Clear Sky Science · en
Isotherm, kinetics, and optimization modeling of Cr(VI) ions and methylene blue dye adsorption from water by an aminobiochar hydrogel
Turning Fruit Waste into a Water Cleaner
Every glass of water we drink can carry invisible hitchhikers—metal ions from industry and brightly colored dye molecules from textiles. Getting these pollutants out is essential, but many current treatments are costly or create new waste. This study explores an inventive solution: transforming discarded orange peels into a soft, sponge‑like material that can soak up dangerous chromium and a common blue dye from water with remarkable efficiency.

From Orange Peels to a Smart Sponge
The researchers began with a simple idea: use an abundant agricultural waste, orange peels, as the starting point for an advanced water‑cleaning material. The peels were first converted into a charcoal‑like substance called biochar using a household‑style microwave and sulfuric acid to activate the surface. Further chemical steps added oxygen‑ and nitrogen‑rich groups, giving the particles “sticky spots” that can latch onto different kinds of pollutants. Finally, these modified biochar particles were locked into a jelly‑like network made from common polymers, forming an aminobiochar hydrogel—essentially a flexible, water‑swollen sponge filled with active carbon.
Capturing Color and Metal in the Same Net
The team tested the hydrogel using two very different contaminants often found together in industrial wastewater: methylene blue, a bright cationic dye, and hexavalent chromium, a highly toxic form of a heavy metal. By carefully adjusting conditions such as pH, contact time, and the amount of hydrogel used, they showed that the material could reach extremely high uptake capacities—up to about 476 milligrams of dye and an impressive 1250 milligrams of chromium per gram of hydrogel under optimal conditions. These values are higher than many previously reported biochar or hydrogel adsorbents, highlighting that the combination of porous biochar and hydrogel network creates an unusually powerful trap for pollutants.
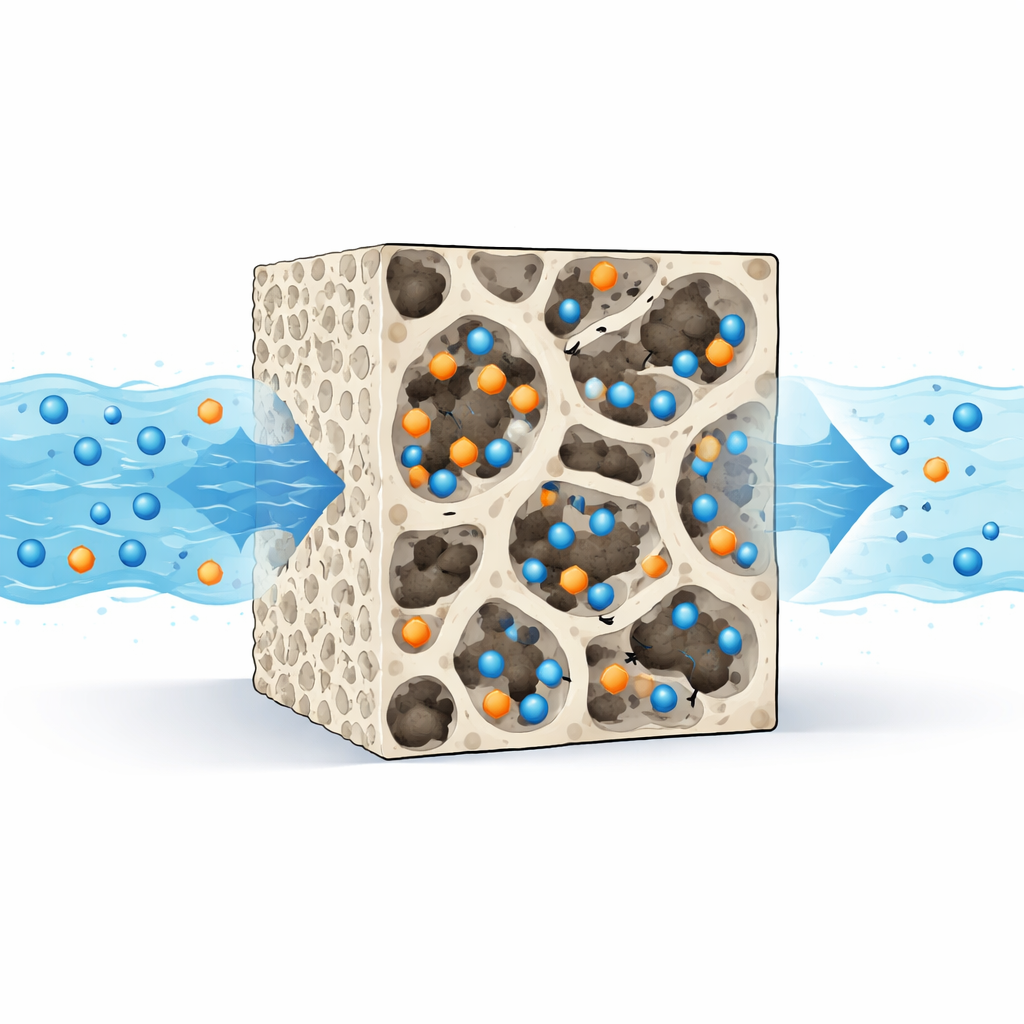
How the Hydrogel Grabs Pollutants
To understand how this works, the researchers probed both the structure and behavior of the new material. Electron microscopy revealed a rough, porous surface, while infrared spectroscopy confirmed the presence of functional groups such as amines, hydroxyls, and carboxyls. These groups control how the hydrogel interacts with different pollutants. The dye, which carries a positive charge, is attracted mainly to negatively charged sites on the hydrogel surface and forms relatively strong chemical‑like bonds; its uptake followed a kinetic pattern known as pseudo‑second‑order, consistent with such interactions. Chromium behaves differently: in acidic water it exists as negatively charged species that are drawn to positively charged sites on the hydrogel, and its uptake followed a pseudo‑first‑order pattern more typical of weaker, physical binding. In both cases, the data indicated that pollutants form a single, dense layer on the surface rather than piling up in many layers.
Finding the Best Operating Conditions
Beyond basic testing, the study used advanced modeling tools to tune performance. A statistical method called response surface methodology varied three key factors—the starting concentration of pollutants, the dose of hydrogel, and the contact time—in a systematic way to find combinations that maximize removal. In parallel, artificial neural networks, inspired by the way brain cells process information, were trained on the experimental data to predict removal efficiency under new conditions. Both approaches agreed on the sweet spots: relatively low pollutant concentrations, sufficient hydrogel dosage, and adequate contact time led to removal percentages above 90 percent for the dye and strongly improved chromium capture, while keeping material use and treatment time practical.
Reusable Material for Cleaner Water
For any real‑world water treatment, an adsorbent must be reusable. The orange‑peel hydrogel passed this test as well: after pollutants were stripped off using simple acid or base rinses, the same material could be cycled through at least six rounds of adsorption and regeneration with only slight performance loss. Taken together, the results show that a low‑cost waste product can be upgraded into a high‑value, regenerable filter for both colored dyes and toxic metals. For non‑specialists, the takeaway is clear: with clever chemistry and careful modeling, everyday food waste like orange peels can be converted into advanced materials that help safeguard water supplies and reduce pressure on landfills.
Citation: Mousa, O.F., Yılmaz, M., El-Nemr, M.A. et al. Isotherm, kinetics, and optimization modeling of Cr(VI) ions and methylene blue dye adsorption from water by an aminobiochar hydrogel. Sci Rep 16, 14172 (2026). https://doi.org/10.1038/s41598-026-49810-7
Keywords: wastewater treatment, biochar hydrogel, chromium removal, dye adsorption, orange peel recycling