Clear Sky Science · en
Tuning the reactivity of g-C3N4 photocatalysts using liquid phase exfoliation
Cleaner Water for a Thirsty World
Access to safe drinking water is one of the biggest health challenges of our time. Many modern chemicals, from textile dyes to stain resistant coatings, are stubborn in the environment and hard to remove with standard treatment plants. This study explores a promising way to break down such pollutants using sunlight and a more sustainable catalyst material, offering a path toward cleaner water without relying on harsh chemicals or energy hungry UV lamps.
Using Light to Break Down Pollution
Photocatalysts are materials that use light to trigger chemical reactions that can tear apart unwanted molecules. When light hits the catalyst, it can kick electrons into motion and create highly reactive species that attack pollutants in water. A long used material for this purpose is titanium dioxide, but it mainly absorbs UV light and has raised some safety concerns. The authors instead focus on graphitic carbon nitride, a metal free, layered material that can be made from common nitrogen rich compounds and can use more of the visible part of sunlight.
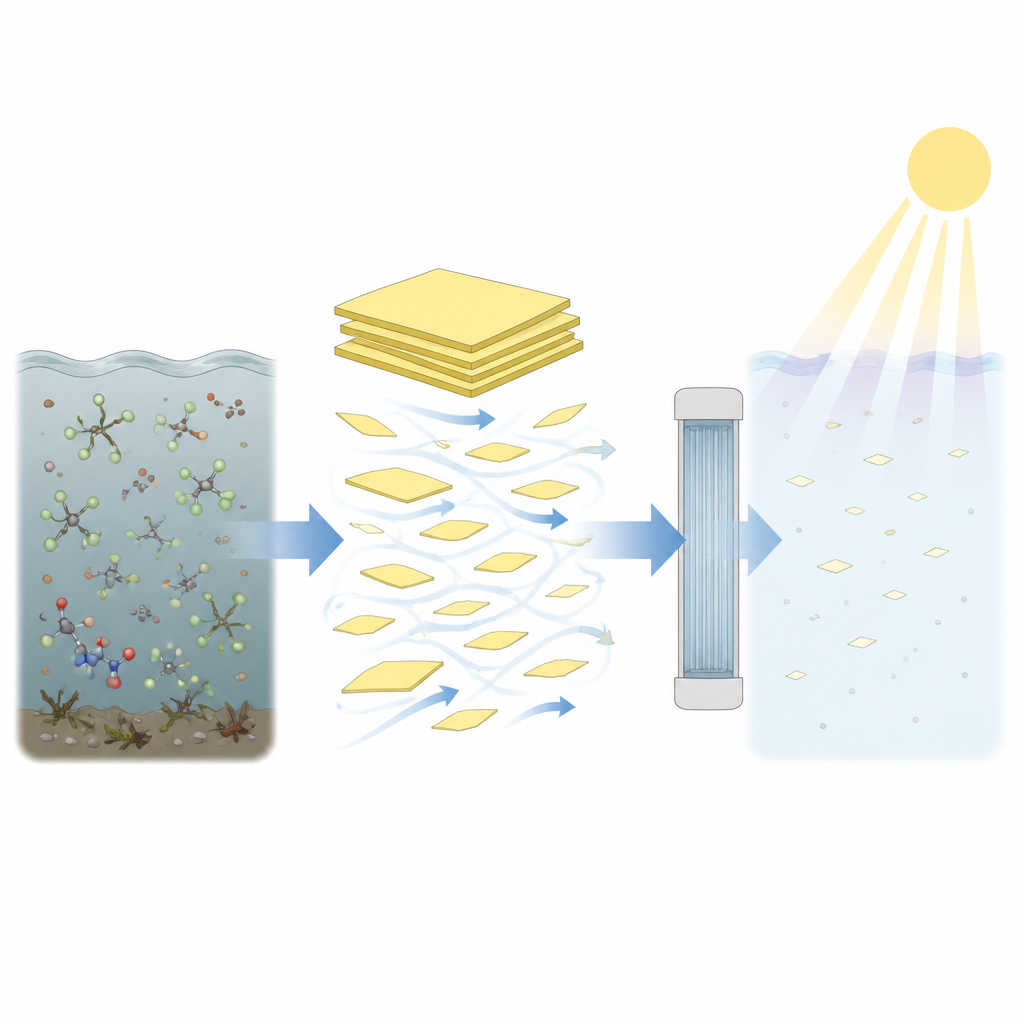
Peeling Layers to Boost Power
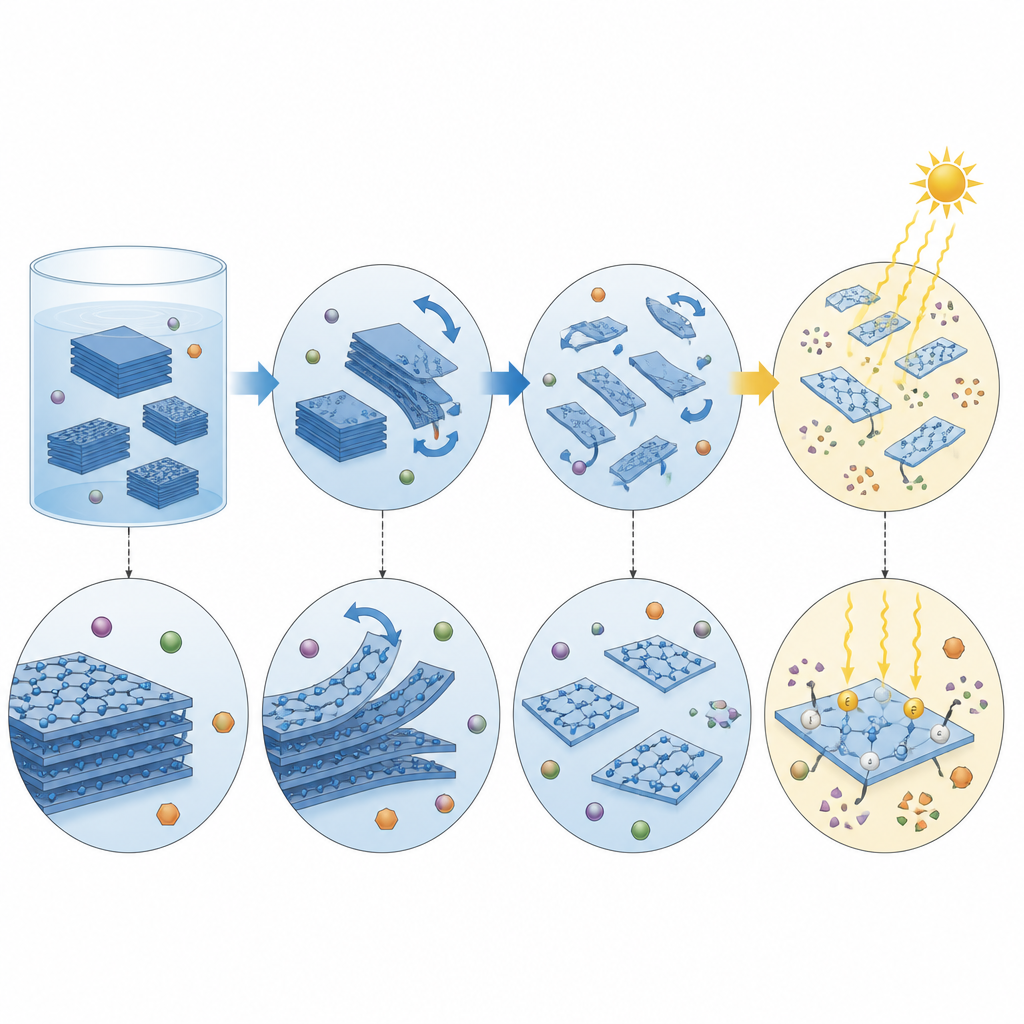
The key idea in this work is surprisingly simple: take the stacked layers of graphitic carbon nitride and peel them into thinner pieces using fast liquid stirring, a process called liquid phase exfoliation. The team compared two practical stirring tools, an industrial homogeniser and a modified kitchen blender, both of which create strong shear forces in liquids. These forces are high enough to separate the layers without shredding the internal structure of the material. By tuning the solvent mixture, they found that a blend rich in ethanol disperses the layers well, while pure water is still suitable when ethanol must be avoided.
Smaller Pieces, More Active Surface
By examining particle size, light absorption and molecular fingerprints of the material, the researchers showed what changes and what stays the same during exfoliation. Within about ten minutes, large particles on the order of tens of micrometers shrink to a few micrometers, greatly increasing surface area. Microscopy images reveal that the material breaks up mainly along its natural layers, and spectroscopic tests confirm that the basic carbon nitrogen framework and electronic structure are largely preserved. The bandgap, which sets what light the material can absorb, changes only slightly, suggesting that the main benefit of exfoliation comes from more exposed surface and shorter paths for charges to reach that surface before they recombine.
Putting the Catalysts to the Test
To see how these structural tweaks influence real performance, the team built a small flow through reactor where water containing model dyes passes over the catalyst under a 365 nm light source. Compared with the starting bulk powder, exfoliated graphitic carbon nitride removes certain dyes up to about two and a half times faster. This improvement appears after just ten minutes of shear processing, and longer times add little extra gain. The catalysts can also slowly attack very strong carbon fluorine bonds, which are a hallmark of stubborn pollutants such as some stain repellents and pesticides. While only a small fraction of fluorine was released in the tests, this shows that the material can begin to tackle some of the toughest contaminants.
Why Mixing Matters More Than Mixing Materials
The authors also explored whether combining exfoliated graphitic carbon nitride with another layered semiconductor, molybdenum disulfide, could further improve performance. These hybrid structures were successfully formed and showed clear signs of interaction between the two components. However, under the specific test conditions, they did not outperform the simply exfoliated graphitic carbon nitride alone. This suggests that, at least for the dyes and light source used here, the biggest gain comes from mechanical peeling of the main catalyst rather than from more complex pairing with a second material.
A Simple Step Toward Safer Water
In everyday terms, this study shows that giving a sustainable catalyst material a strong, carefully controlled “stir” can make it significantly better at cleaning polluted water. By peeling layered graphitic carbon nitride into thinner pieces without adding new chemicals, the researchers boost its ability to break down colorful dyes and even begin to attack some of the most resilient fluorinated bonds. The approach uses scalable, industry friendly mixing techniques and avoids harsh reagents, making it a practical step toward real world water treatment systems that harness light to deal with persistent pollutants.
Citation: Brown, J., Ramirez, I., Burt, J. et al. Tuning the reactivity of g-C3N4 photocatalysts using liquid phase exfoliation. npj 2D Mater Appl 10, 54 (2026). https://doi.org/10.1038/s41699-026-00690-5
Keywords: photocatalysis, graphitic carbon nitride, water treatment, liquid phase exfoliation, persistent pollutants