Clear Sky Science · en
Ginger phytochemical corona enhances hemocompatibility of metal oxide nanoparticles for blood-contacting applications
Spicing Up Safer Nanomedicine
Modern medicine increasingly relies on tiny particles made of metal oxides to carry drugs, fight infections, or help with imaging. But when these nanoparticles meet blood, they can damage cells, trigger clots, or stir up the immune system. This study explores an everyday kitchen ingredient—ginger—as a natural way to coat such particles, making them gentler and safer for use in blood-contacting medical devices and treatments.
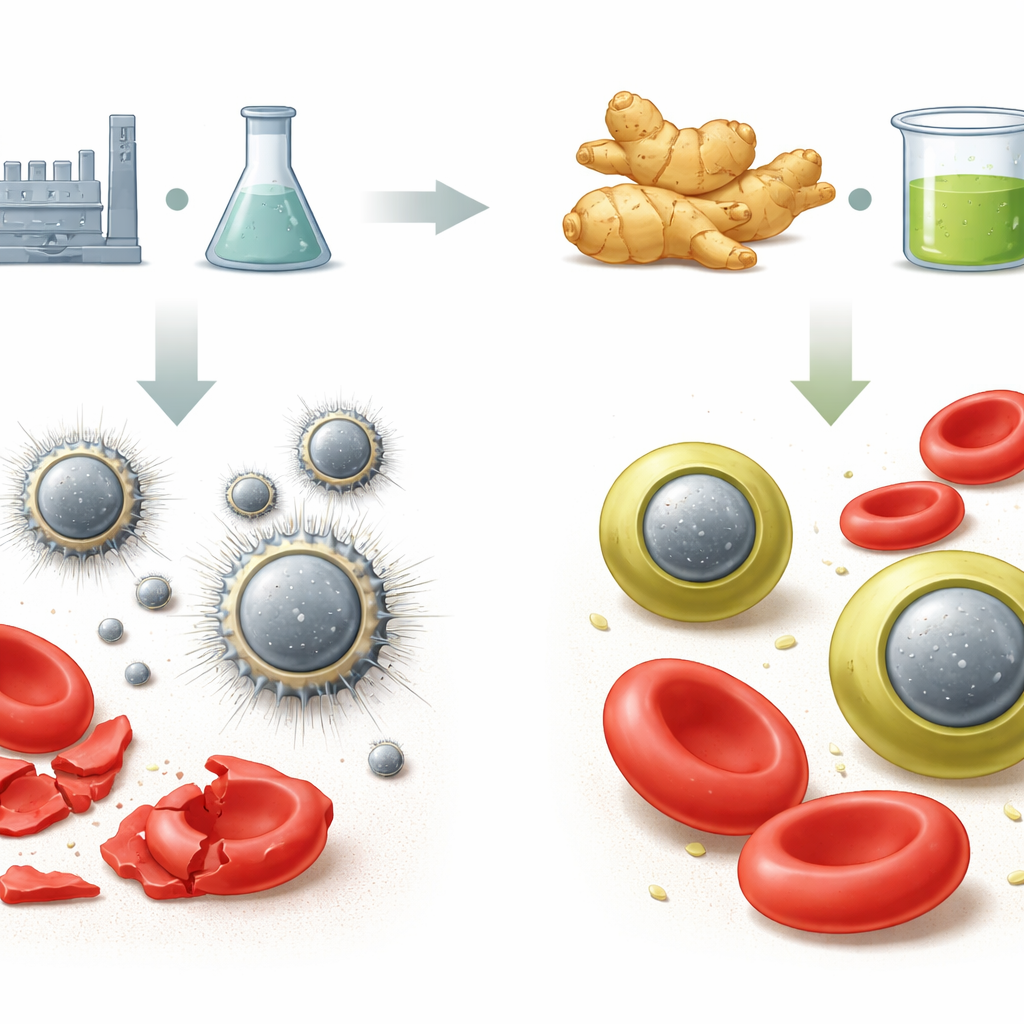
Why Tiny Particles Need a Gentle Touch
Metal oxide nanoparticles such as titanium dioxide, zinc oxide, magnesium oxide, and calcium oxide are attractive tools because of their small size and special surface properties. Unfortunately, when made by standard chemical methods, they can be harsh on blood. Their bare, highly reactive surfaces can puncture red blood cells, generate harmful oxidants, and bind blood proteins in ways that attract the immune system. For these particles to be used safely in intravenous drugs, coatings, or sensors, they must be “hemocompatible”—that is, able to circulate in blood without causing harm.
Turning Ginger into a Protective Coat
The researchers prepared two versions of each metal oxide: one by a usual laboratory precipitation method, and another using a water extract of ginger rhizomes as the reducing and stabilizing agent. This “green” route wrapped the particles in a thin layer of ginger phytochemicals—a so‑called phytochemical corona—rich in antioxidant phenolic compounds like gingerols and shogaols. Detailed measurements confirmed that this natural layer clung firmly to the nanoparticle surface, slightly increasing particle size, improving dispersion, and changing the way the particles interacted with light and with surrounding molecules.
How Ginger Calms Blood Reactions
Once any nanoparticle enters blood, proteins from plasma quickly adsorb to its surface to form a “protein corona” that the body actually “sees.” Chemically made particles developed thick, messy coronas stuffed with proteins that flag foreign objects to the immune system and promote clot formation. In contrast, ginger-coated particles attracted much thinner coronas and far fewer of these “opsonin” proteins. Remarkably, they became enriched in apolipoprotein A‑I, a major component of “good” HDL cholesterol that tends to quiet immune reactions and prolong circulation. At the same time, the ginger layer cut the buildup of reactive oxygen species inside red and white blood cells by more than half, greatly easing oxidative stress.
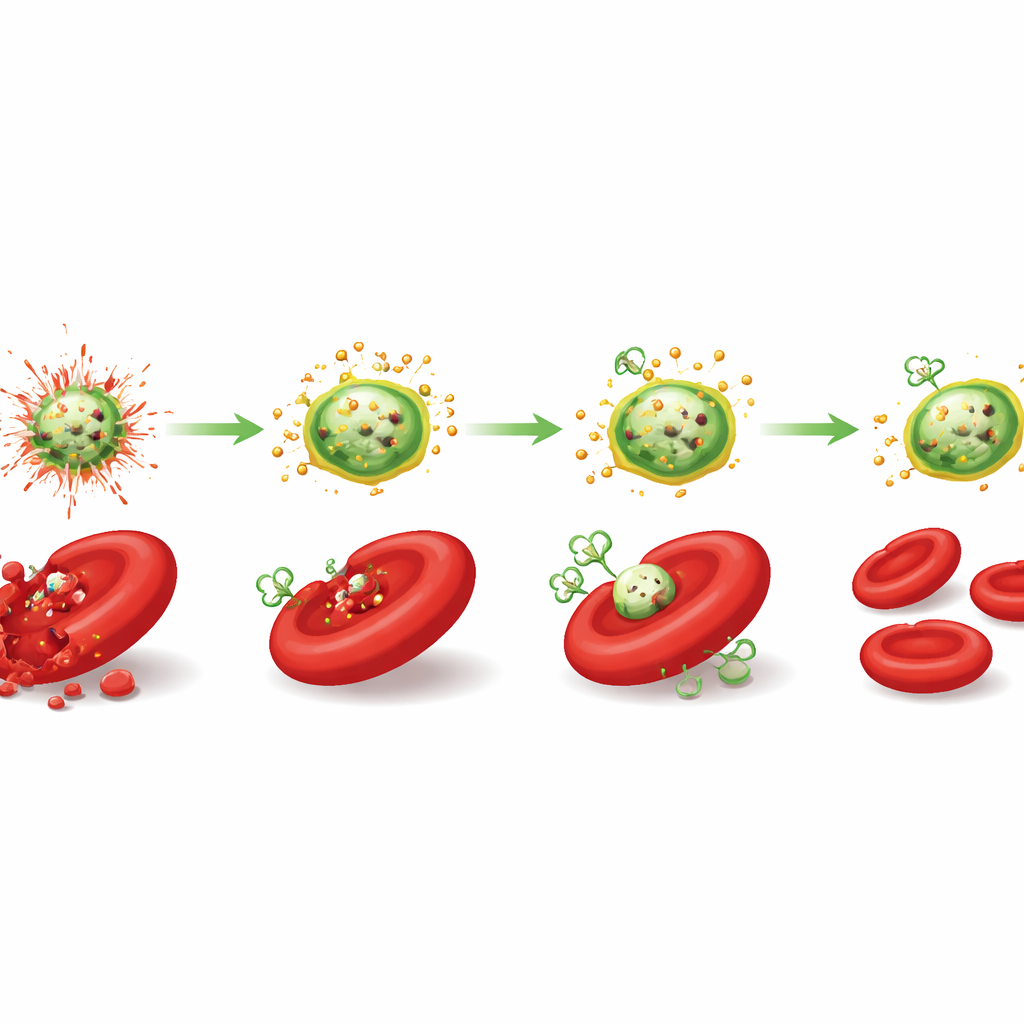
Protecting Blood Cells and Clotting Balance
In tests with human blood, the difference between coated and uncoated particles was striking. Bare metal oxide nanoparticles, especially zinc oxide, caused dose‑dependent rupture of red blood cells, visible changes in their normal doughnut-like shape, increased clumping (seen as a higher sedimentation rate), and reduced survival of key immune cells called peripheral blood mononuclear cells. Ginger-coated versions stayed largely harmless even at several times higher doses: they caused less than 2% red blood cell rupture, preserved over 92% normal cell shapes, and kept immune cell viability above 93% at the highest tested concentrations. Effects on blood clotting times also shifted toward a safer, more neutral profile for the ginger-coated zinc and magnesium particles, meaning they neither strongly promoted nor strongly blocked clot formation.
Four Simple Ways Ginger Helps
Putting all the experiments together, the authors propose four mutually reinforcing ways in which ginger makes these nanoparticles blood‑friendly. First, the organic shell acts as a physical cushion, keeping the hard inorganic core from scraping and piercing cell membranes. Second, chemical groups on the ginger compounds can grab and hold metal ions, slowing their release into the surrounding fluid. Third, the same phenolic groups neutralize reactive oxygen species before they can damage lipids, proteins, or DNA. Fourth, by tuning which blood proteins stick to the surface, the ginger coat encourages formation of a “stealth” protein corona that avoids immune attack.
What This Means for Future Treatments
For patients, the key outcome is a wider safety margin: the maximum “safe” concentration of these nanoparticles in contact with blood jumped from below 125 micrograms per milliliter for the chemically made versions to more than 500 micrograms per milliliter for the ginger-coated ones. Among the four metals, ginger-coated zinc oxide showed the greatest transformation—from clearly damaging to essentially gentle. While this work was done in laboratory settings rather than in living organisms, it suggests that simple plant extracts like ginger could offer a low-cost, sustainable way to tame otherwise aggressive nanomaterials, paving the way for safer drug carriers, coatings on medical implants, and other technologies that must coexist peacefully with our blood.
Citation: Said, A.H., Ebnalwaled, A.A., Samir, M. et al. Ginger phytochemical corona enhances hemocompatibility of metal oxide nanoparticles for blood-contacting applications. Sci Rep 16, 14692 (2026). https://doi.org/10.1038/s41598-026-50697-7
Keywords: green nanotechnology, ginger-coated nanoparticles, blood compatibility, metal oxide nanomedicine, protein corona