Clear Sky Science · en
Knocking out OsPht1;9-1;10 genes decreases arsenic accumulation in rice (Oryza sativa) grains
Why the safety of rice matters
For billions of people, especially across Asia, rice is eaten every single day. Yet in many regions, the water and soils used to grow rice contain arsenic, a toxic element linked to cancers and other serious illnesses. Because rice plants are unusually good at pulling arsenic from the soil and sending it into their grains, our daily bowl of rice can quietly become a major source of exposure. This study explores a way to make rice plants that naturally load much less arsenic into their grains—without sacrificing yield or essential nutrients—offering a path toward safer staple foods.
How arsenic sneaks into rice
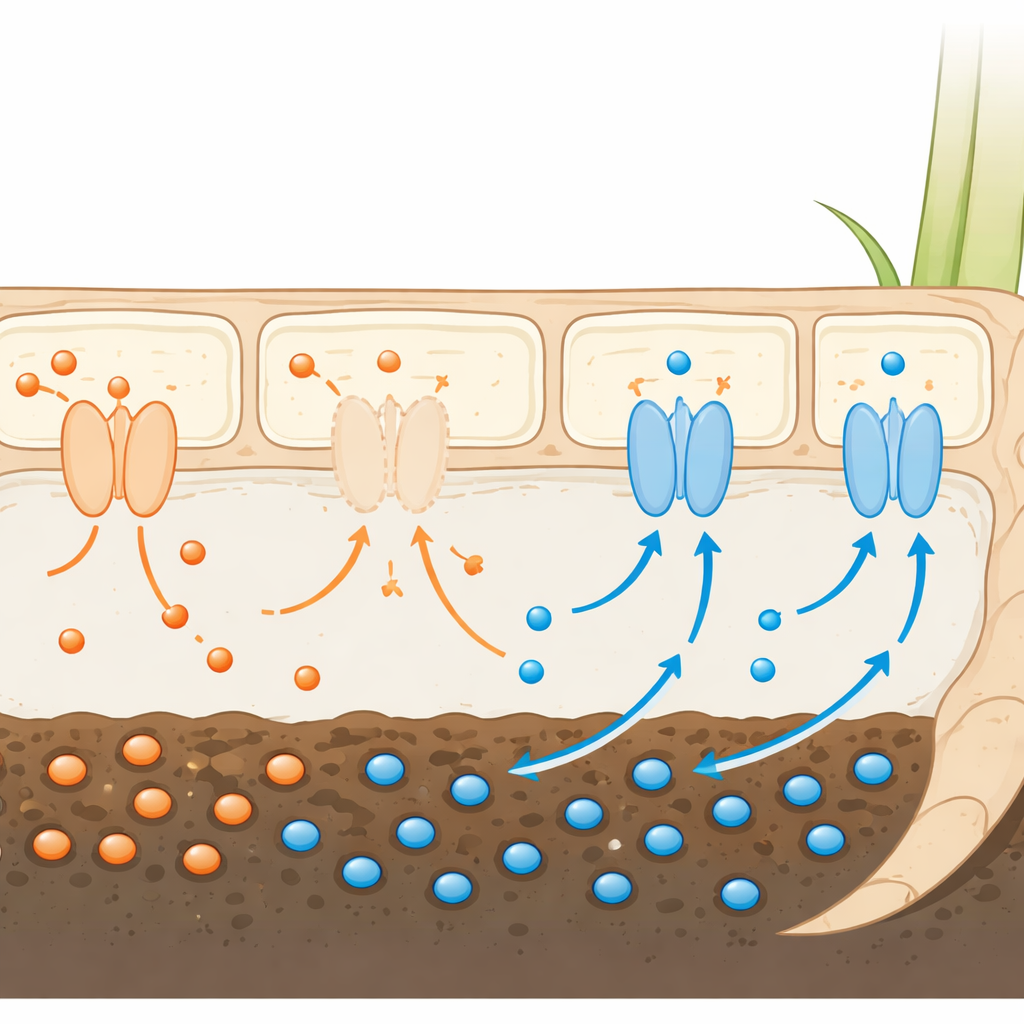
Arsenic in flooded rice paddies occurs mainly in two forms, one of which, arsenate, closely resembles phosphate, a vital plant nutrient. Rice roots rely on specialized protein “gateways” to bring phosphate in from the soil. Because arsenate looks so similar, it can slip through these same gateways and hitch a ride into the plant. Earlier work had shown that turning off some of these gateways can cut arsenic entry, but this often starves the plant of phosphate and sharply lowers grain yield. The challenge has been to find gateways that arsenate uses, but that rice does not depend on heavily for its own nutrition.
Finding the right gateways to target
Rice has at least 13 related phosphate gateways known as Pht1 transporters. The authors focused on two of them, called OsPht1;9 and OsPht1;10, because earlier data suggested they respond strongly when plants encounter arsenate. By examining gene activity in rice roots exposed to arsenate and to low or high phosphate, the team found that OsPht1;10, in particular, is switched on when arsenate is present under normal phosphate conditions. Tests in engineered yeast cells, which lacked their own phosphate gateways, showed that both OsPht1;9 and OsPht1;10 are very effective at bringing arsenate into cells—more so even than a previously known high-capacity arsenate transporter.
Engineering rice to block arsenic, not nutrients
To see what these gateways do inside real plants, the researchers used CRISPR gene-editing to create rice lines in which OsPht1;9 and OsPht1;10 were both disrupted. In hydroponic experiments, these double-mutant plants grew better than normal plants when arsenate was present, with longer roots and higher tolerance. Measurements revealed that their shoots contained 46–66% less arsenic, and the sap that carries water and nutrients from roots to leaves had up to one-third less arsenic. Importantly, under typical phosphate levels the edited plants did not show reduced phosphate content, suggesting that other gateways easily compensated for nutrient uptake while arsenate entry through OsPht1;9 and OsPht1;10 was sharply curtailed.
Field tests in real soils
Laboratory tests can be promising but must be confirmed in the field. The team therefore grew the edited and normal rice plants for full seasons in arsenic-contaminated soils at two locations in southern China, over several different years. Across all trials, plants lacking both OsPht1;9 and OsPht1;10 produced grain yields comparable to regular rice, showing that their overall vigor was not compromised. Yet arsenic levels in whole plants fell by up to 62%, and grain arsenic dropped by roughly 19–67%, depending on year and site. Single-gene mutants, in which only OsPht1;9 or only OsPht1;10 was altered, did not show consistent reductions, underscoring that both gateways must be shut to meaningfully cut arsenic flow to the grain.
What this means for safer rice
This work identifies OsPht1;9 and OsPht1;10 as prime genetic “valves” that channel arsenate into rice without being essential for the plant’s phosphate nutrition under typical farm conditions. By editing both genes, the researchers created rice lines that accumulate far less arsenic in their grains while maintaining normal yields and key micronutrients. Because many countries restrict transgenic approaches that add foreign DNA, these native-gene editing targets—and naturally occurring variants of the same genes—offer a practical route for breeders to develop low-arsenic rice varieties. In the long run, such crops could help reduce a major hidden source of arsenic exposure for millions of people who depend on rice as their daily staple.
Citation: Feng, H., Chen, C., Xu, M. et al. Knocking out OsPht1;9-1;10 genes decreases arsenic accumulation in rice (Oryza sativa) grains. Commun Biol 9, 518 (2026). https://doi.org/10.1038/s42003-026-09741-5
Keywords: arsenic in rice, phosphate transporters, gene-edited crops, food safety, rice breeding