vulnerable types from being lost. When meals are extremely frequent or huge, the gut community starts to resemble the microbes in the food alone, and local growth and competition matter less. Between these two extremes, the researchers find a “sweet spot” where the average diversity over time is maximized. They call the combination of feeding interval and microbial dose that achieves this the Maximal Diversity Strategy. In simple two-species cases, they can even draw a curve of meal sizes and spacings that keep both types present and most evenly balanced.
What happens when the gut hosts many species
Real gut communities contain many types, so the team extended their analysis to hundreds or even thousands of microbial types with differing growth and removal rates. Using a mix of analytical formulas and computer simulations, they show that as the number of types increases, an optimal strategy almost always exists. Under this strategy, the diversity sustained in the gut converges to the diversity already present in the food. At the same time, the best feeding rate—the average number of microbes entering per unit time—tends toward the average rate at which microbes are cleared from the gut. In other words, for a rich community, maximizing diversity roughly means matching microbial inflow to microbial outflow while supplying a microbially diverse diet.
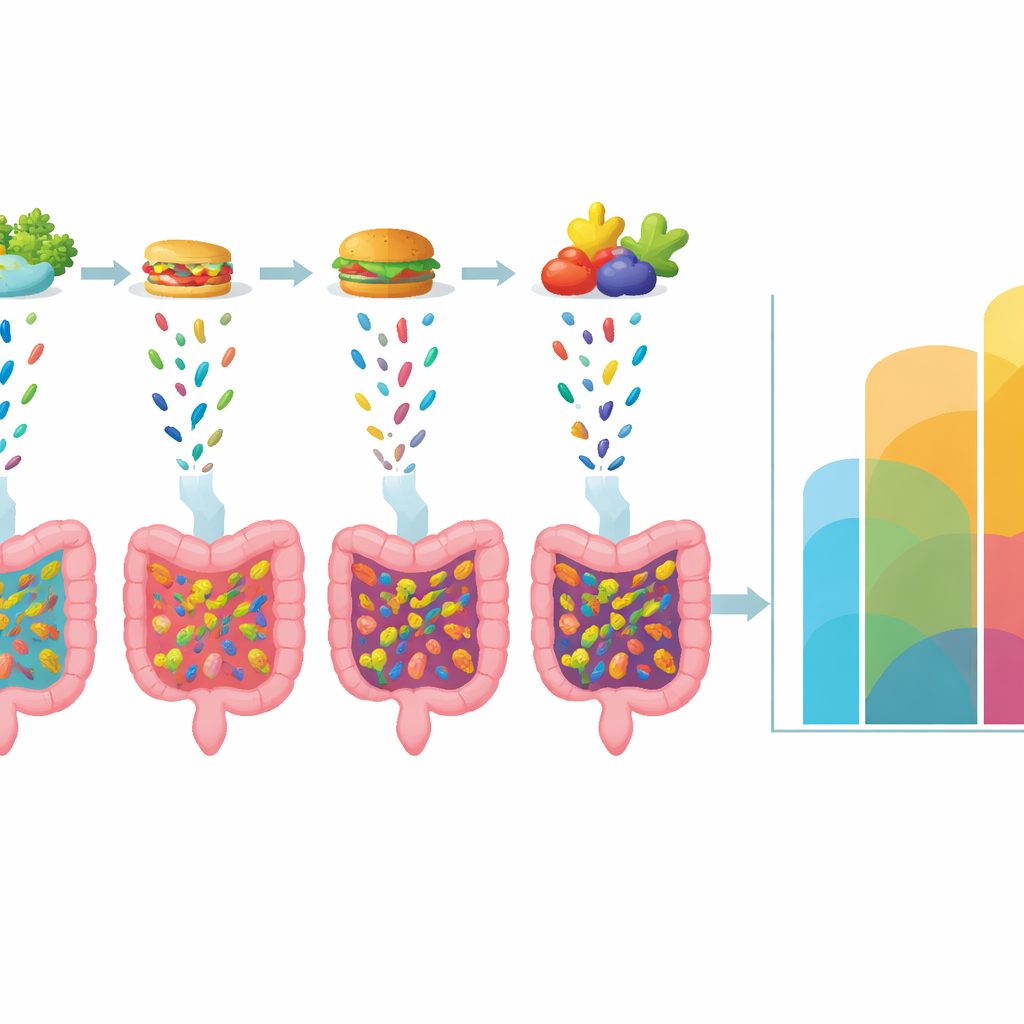
Robust patterns despite real-world variation
The authors tested how sensitive their conclusions are to the details of microbial behavior. They varied how fast species grow, how quickly they are removed, and how unevenly they appear in the food. They also allowed the timing and size of feeding events to fluctuate, mimicking irregular meals and variable probiotic doses. As long as this “dispersal noise” was not extreme, the same core pattern held: there is a broad region of feeding schedules that keep diversity high, and the overall feeding rate, more than the precise timing, is what matters most. Some parameter choices slow how quickly the system reaches the ideal balance, but they rarely remove the possibility of an optimal strategy altogether when many types are present.
What this means for diets and probiotics
This study does not claim that maximizing diversity automatically guarantees health, nor does it track specific disease outcomes. Instead, it offers a clear, testable ecological principle: when many microbial types are competing in the gut, there should be a feeding regime—defined by how often and how microbe-rich meals or live biotherapeutic products are—that best preserves a rich community. For simple experimental systems and smaller animals, the authors estimate that natural feeding patterns may already fall near this balance, while in humans current probiotic doses may be far below what the model suggests would maximize diversity. Their work points toward using quantitative ecology to design better intake protocols for probiotics and other live microbe therapies, and to inspire controlled experiments where the microbial content and timing of meals are tuned independently of calories and nutrients.
Citation: Marquioni, V.M., Hofacker, AC., Villavicencio, J.V. et al. Modulating microbial intake helps to maintain the gut microbiome diversity. Commun Biol 9, 533 (2026). https://doi.org/10.1038/s42003-026-09867-6
Keywords: gut microbiome, microbial diversity, probiotics, intermittent feeding, ecological modeling