Clear Sky Science · en
Design, synthesis, and insecticidal potency of novel 3-methyl-pyrazole derivatives against Culex pipiens larvae
Why new mosquito killers matter
Mosquitoes are more than a backyard nuisance: they spread viruses and parasites that threaten both human and animal health. One widespread species, the common house mosquito Culex pipiens, helps transmit West Nile virus, bird malaria, and other infections, and it can even contaminate raw milk with harmful bacteria. Yet the chemical sprays and larvicides we rely on are losing their punch as mosquitoes evolve resistance. This study explores a fresh family of lab‑made molecules aimed specifically at killing mosquito larvae before they grow into biting adults, with the long‑term goal of safer and more effective control tools.
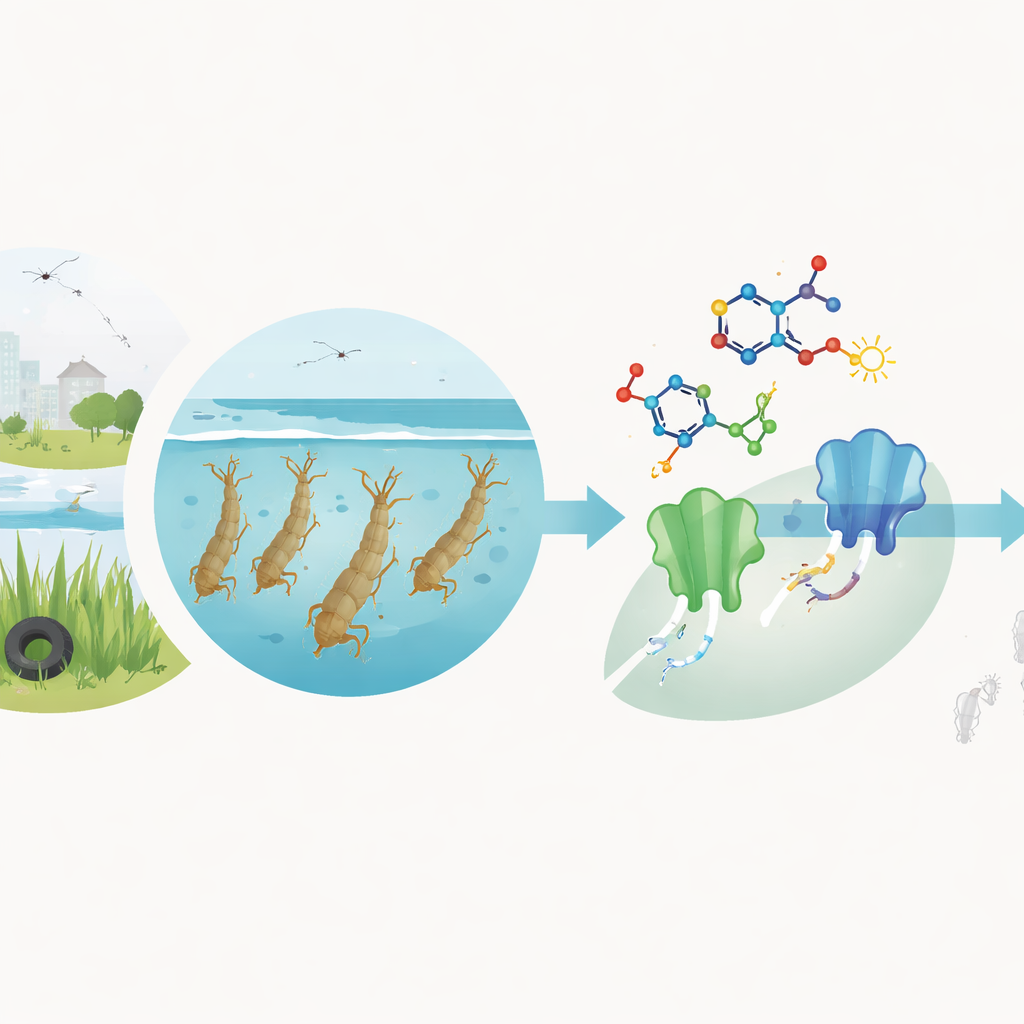
Building new weapons in the lab
The research team designed and synthesized nineteen different compounds that all share a small ring‑shaped chemical core known as a 3‑methyl‑pyrazole. Around this core, they systematically attached various extra groups, such as sulfur‑containing fragments, aromatic rings, and strongly pulling or donating substituents. These changes were not random tinkering: each was chosen because similar features are found in successful commercial insecticides. The compounds were carefully characterized using standard analytical methods to confirm their structures and purity, creating a focused “library” of candidates for biological testing.
Putting larvae to the test
To find out how well these new molecules work, the scientists exposed lab‑reared Culex pipiens larvae to a range of doses, following World Health Organization guidelines. They compared survival after 24 hours and calculated the concentration needed to kill half the larvae (LC50) for each compound. Two derivatives, labeled 7 and 12, stood out dramatically. They were active at a fraction of a microgram per milliliter—hundreds of times more potent than chlorpyrifos, a widely used reference insecticide tested side‑by‑side. Several other molecules showed moderate effects, but none matched the strength of these top performers, highlighting how small tweaks in chemical structure can make the difference between weak and powerful larvicides.
Targeting the mosquito nervous system
The next question was how these molecules kill. Observations of the larvae revealed twitching, hyperactivity, loss of coordination, and final paralysis—classic signs of nerve disruption. Guided by this, the team focused on two key players in mosquito nerve signaling: an enzyme that breaks down the messenger acetylcholine, and a receptor that responds to that messenger and opens an ion channel in nerve cells. Using computer‑based docking studies, they virtually fitted all nineteen molecules into three‑dimensional models of these targets. Compounds 7 and 12 nestled into the same regions used by known insecticides, forming a dense web of hydrogen bonds and other stabilizing contacts, often matching or exceeding the interactions predicted for chlorpyrifos and several modern neonicotinoid products.
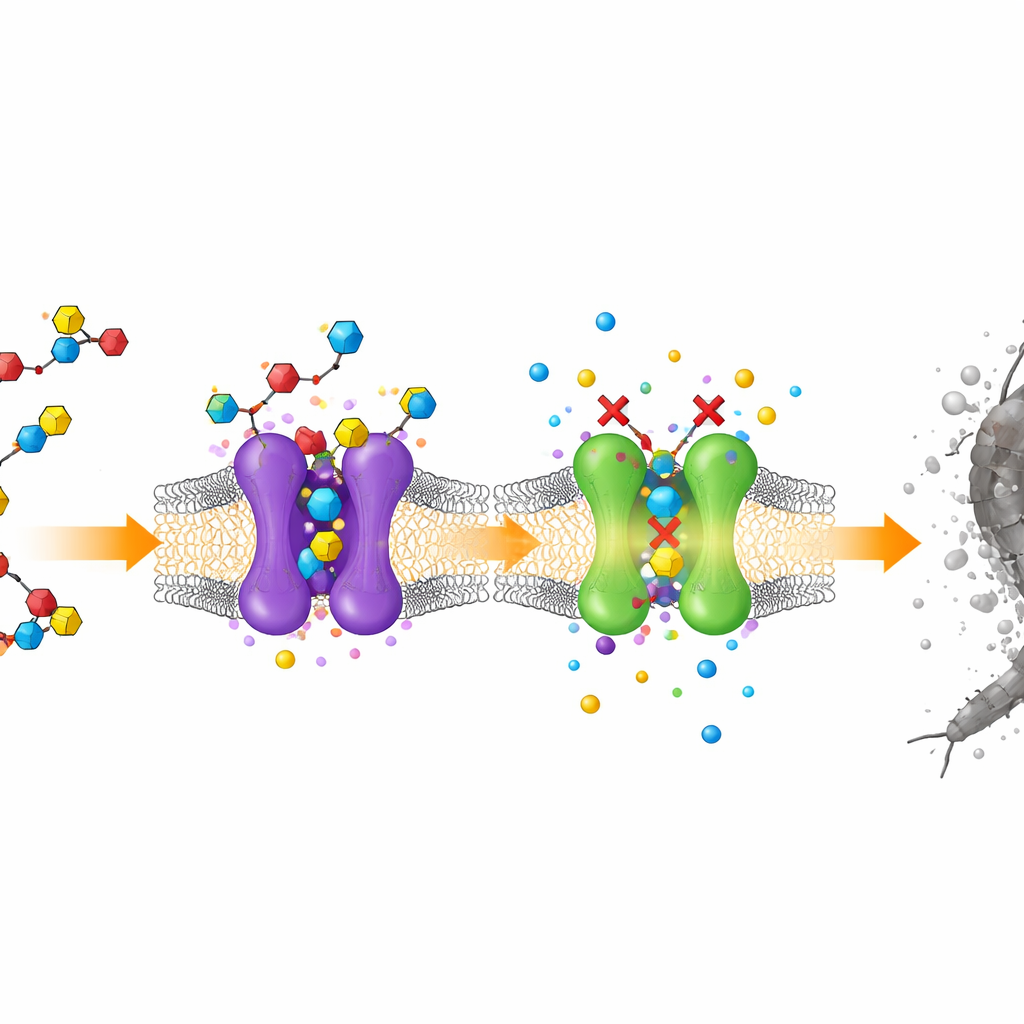
Watching molecules move in real time
Docking snapshots show only a frozen moment, so the researchers went further with molecular dynamics simulations, which track how atoms move over time in a virtual water‑filled environment. They followed the behavior of compounds 7 and 12 bound to the mosquito enzyme for 100 billionths of a second and compared them with chlorpyrifos under the same conditions. The enzyme structure remained stable, and the new molecules stayed snugly lodged in the active site, maintaining many of their key contacts. In contrast, the reference insecticide showed more fluctuations and fewer long‑lasting interactions. These simulations support the idea that the new compounds not only fit well initially, but also remain tightly bound long enough to effectively block the enzyme’s role in nerve signaling.
What this means for future mosquito control
Taken together, the chemistry, larval tests, and computer models paint a consistent picture: carefully tuned 3‑methyl‑pyrazole derivatives—especially compounds 7 and 12—are extremely powerful killers of Culex pipiens larvae, likely by jamming critical steps in their nervous system. While this work is still at an early stage, it outlines a roadmap for designing next‑generation larvicides that could work at very low doses and help overcome existing resistance. Before any field use, however, these molecules must be checked for safety in non‑target species, tested directly on the target enzymes, and evaluated against other important mosquito vectors like Aedes and Anopheles. If those hurdles are cleared, this new chemical family could become an important part of an integrated, more sustainable strategy to curb mosquito‑borne disease.
Citation: Nofal, H.R., Ali, A.K., Ismail, M.F. et al. Design, synthesis, and insecticidal potency of novel 3-methyl-pyrazole derivatives against Culex pipiens larvae. Sci Rep 16, 14699 (2026). https://doi.org/10.1038/s41598-026-50895-3
Keywords: mosquito control, Culex pipiens, larvicide, acetylcholinesterase inhibition, insecticide resistance