Clear Sky Science · en
Ensemble learning on serum metabolic fingerprints for early detection of lung adenocarcinoma
Why early lung cancer detection matters
Lung adenocarcinoma, the most common form of lung cancer, often grows quietly for years before causing symptoms. By the time it is found, it can already be difficult to treat. Today’s main screening tool, low-dose CT scans, frequently spots tiny lung nodules that turn out not to be cancer, leading to anxiety, repeated scans, and sometimes unnecessary surgery. This study explores whether a simple blood test, interpreted with modern machine learning, could help flag early lung cancer and distinguish dangerous growths from those that are less worrisome.
Reading cancer clues in a blood sample
The researchers focused on small molecules called metabolites that circulate in the blood and reflect how our cells use and transform energy. They collected serum samples from 199 people: healthy volunteers and patients spanning the earliest stages of lung adenocarcinoma, from pre-cancerous nodules to minimally invasive and fully invasive tumors. Using high-resolution mass spectrometry, they took an unbiased snapshot of nearly a thousand different metabolites in each sample, capturing a broad picture of how the body’s chemistry shifts as cancer develops.
How body chemistry shifts as tumors grow
When the team compared the chemical fingerprints of the four groups, clear patterns emerged. Many substances involved in bile acids, fats, amino acids, and the building blocks of DNA and RNA changed progressively from healthy individuals to those with pre-invasive lesions and then to invasive tumors. Some metabolites steadily rose, others steadily fell, and a few peaked or dipped at intermediate stages. These stepwise changes suggest that the blood chemistry of people with early lung growths is already being rewired well before the cancer becomes advanced, offering a window for earlier detection if the right signals can be captured.
Teaching algorithms to spot cancer fingerprints
Because no single metabolite tells the whole story, the researchers turned to ensemble machine learning—an approach that combines multiple prediction models into a single, more robust decision-maker. They first narrowed the list of candidate metabolites with statistical tests and a feature-selection method that prizes the strongest, least redundant signals. These selected markers were then fed into AutoGluon, a software framework that builds and blends several types of models, such as decision trees and gradient boosting machines, to classify samples as healthy or diseased and to distinguish among disease stages.
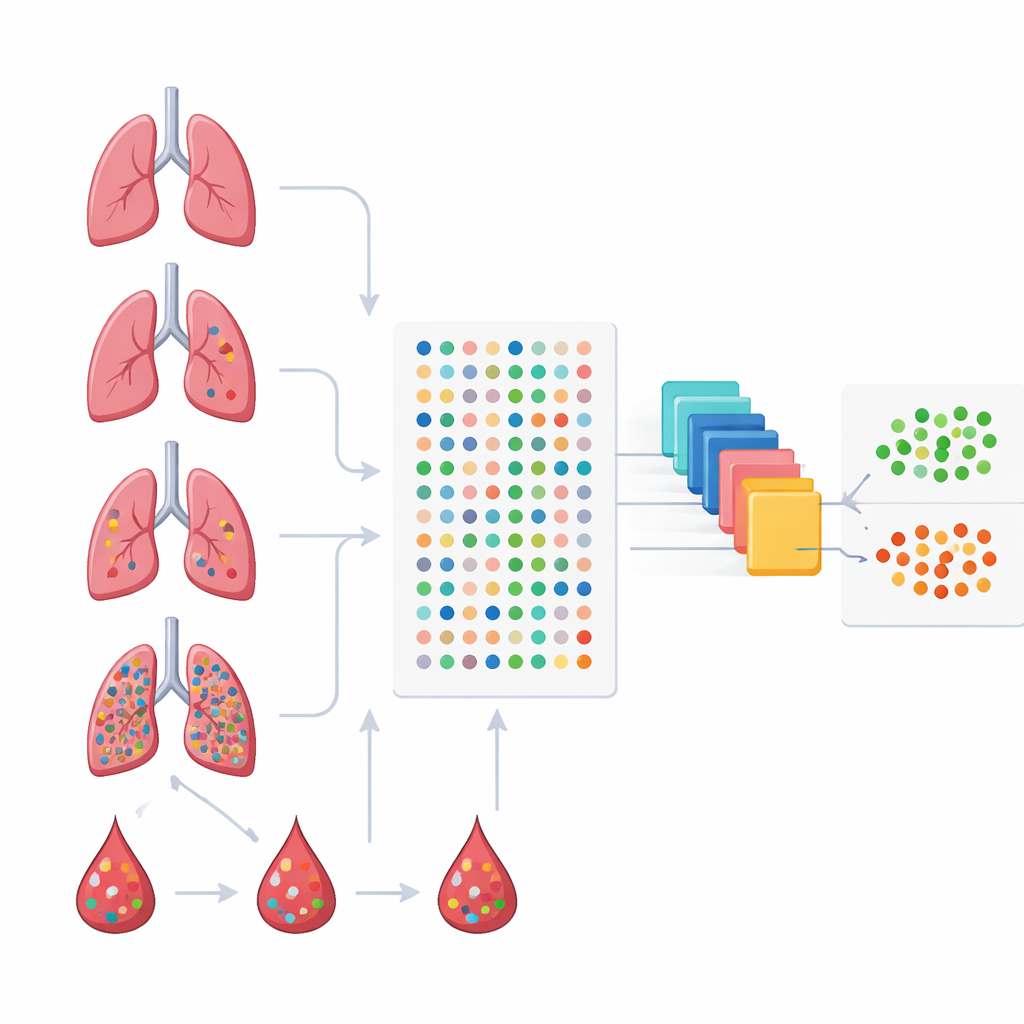
Small panels of molecules with big diagnostic power
The machine learning pipeline produced compact sets of metabolites that together carried strong diagnostic information. One panel of six molecules in blood could almost perfectly separate healthy people from all patients with lung lesions, including the earliest stage tumors, in the study cohort. Another six-molecule panel was tailored specifically to detect invasive stage I cancer, achieving accuracy on par with or better than many existing metabolite-based tests. A separate four-molecule panel distinguished pre-invasive nodules from those that had already broken through into invasive disease—an especially important clinical question because it influences whether surgeons recommend close monitoring or more aggressive operations.
What this could mean for patients
Although this work needs to be confirmed in larger and more diverse groups of patients, it points toward a future in which a routine blood draw could help identify people whose lung nodules are truly dangerous, while sparing others from unnecessary procedures. By capturing the subtle chemical shifts that accompany the earliest steps of tumor formation, and interpreting them with powerful yet interpretable algorithms, the study lays groundwork for minimally invasive tools that could complement CT scans, guide treatment choices, and ultimately catch lung adenocarcinoma when it is most curable.
Citation: Cai, C., Xu, W., Yang, S. et al. Ensemble learning on serum metabolic fingerprints for early detection of lung adenocarcinoma. npj Precis. Onc. 10, 149 (2026). https://doi.org/10.1038/s41698-026-01342-z
Keywords: lung adenocarcinoma, serum metabolomics, early cancer detection, machine learning biomarkers, noninvasive diagnosis