Clear Sky Science · en
Anthraquinones derived from soil actinomycetes combat multidrug-resistant Staphylococcus aureus
A New Ally Against Tough Germs
Drug-resistant infections are a growing worry in hospitals, farms, and even our kitchens. One of the most dangerous culprits is a germ called Staphylococcus aureus, which can shrug off many common antibiotics. This study reports a promising new weapon: a small natural molecule, dug from ordinary soil, that can kill stubborn staph germs, break down their protective slime layers, and do so with little apparent harm to animals or human cells.
Medicine Hidden in the Dirt
The story begins with soil-dwelling microbes called actinomycetes, famous for producing many of our best antibiotics. The researchers previously isolated several related molecules known as anthraquinones from a Streptomyces strain in soil. In this work, they focused on the most powerful one, nicknamed 13394-2. In lab tests, tiny amounts of this compound stopped the growth of multiple strains of drug-resistant Staphylococcus aureus, including notorious hospital and community strains. Even at very low doses, it slowed bacterial growth; at slightly higher doses it wiped the bacteria out completely, performing well across isolates collected from different regions and resistance profiles.
Breaking the Germs’ Armor and Slimy Shields
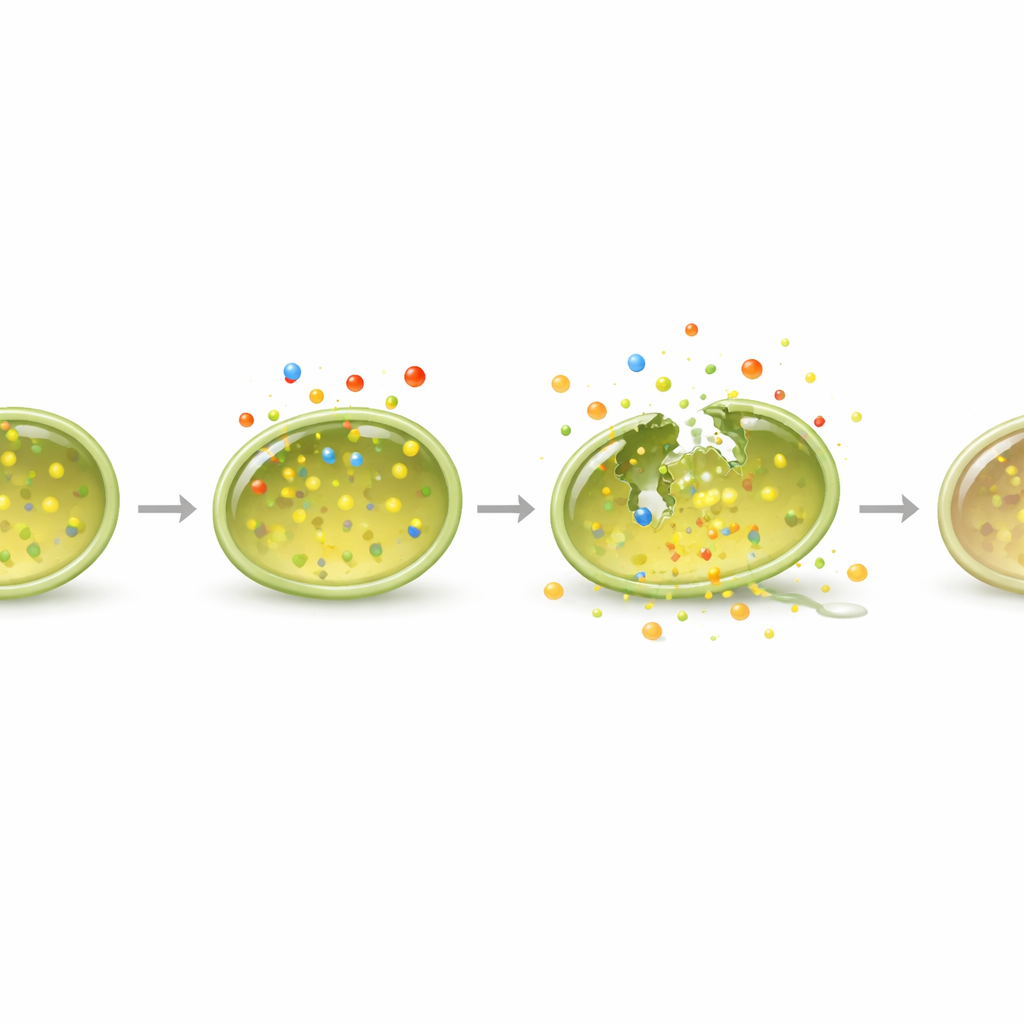
To see how 13394-2 actually kills, the team examined treated bacteria under high-powered microscopes and with fluorescent dyes. Untreated cells looked round and smooth; once exposed to the compound, many became misshapen, collapsed, and leaky. Tests that track entry of DNA-binding dyes showed that the cell envelope became porous, and live/dead staining revealed a strong shift toward dead cells. Importantly, the compound also attacked biofilms—the sticky, layered communities that let bacteria cling to surfaces and resist drugs. 13394-2 not only blocked new biofilms from forming, it also broke down mature biofilms more effectively than the standard antibiotic vancomycin, stripping away a major line of bacterial defense.
Shutting Down the Germs’ Inner Power
The damage was not limited to the surface. Inside the cells, 13394-2 triggered a surge of reactive molecules linked to oxidative stress and rapidly drained energy stores in the form of ATP. Measurements showed that the usual energy-driving force across the membrane collapsed, leaving the bacteria unable to power essential processes. To understand the deeper ripple effects, the researchers analyzed which genes turned on or off after treatment. They found sweeping changes: genes involved in building proteins and adjusting membrane fats were boosted, while central energy pathways were dialed down. Together, these shifts suggest bacteria are trying to repair themselves under stress but ultimately starve for energy and fail.
Zeroing In on a Vulnerable Part
Diving further, the team looked at a key enzyme called FabF, which helps bacteria make fatty acids—the building blocks of their membranes. Computer simulations suggested that 13394-2 fits snugly into FabF’s active pocket, forming stable bonds much like a related natural compound already known to block this enzyme. Follow-up experiments with purified FabF protein confirmed direct, tight binding and showed that the enzyme becomes more rigid and stable when the compound is present, a hallmark of specific interaction. This points to a dual attack: 13394-2 both punches holes in the membrane and likely disrupts the machinery that makes new membrane material, pushing cells toward irreversible breakdown.
From Lab Bench to Living Hosts and Everyday Life
Any potential antibiotic must not only kill bacteria but also be reasonably safe. In cultured mammalian cells, 13394-2 was only harmful at concentrations dozens of times higher than those needed to stop Staphylococcus aureus, yielding a healthy safety margin. Blood cell tests showed minimal damage, and mice given the compound did not develop signs of liver or kidney injury. In infection models, the molecule shined: it improved survival in larvae and mice with bloodstream staph infections, reduced bacterial loads in organs, and lessened tissue damage. On infected skin wounds, it both tamed the infection and sped healing. Beyond medical uses, it sharply cut bacterial contamination on plastic cookware and fresh meat, suggesting possible roles in food safety and surface disinfection.
What This Means for Everyday Health
Together, the results paint 13394-2 as a promising new type of antibiotic candidate. Unlike many existing drugs that hit a single target, this soil-derived molecule appears to damage bacterial membranes, scramble metabolism, and latch onto a crucial enzyme for building the cell’s outer barrier. It works against notoriously hard-to-treat staph strains, clears their biofilms, and shows encouraging safety in early tests. While much work remains—such as fine-tuning its structure, studying how the body handles it, and testing it in more advanced models—this study demonstrates that the search for next-generation antibiotics can still start with a scoop of soil and end with a powerful new tool against resistant infections.
Citation: Wang, C., Li, X., Zhang, Z. et al. Anthraquinones derived from soil actinomycetes combat multidrug-resistant Staphylococcus aureus. Commun Biol 9, 500 (2026). https://doi.org/10.1038/s42003-026-09739-z
Keywords: antibiotic resistance, MRSA, natural products, anthraquinones, biofilm disruption