Clear Sky Science · en
Normative modeling of MEG brain oscillations across the human lifespan
Listening to the Brain’s Quiet Background
Our brains are never truly at rest. Even when we sit quietly with our eyes open or closed, billions of nerve cells hum together in rhythmic patterns. These invisible brain rhythms change as we grow up, grow older, and when disease takes hold. This study set out to build something like growth charts for these rhythms – a way to see what is typical for a person’s age and sex – and then use those charts to spot when an individual brain falls outside the usual range.
Why Brain Rhythms Matter
Doctors have long used height and weight charts to track a child’s physical development. In recent years, brain researchers have begun doing something similar for brain structure, using MRI scans to map how brain volume and thickness usually change with age. But many brain disorders are not only about structure; they also disrupt how brain cells fire together over time. Techniques like magnetoencephalography (MEG) can capture these fast electrical rhythms, yet there has been no large, lifespan-wide reference for what “normal” brain rhythms look like. Without that reference, it is hard to say whether a given person’s brain activity is unusual, and in what way.
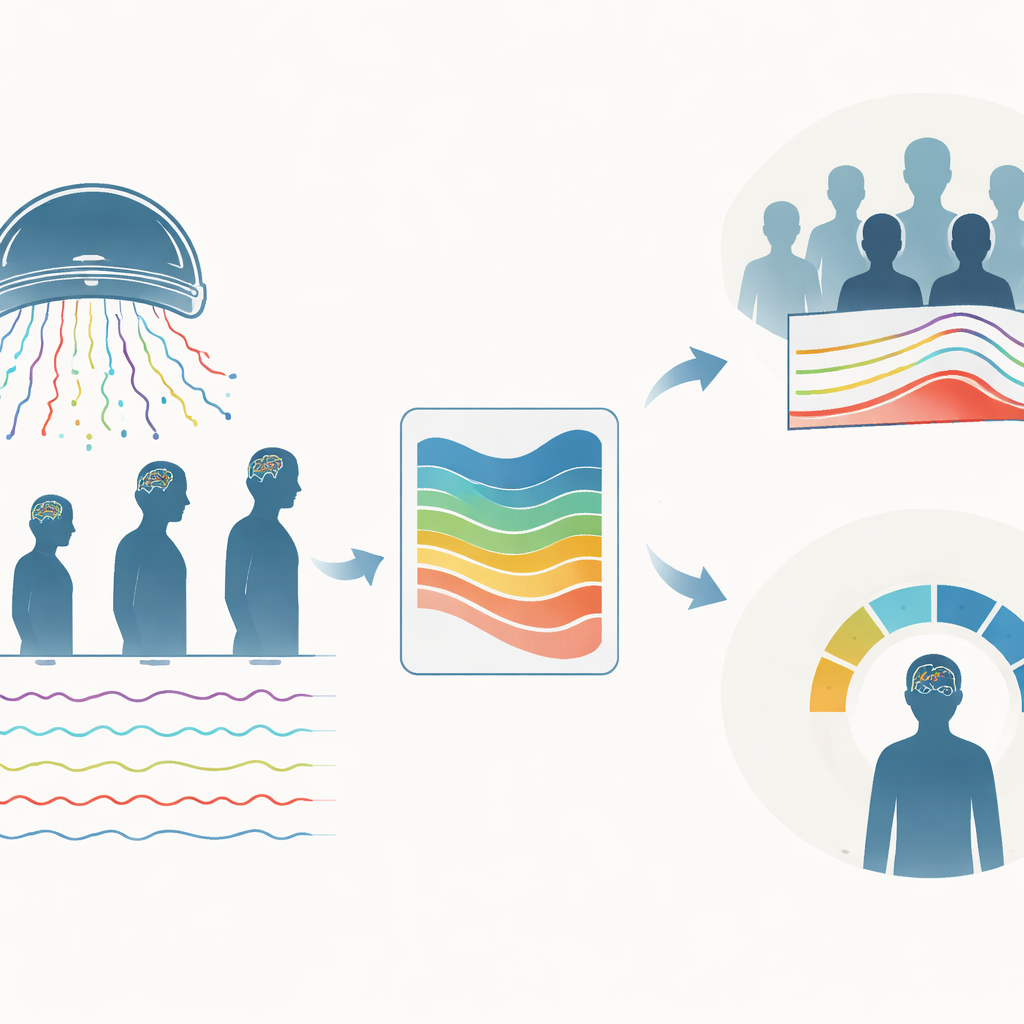
Building Lifespan Charts of Brain Activity
The authors created a framework they call MEGaNorm to fill this gap. They pooled resting-state MEG recordings from 1,846 healthy people aged 6 to 88 years, collected at six sites using three different scanner types, plus 160 people with Parkinson’s disease. From each recording, they cleaned away artifacts such as blinks and heartbeats, then calculated how much of the signal fell into four familiar rhythm ranges: slower theta waves, mid-range alpha and beta waves, and faster gamma waves between 3 and 40 cycles per second. Crucially, they first subtracted a broadband “background hiss” that is always present in brain signals, so the remaining measurements reflected the true rhythmic peaks rather than general noise or age-related shifts in overall power.
Capturing Normal Differences, Not Just Averages
Rather than focusing only on average values, the team used a hierarchical Bayesian approach to estimate full curves describing how each rhythm typically varies with age, and how wide that variation is. Their models allowed the spread and shape of the data to change with age, and they explicitly accounted for differences between recording sites and between males and females. This produced centile curves – analogous to the 5th, 50th, or 95th percentiles on a growth chart – for each rhythm across the lifespan. They then turned these into two kinds of visual tools. Population-level charts (P-NOCs) show, in five-year age steps, how much each rhythm usually contributes to the overall pattern of brain activity. Individual-level charts (I-NOCs) let a clinician or researcher take a single person’s measurements and see where they fall relative to same-age, same-sex, same-scanner norms.
How Brain Rhythms Evolve with Age
The growth charts reveal that brain rhythms follow distinct life courses rather than changing in lockstep. Theta power traces a U-shaped curve, being relatively high in childhood, dipping around midlife, then rising again in later years. Alpha power, often linked to relaxed wakefulness, shows the opposite: it climbs through adolescence, peaks in early adulthood, and gradually declines. Beta power tends to increase up to about age 50, then flattens, while slow gamma remains modest but relatively stable. Around midlife there is a notable shift in how rhythms balance each other: before roughly 50 years, falling alpha is partly offset by rising beta; afterwards, beta plateaus and increasing theta steps in as alpha continues to wane. These patterns underscore that healthy aging involves a redistribution among rhythms, not simply a uniform slowing or weakening.
Spotting Hidden Differences in Parkinson’s Disease
To test the clinical value of their charts, the researchers applied MEGaNorm to the Parkinson’s disease group. They treated all patients as unknowns, feeding their data through the same pipeline and asking how far each person’s rhythms deviated from the age-, sex-, and site-matched norms. The largest and most reliable departures appeared in theta and beta bands: many patients showed unusually strong theta, unusually weak beta, or both, and these deviations helped distinguish them from healthy controls better than alpha or gamma changes. Importantly, patients did not all cluster into a single “Parkinson’s pattern.” Instead, they spread along a continuum from high-theta/low-beta profiles to low-theta/high-beta profiles, with some individuals sitting squarely in the normal range. This diversity would be blurred or lost in traditional group-average comparisons.
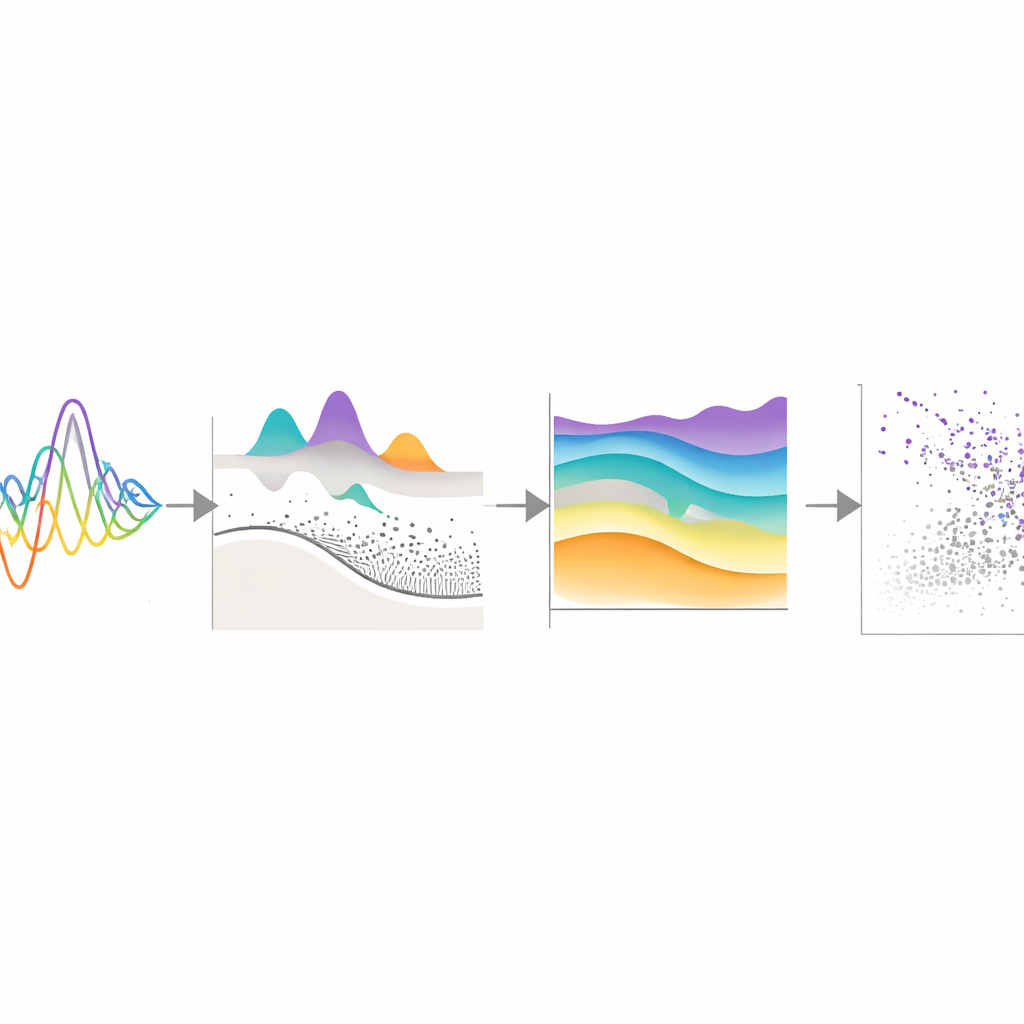
Toward More Personal Brain Health Benchmarks
In everyday terms, this work turns messy measurements of brain “background noise” into intuitive charts that say, for example, “for someone like you, this level of beta activity is higher than 95 percent of your peers.” Because the framework is built to be updated as new data arrive, and can be adapted locally without sharing sensitive recordings, it offers a practical foundation for more personalized brain health assessments. While further testing in different populations and disorders is needed, MEGaNorm shows how we might one day track functional brain changes over time much like we track blood pressure or cholesterol, catching early signs of disease and tailoring treatment based on each individual’s unique rhythm profile.
Citation: Zamanzadeh, M., Verduyn, Y., de Boer, A. et al. Normative modeling of MEG brain oscillations across the human lifespan. Commun Biol 9, 604 (2026). https://doi.org/10.1038/s42003-026-09825-2
Keywords: brain oscillations, magnetoencephalography, lifespan development, Parkinson’s disease, precision psychiatry