Clear Sky Science · en
Epigenome-wide DNA methylation patterns associated with disease activity in systemic lupus erythematosus
Why tiny DNA tags matter in lupus
Systemic lupus erythematosus (lupus) is an autoimmune disease in which the immune system turns against the body, causing fatigue, pain, and damage to organs ranging from the skin to the brain. Many patients never reach full remission, even on modern treatments, and still struggle with brain fog, mood changes, and aching joints. This study explores whether chemical tags on DNA in blood cells—marks that help control which genes are turned on or off—might be linked to how active a woman’s lupus is, and whether they can offer clues to hidden processes driving lingering symptoms.
A closer look at lupus activity in the real world
The researchers focused on 48 women with long-standing lupus being treated at a Swedish clinic. All met standard criteria for the disease, but they differed in how active their lupus was at the time of blood sampling. Using a widely used disease activity score, they grouped the women into those with no measurable activity and those with ongoing, generally mild activity. Everyone was on routine care, so the team was not comparing sick people with healthy volunteers, but rather looking for molecular differences between women whose lupus seemed quiet and those whose disease still smoldered.
Reading chemical marks on the genome
The team analyzed DNA from whole blood using a high-density array that measures hundreds of thousands of sites where methyl groups—small chemical tags—can attach along the genome. These methyl marks help tune gene activity without changing the DNA code itself. After strict quality checks, more than 700,000 sites were examined. The scientists compared the two patient groups while adjusting for age, ancestry, smoking, body weight, and the mix of blood cell types, to make sure that any differences reflected disease activity rather than simple demographic or biological variation.
Subtle but widespread differences, not a single smoking gun
No single DNA site stood out strongly enough to pass very strict statistical correction, suggesting that lupus activity is not driven by one dramatic on–off switch in blood cells. Instead, thousands of sites showed modest differences between women with and without detectable disease activity. When the researchers looked at stretches of DNA containing several neighboring sites that shifted together, they uncovered 36 regions with consistent changes. Many of these regions lay within genes involved in immune defenses, cell death, and the delicate dialogue between the nervous and immune systems. One intriguing pattern was the enrichment of a binding motif for REST, a protein best known for silencing nerve-related genes in non-nerve tissues, hinting that gene networks usually associated with the brain may be unusually regulated in immune cells during active lupus.
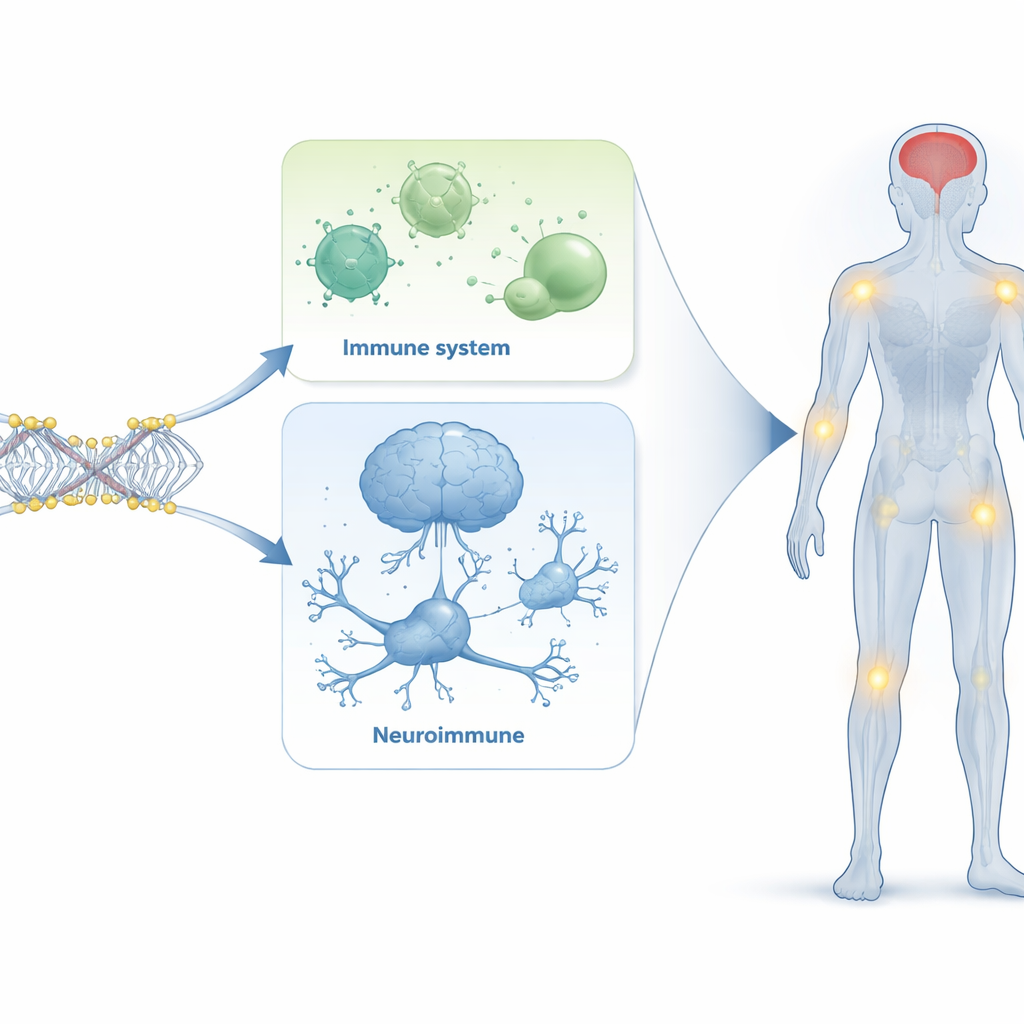
Links between immune signals, the brain, and lasting symptoms
Several of the altered regions mapped to genes tied to inflammation and antiviral responses, echoing the long-recognized role of overactive immune pathways in lupus. Others were associated with nerve cell communication and brain function, including genes related to mood, memory, and seizure disorders. Because these findings come from blood, they do not prove that the same changes occur in the brain itself. However, they support the idea that the immune system and nervous system are tightly intertwined in lupus, and that epigenetic shifts in blood cells may mirror or influence pathways connected to cognitive problems, anxiety, and fatigue that persist even when traditional measures of inflammation look controlled.
What this work means for people living with lupus
This study does not deliver a ready-to-use blood test for lupus activity, nor does it pinpoint a single new drug target. Instead, it offers early evidence that disease activity is linked to subtle, coordinated changes in DNA methylation across networks of immune and neuroimmune genes. For patients, that means researchers are beginning to map the molecular fingerprints of flares and low-level activity, including pathways that might explain why the brain and mood are affected. Larger, long-term studies in more diverse and precisely defined groups of patients will be needed to confirm these patterns and test whether tracking such epigenetic marks could one day help tailor treatment or predict who is at risk for lingering symptoms.
Citation: Ravaei, A., Fatima, T., Wincup, C. et al. Epigenome-wide DNA methylation patterns associated with disease activity in systemic lupus erythematosus. Sci Rep 16, 14287 (2026). https://doi.org/10.1038/s41598-026-51708-3
Keywords: systemic lupus erythematosus, DNA methylation, epigenetics, autoimmune disease, neuroimmune pathways