Clear Sky Science · en
Electrochemical and quantum chemical exploration on L-ornithine L-aspartate as a sustainable corrosion inhibitor for AISI 1018 steel in acidic environment
Turning Waste Pills into Metal Protection
Everyday products from cars to chemical tanks rely on steel, but when that steel meets strong acids, it can quickly rust away, costing industries trillions of dollars worldwide. This study explores an unexpected way to protect steel: by reusing expired medicine. The researchers show that an out‑of‑date nutritional drug, L‑ornithine L‑aspartate, can be transformed from pharmaceutical waste into a powerful shield that keeps steel safe in harsh acidic conditions.
Why Acid Eats Metal So Quickly
Steel structures used in refineries, pipelines, and chemical processing plants are often in contact with acidic liquids such as hydrochloric acid. Without protection, the acid strips metal atoms from the surface, leaving pits, cracks, and eventually holes. Traditional chemical additives can slow this attack, but many are toxic, expensive, or work poorly in very strong acids. The team behind this work set out to find a cleaner, cheaper option that also deals with another growing problem: what to do with the tons of expired drugs that pharmacies and hospitals must discard each year.
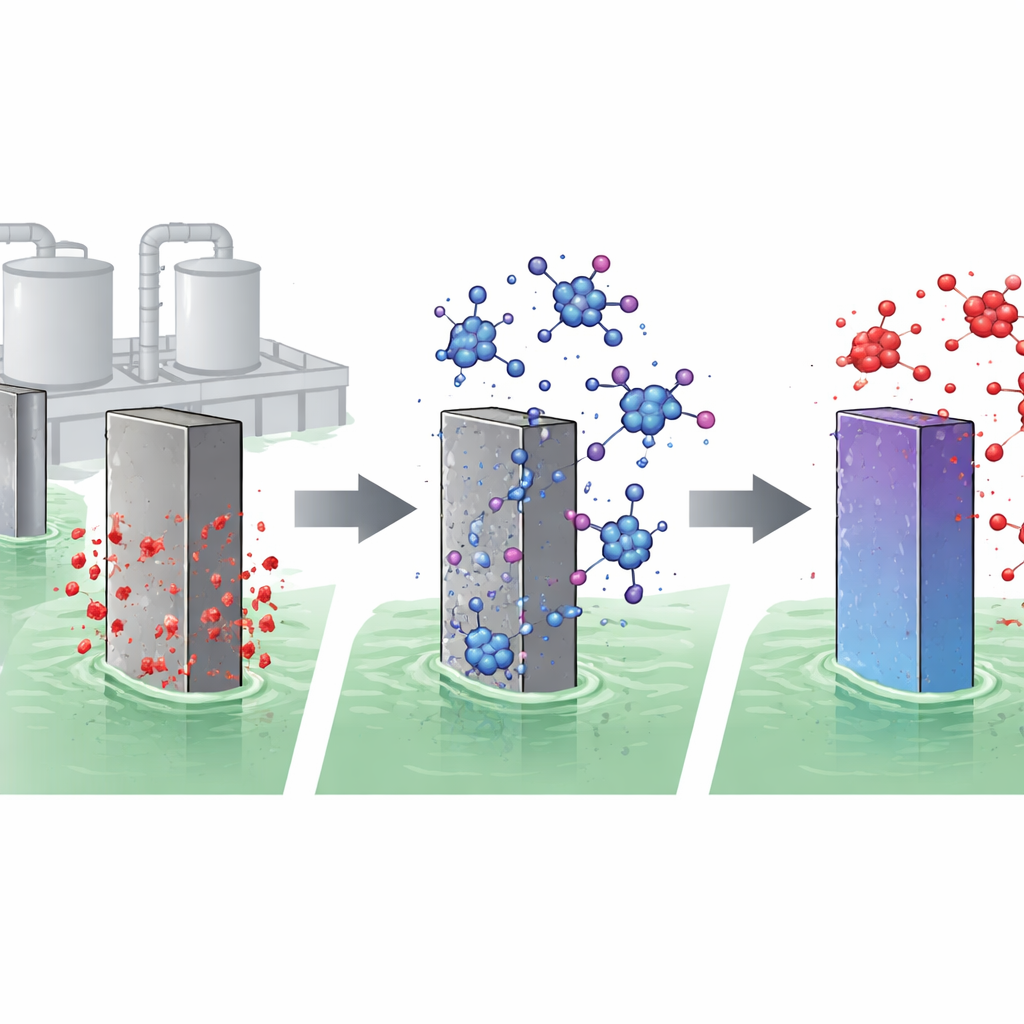
Giving an Expired Drug a Second Life
The researchers focused on L‑ornithine L‑aspartate, a compound made from two amino acids and usually prescribed as a liver support supplement. Even six months after its expiry date, careful testing showed that almost all of the drug molecules were still chemically intact. The scientists dissolved this expired material in a strong hydrochloric acid solution and immersed samples of a common construction steel, AISI 1018, to see whether the drug could slow corrosion. They then used several complementary methods—weight‑loss measurements, electrical tests, surface imaging, and computer simulations—to build a full picture of how well the drug works and why.
How the Invisible Shield Forms on Steel
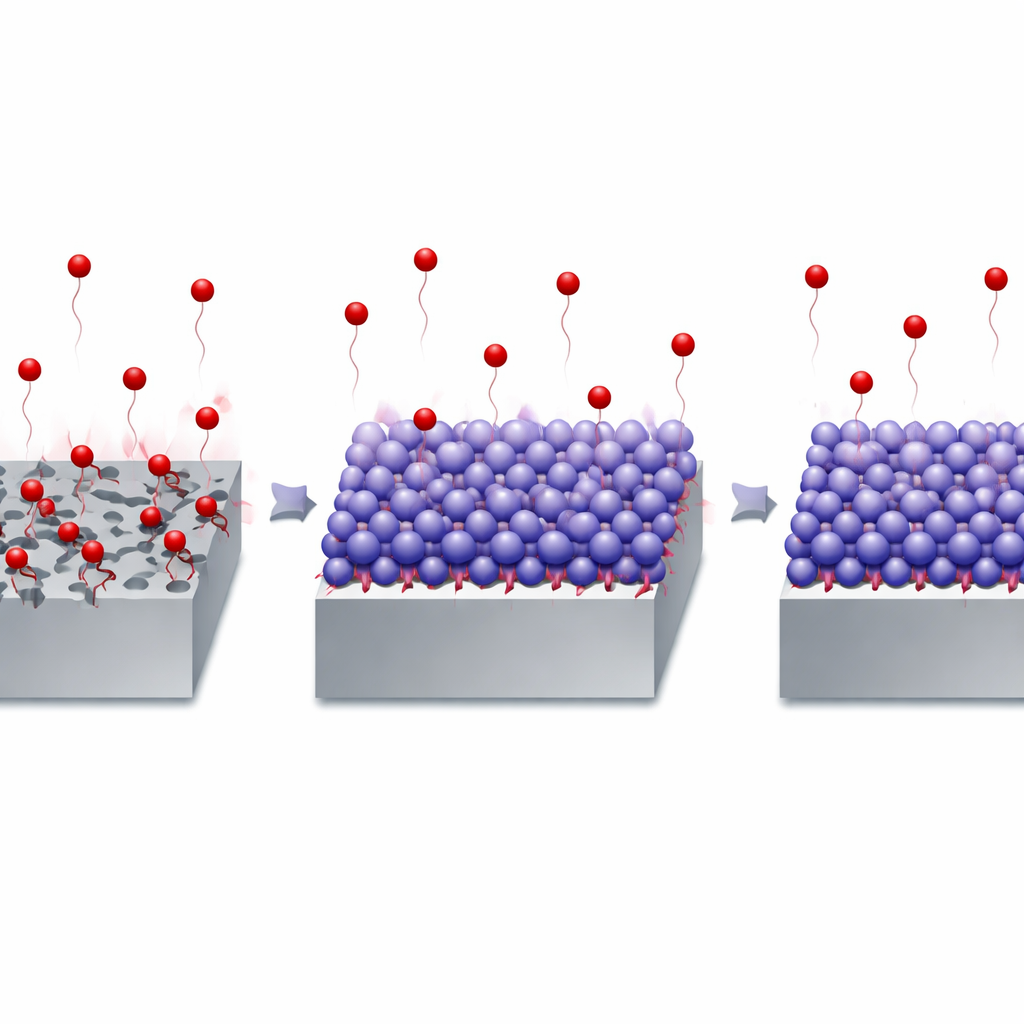
When the expired drug is added to the acid, its molecules are attracted to the steel surface and stick to it, forming a thin organic film. Electrochemical measurements show that as the drug concentration increases, the flow of corrosive current at the steel surface drops sharply. At the highest tested level, the corrosion rate falls by more than 90 percent, and the steel loses almost no mass over hours in hot acid. Microscopic images back this up: steel exposed to acid alone looks rough and deeply pitted, while steel protected by the drug is smooth and largely free of damage. Computer models of how single molecules land on an iron surface reveal that the drug clings strongly and spreads out, creating a tight barrier that blocks aggressive chloride ions and hydrogen from reaching the metal.
Stable Protection Under Real‑World Conditions
The team also tested how the protection changes with temperature, since many industrial processes run hot. As the acid is warmed from room temperature to near the boiling point of water, corrosion speeds up, but the expired drug still keeps more than 90 percent of the damage at bay. Calculations show that the drug raises the energy barrier that corrosion reactions must overcome, meaning the metal surface becomes harder to attack. Studies of how the molecules arrange themselves suggest they first occupy the most reactive spots on the steel and then spread over less active areas, forming a somewhat uneven but highly effective multilayer coating. Interestingly, fresh and expired versions of the drug perform almost identically, confirming that the “waste” material still has all the right features to protect metal.
What This Means for Industry and the Environment
In simple terms, this work shows that a safe, inexpensive liver supplement—long past its medical shelf life—can act as a highly efficient rust‑stopper for steel in extremely harsh acid. By turning an environmental burden (expired pharmaceuticals) into a protective coating for industrial metals, the approach links cost savings with cleaner chemistry. If scaled up, similar strategies could help factories reduce both corrosion‑related failures and drug disposal problems, moving one step closer to a circular economy where materials are reused rather than thrown away.
Citation: Alshammari, O.A.O., Abdelwahab, A., Jeilani, Y.A. et al. Electrochemical and quantum chemical exploration on L-ornithine L-aspartate as a sustainable corrosion inhibitor for AISI 1018 steel in acidic environment. Sci Rep 16, 13029 (2026). https://doi.org/10.1038/s41598-026-49295-4
Keywords: corrosion inhibition, expired pharmaceuticals, steel protection, green chemistry, acidic environments