Clear Sky Science · en
Evaluation of functionalized graphene oxide as a nanostructured sensor for lead ion detection in aqueous solutions via quartz crystal microbalance
Why cleaner water needs smarter tools
Lead in drinking water is a silent hazard that cannot be seen, smelled, or tasted, yet it can harm the brain, blood, liver, and kidneys even at low levels. Governments set strict limits for how much lead is allowed, but current lab tests often require expensive machines, trained staff, and a trip to a central facility. This study explores a simpler, portable sensor that could help track lead in water more easily and cheaply, supporting safer tap water in homes, schools, and factories.
A tiny crystal that can feel extra weight
At the heart of the new device is a quartz crystal that vibrates at a precise frequency, much like a very stable tuning fork. When a small amount of material sticks to its surface, the crystal slows down slightly, and this tiny shift can be measured with high accuracy. This type of device is called a quartz crystal microbalance, and it is already used in many research labs. The challenge here is to cover the crystal with a special coating that catches lead ions from water while largely ignoring other metals.
Building a sticky surface for lead
The researchers started with graphene oxide, a sheet-like form of carbon that is thin, strong, and easy to modify. They attached molecules that carry sulfur and silicon to its surface so that the coating would have many tiny hooks that attract lead. Careful tests using microscopes, light-based methods, and surface measurements showed that the modified material became rougher, more water-friendly, and carried a stable negative charge. All of these features help expose more active spots to the water, making it easier for lead ions to reach and bind to the surface.
How the sensor behaves in different waters
To test the sensor, the team flowed water containing known amounts of lead over the coated crystal under controlled conditions. As lead stuck to the surface, the vibration frequency dropped in real time, allowing the team to track how quickly and how strongly the ions were captured. The sensor worked across a range of water acidity, with the strongest overall response in alkaline water and reliable performance in neutral water that matches typical drinking water. In neutral conditions, it could detect lead at levels as low as the limit recommended by the World Health Organization, and its response increased in a straight line with rising lead concentration over the tested range.
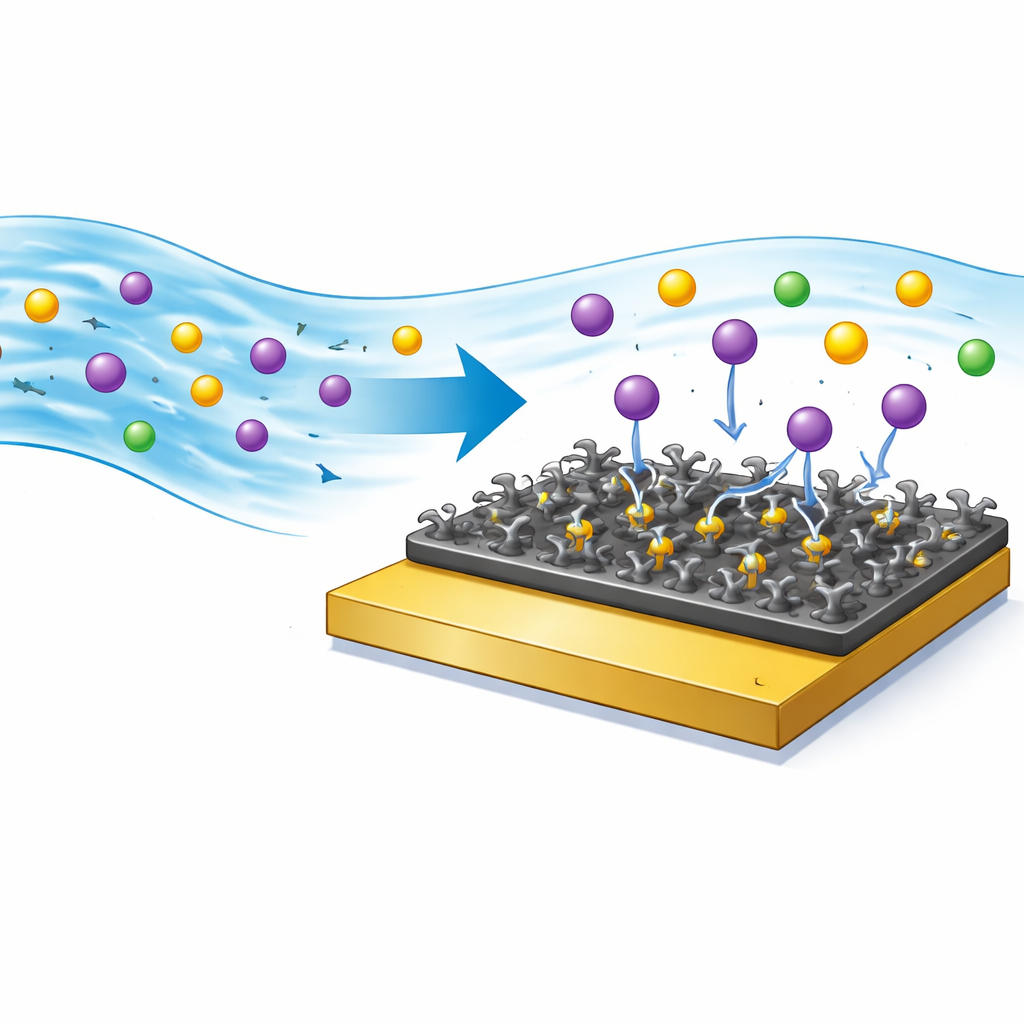
Picking out lead among many metals
Real water rarely contains a single metal, so the team challenged the sensor with other common ions such as zinc, copper, calcium, magnesium, and iron. Even when these were present in higher amounts than lead, the sensor signal dropped far more when lead was in the mix, showing that the sulfur-bearing sites on the surface have a special pull on lead ions. Additional tests on real samples from industrial wastewater, groundwater, and tap water showed that the sensor could still recover lead levels accurately when compared with a standard laboratory method. After each use, the surface could be cleaned with a common chemical treatment, restoring most of its original performance over at least ten cycles.
What this means for everyday water checks
Overall, the study shows that combining a vibrating quartz crystal with a carefully tailored graphene-based coating can create a sensitive and selective lead sensor that uses simple hardware and small amounts of material. While this is still an early proof-of-concept that needs longer-term testing and wider trials outside the lab, it points toward compact devices that could monitor lead in water supplies more often and closer to where people actually drink. In time, such tools could complement traditional lab tests and make continuous water-quality checks more practical and affordable.
Citation: Kilany, H.A., Elsherif, R.M. & Gawad, S.A.A. Evaluation of functionalized graphene oxide as a nanostructured sensor for lead ion detection in aqueous solutions via quartz crystal microbalance. Sci Rep 16, 14707 (2026). https://doi.org/10.1038/s41598-026-50889-1
Keywords: lead in drinking water, graphene oxide sensor, quartz crystal microbalance, heavy metal detection, water quality monitoring