Clear Sky Science · en
Single-cell insights into trophoblast heterogeneity and adaptive dysfunction in selective fetal growth restriction
When One Twin Falls Behind
For parents expecting twins, it can be deeply worrying when one baby stops growing as well as the other, even though they share the same womb and placenta. This condition, called selective fetal growth restriction, not only threatens the smaller twin’s health before birth but can also shape long-term development. This study uses cutting-edge single-cell analysis to peer inside the shared placenta of such twin pregnancies, revealing how tiny changes in specific placental cells and immune responses can tip the balance between healthy growth and dangerous shortage.
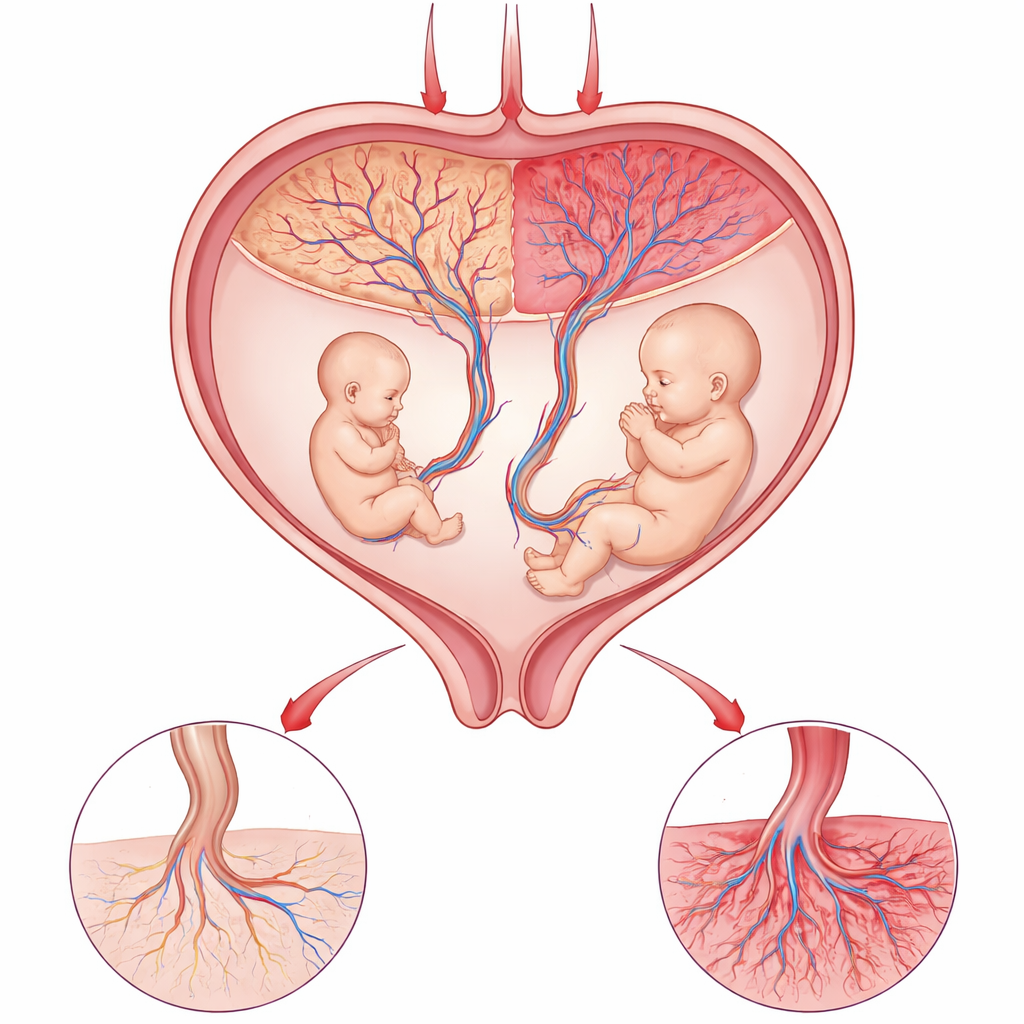
The Placenta’s Delicate Balancing Act
The placenta is the lifeline between mother and baby, delivering oxygen and nutrients while removing waste. In certain identical twin pregnancies, both babies share a single placenta but occupy different territories within it. When one twin becomes much smaller than the other, doctors know that something has gone wrong locally in that shared organ. Because the twins have the same mother and nearly the same genes, they provide a powerful natural experiment: any major differences in growth are likely to come from how their respective parts of the placenta are working, rather than from differences in the babies themselves.
Zooming In on Individual Placental Cells
The researchers collected placental samples from three pregnancies in which one twin was significantly smaller than the other. Using single-cell RNA sequencing—a method that reads the activity of thousands of genes in individual cells—they profiled more than 175,000 cells from the different placental regions. This allowed them to sort cells into distinct types, including support cells, blood vessel cells, and several kinds of immune cells. They focused on a key group called villous cytotrophoblasts, a layer of cells that lines the tiny finger-like projections where maternal blood meets the fetal circulation and that continually replenishes and repairs the surface of the placenta.
Two Key Cell States: Structure Versus Survival Mode
Within these villous cytotrophoblasts, the team discovered two major states with very different roles. One group, marked by a molecule called TP63, acted like structural caretakers: they supported the barrier between mother and baby, maintained cell-to-cell connections, and helped keep the tissue organized. The other group, marked by an enzyme called LDHA, was wired for intense energy production and stress coping, turning up pathways linked to mitochondria, oxidative stress, and protein quality control. In the smaller twins’ placental territories, the protective TP63 cells were depleted, while the stress-driven LDHA cells expanded and cycled more actively. Computer-based “trajectory” analysis suggested that cells were being pushed along a path from healthy structural roles toward a survival mode, with fewer cells reaching the fully fused state that normally maintains a smooth, efficient exchange surface.
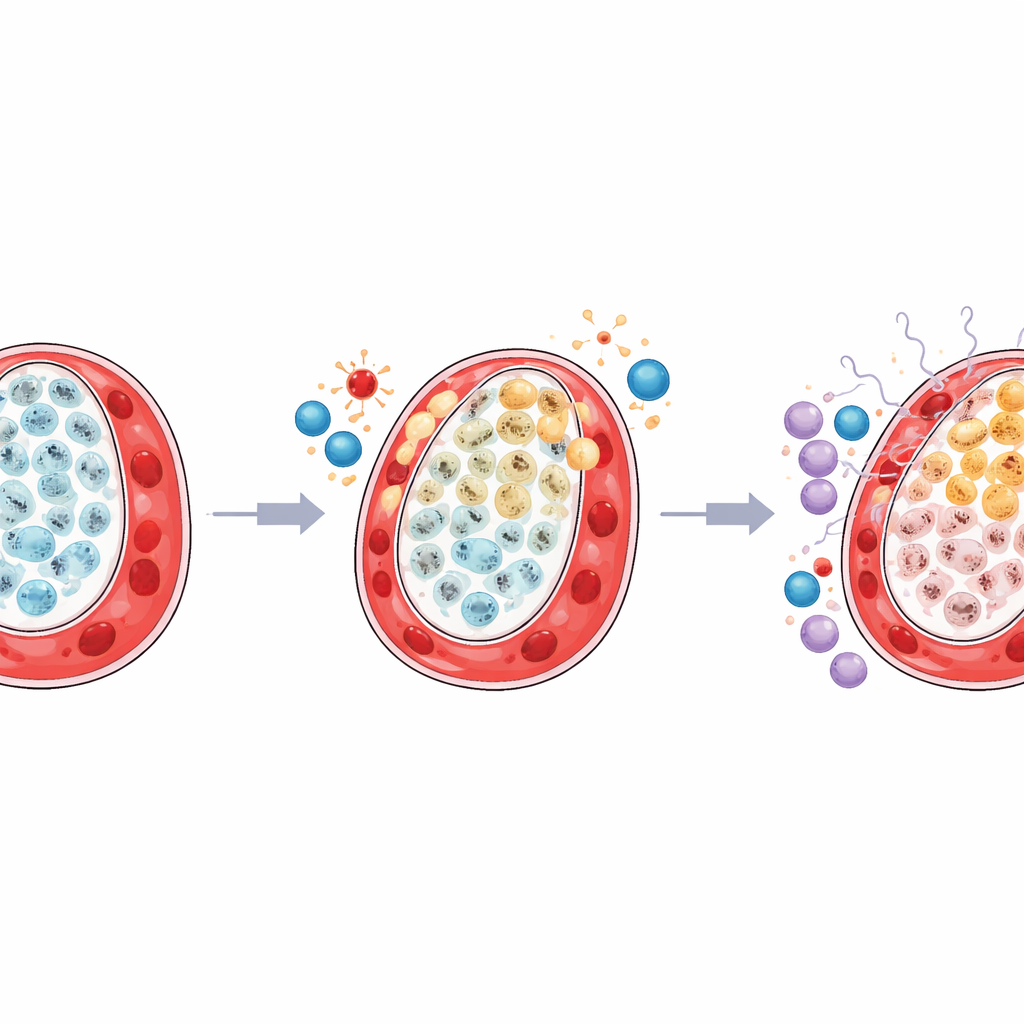
Inflamed Neighborhood Around Stressed Cells
The study also found that the surrounding immune environment in the smaller twin’s placental territory was reshaped in a harmful way. A helpful type of resident macrophage, known as Hofbauer cells, which normally supports tissue health and manages metabolism, was reduced. At the same time, certain natural killer cells, primed for antiviral and inflammatory responses, became more abundant and more active. Detailed mapping of signals passed between cells showed that the stressed LDHA-type trophoblasts engaged more strongly with immune cells through pathways driven by interferon and related alarm molecules. Meanwhile, communication between the structural TP63 cells and supportive stromal and blood vessel cells weakened, suggesting that both physical support and calm immune regulation were being lost.
Three-Way Breakdown That Limits Fetal Growth
Taken together, the work paints a clear, accessible picture of what goes wrong in the placenta when one twin falls behind. Structural support cells that keep the exchange surface tight and orderly are exhausted, energy-hungry stress cells take over, and the immune system shifts into a more inflammatory state. These three threads—loss of structure, metabolic overload, and chronic inflammation—reinforce each other and gradually undermine the placenta’s ability to deliver enough oxygen and nutrients to the smaller baby. By pinpointing the specific cell types and signaling pathways involved, this study provides a roadmap for future tests and treatments aimed at spotting placental trouble earlier and perhaps nudging these cells back toward a healthier balance.
Citation: Bi, Y., Yang, J., Li, X. et al. Single-cell insights into trophoblast heterogeneity and adaptive dysfunction in selective fetal growth restriction. Commun Biol 9, 387 (2026). https://doi.org/10.1038/s42003-026-09798-2
Keywords: placenta, twin pregnancy, fetal growth restriction, single-cell RNA sequencing, trophoblast cells