Clear Sky Science · en
Validation of circulating miR-323a-3p and miR-625-3p to classify hypertrophic cardiomyopathy in Friedreich’s ataxia
Why tiny blood signals matter for a rare heart problem
Friedreich’s ataxia is a rare inherited disease that usually begins in childhood, affecting movement and balance. Many people with this condition also develop a serious thickening of the heart muscle called hypertrophic cardiomyopathy, which is a leading cause of early death in these patients. Doctors can see heart changes with ultrasound scans and can measure heart damage proteins in the blood, but these tools often detect trouble only after the heart has already remodeled. This study asks whether very small molecules circulating in the blood can flag heart risk earlier and more accurately.
Looking for early warning signs in the blood
The research team focused on microRNAs, short pieces of genetic material that help control how cells behave and that can be measured in blood. Earlier work from the same group had identified seven microRNAs that seemed to differ between people with Friedreich’s ataxia and healthy volunteers. In this new study, they set out to test that signal in a fresh group of 34 patients and 34 matched healthy controls. They also carefully reviewed medical records and heart ultrasound data to see which patients had developed thickened heart muscle and which had not, and whether they also had diabetes, another common problem in this disease.
Confirming a blood fingerprint of the disease
When the team re-measured the seven candidate microRNAs in the new group, five of them were clearly higher in patients than in healthy people. This suggests that these microRNAs form a repeatable blood fingerprint of Friedreich’s ataxia, not just a one-time finding. Several of these molecules were also higher in patients who had diabetes compared with those who did not, hinting that the same blood signals might track both nerve damage and problems with sugar control. These results support the idea that microRNAs in blood can reflect the wide-ranging effects of this condition throughout the body.
Linking blood markers to heart thickening
The most striking results involved two particular microRNAs, called miR-323a-3p and miR-625-3p. Among Friedreich’s ataxia patients, those who already had hypertrophic cardiomyopathy showed higher levels of miR-323a-3p and lower levels of miR-625-3p in their blood. The researchers then compared how well these molecules and standard heart ultrasound measures could separate patients with thickened hearts from those without. Each of the two microRNAs, as well as certain wall thickness measurements, showed useful accuracy on its own. However, when the levels of miR-323a-3p and miR-625-3p were combined in a simple statistical model, the ability to classify patients improved, with better sensitivity and specificity than traditional heart blood proteins reported in earlier studies.
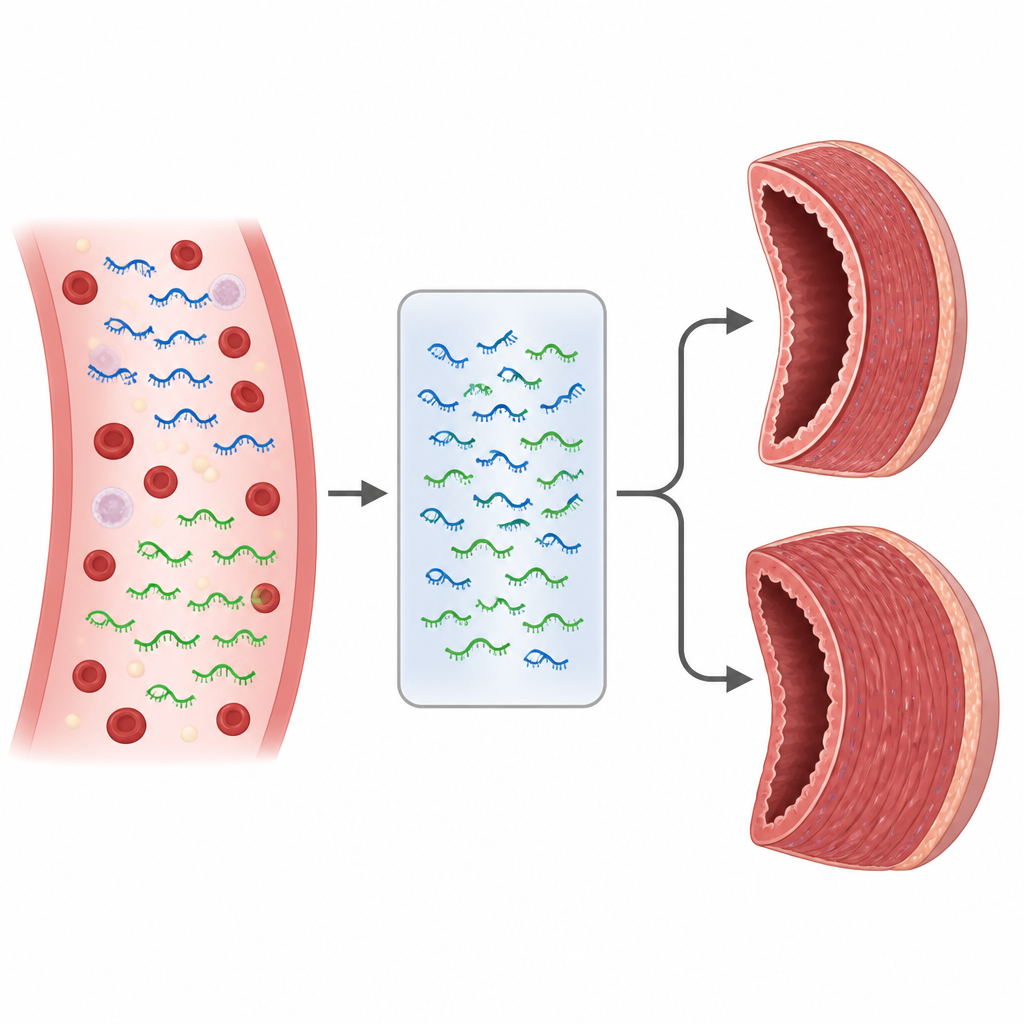
Clues to how heart scarring might develop
Beyond diagnosis, the patterns of these microRNAs also offer clues about what is happening inside the heart. Previous laboratory work suggests that miR-323a-3p can drive the build-up of collagen and scarring in heart tissue by disturbing natural braking systems that limit stiffening and overgrowth. In contrast, miR-625-3p appears to dampen certain growth signals. The combination of increased miR-323a-3p and reduced miR-625-3p seen in affected patients could therefore tilt the balance toward thicker, stiffer heart muscle, a hallmark of the cardiomyopathy in Friedreich’s ataxia. Although this study did not directly examine heart tissue, the matching blood patterns fit with this emerging picture.
What this means for patients and care
Overall, the study shows that a pair of tiny blood signals can help distinguish Friedreich’s ataxia patients with heart thickening from those without, and may do so more effectively than current blood tests. If confirmed in larger and more diverse groups over time, a two-microRNA blood test could allow doctors to sort patients into higher and lower cardiac risk categories and tailor how often they need heart imaging and follow-up. For families facing this lifelong condition, such a simple blood draw could eventually offer earlier warning of heart trouble and support more personalized monitoring, even though more work is needed before it becomes part of routine care.
Citation: Ibáñez-Cabellos, J.S., Baviera-Muñoz, R., Alemany-Perna, B. et al. Validation of circulating miR-323a-3p and miR-625-3p to classify hypertrophic cardiomyopathy in Friedreich’s ataxia. Sci Rep 16, 15056 (2026). https://doi.org/10.1038/s41598-026-50975-4
Keywords: Friedreich’s ataxia, hypertrophic cardiomyopathy, microRNA biomarkers, cardiac imaging, diabetes comorbidity