Clear Sky Science · en
Eco friendly obtained zirconium oxide crystals for efficient separation of rare earth elements from acidic media
Turning Waste Peels into Valuable Metals
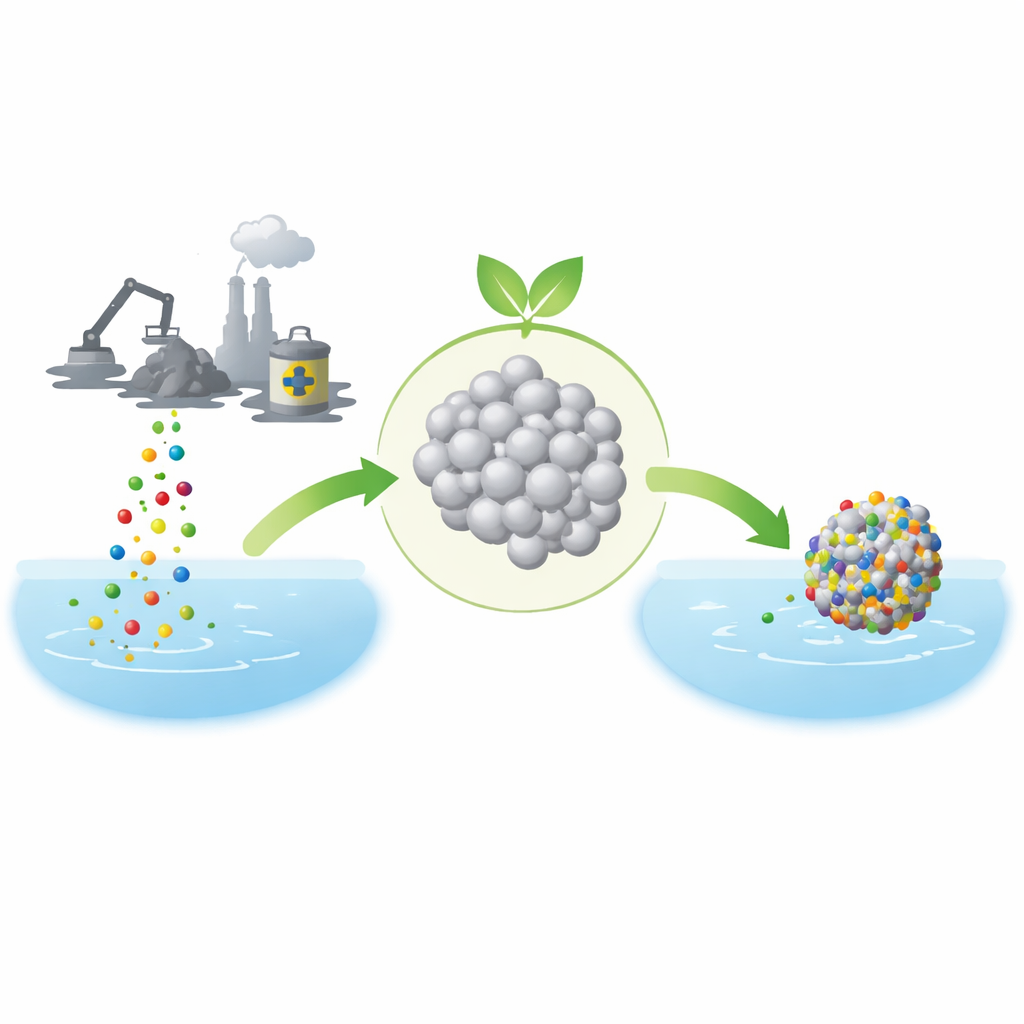
Modern technologies, from smartphones to wind turbines, rely on rare earth elements—metallic ingredients that are hard to separate and even harder to recycle cleanly. This study shows how an everyday kitchen scrap, pomegranate peel, can help create a simple material that pulls valuable rare earth metals out of acidic waste streams. The work points toward cleaner ways to recover these strategic resources while reducing both industrial pollution and radioactive waste.
Why Rare Metals Matter
Rare earth elements such as lanthanum, europium, and samarium are essential for bright screens, powerful magnets, advanced ceramics, and nuclear technology. Although they are not truly rare in Earth’s crust, they are scattered at low concentrations, making mining and separation complex, costly, and polluting. Large volumes of liquid waste from mining, metal processing, and nuclear fuel handling carry these elements away, both squandering their value and posing environmental and health risks similar to heavy metals. Finding low-cost methods to concentrate and separate these metals from harsh, acidic solutions is therefore both an economic and environmental priority.
A Green Powder from Fruit Peels
The researchers set out to make a metal-oxide powder that could latch onto rare earth ions while being cheap and environmentally friendly to produce. They chose zirconium oxide, a robust ceramic already used in dentistry and sensors, and prepared it using an approach known as green synthesis. Instead of relying on toxic chemicals, they boiled discarded pomegranate peels to extract natural plant compounds, then mixed this extract with a zirconium salt solution. By gently adjusting the mixture’s alkalinity and heating it, they obtained tiny zirconium oxide crystals. A suite of analytical tools confirmed the product’s structure, stability, and nanoscale grain size, and showed that its surface was rich in sites where metal ions could attach.
How the Powder Grabs Metal Ions
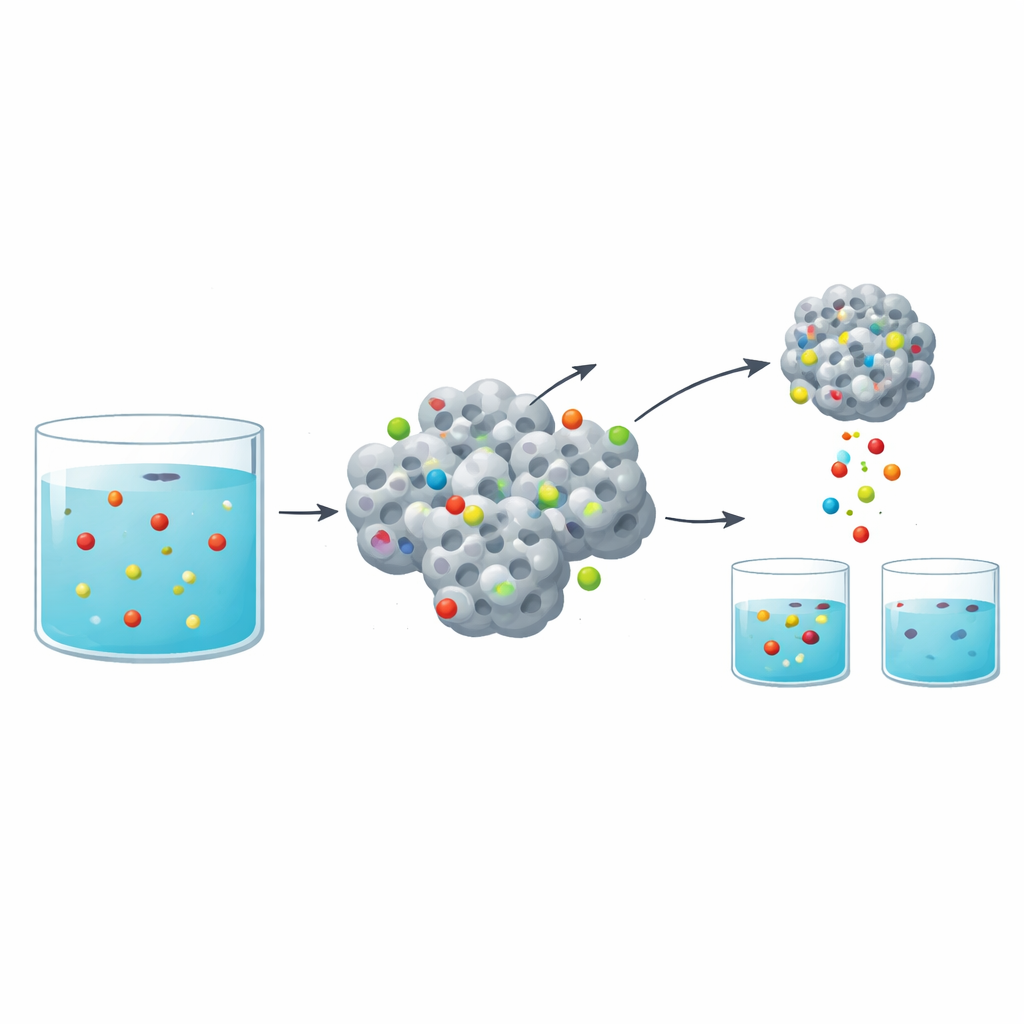
To test performance, the team stirred the zirconium oxide powder into acidic water containing known amounts of lanthanum, europium, and samarium ions. They varied key conditions—acidity, temperature, contact time, initial metal concentration, and the amount of powder—to see how these factors affected removal. At a mildly acidic pH of about 3.5, the material was especially effective, stripping more than 90 percent of each metal from solutions with moderate concentrations. The data showed that uptake was fast during the first hour as many open sites on the powder’s surface were available, then slowed as those sites filled and the system approached equilibrium. Mathematical models of the time-dependent behavior indicated that the metals bonded mainly through a chemisorption process, meaning they formed stronger, more specific interactions than simple physical sticking.
What the Models Reveal About the Surface
By running experiments across a wide span of metal concentrations, the authors could map how much each gram of powder could hold and how tightly it bound the ions. Classic adsorption models suggested that the zirconium oxide behaved partly like a uniform surface of identical sites and partly like a more varied landscape of sites with different strengths. Further analysis of the energy involved in binding supported a mixed mechanism: strong, chemical-like attachment combined with weaker, physical interactions. Additional tests showed that competing ions typical of industrial waste, such as cesium, strontium, and cobalt, did not greatly reduce the capture of the target rare earths, hinting at a useful degree of selectivity.
Using and Reusing the Material
For any real-world cleanup or recycling process, the sorbent material must not be single-use. The researchers therefore tested how easily the captured rare earth ions could be stripped off and the zirconium oxide reused. By washing the loaded powder with a dilute nitric acid solution, they recovered more than 90 percent of the metals and restored most of the powder’s capacity. After five adsorption–desorption cycles, performance had only slightly declined, indicating that the material remained structurally sound and functional under repeated use and acid exposure.
A Simple Route to Cleaner Recovery
In plain terms, this work shows that a stable white powder made with help from fruit waste can efficiently sponge valuable rare earth metals out of harsh, acidic liquids and be reused multiple times. The process runs better at warmer temperatures and moderate acidity, and the underlying physics suggests that the metals are held strongly enough to be captured yet can still be released on demand. While other advanced materials may hold even more metal per gram, this green-synthesized zirconium oxide balances reasonable capacity with simplicity, low cost, and environmental friendliness. It offers a promising path toward cleaner recovery of critical elements from industrial and nuclear waste streams, turning a disposal challenge into a resource.
Citation: El-Tantawy, A., Ali, I.M. Eco friendly obtained zirconium oxide crystals for efficient separation of rare earth elements from acidic media. Sci Rep 16, 14693 (2026). https://doi.org/10.1038/s41598-026-48985-3
Keywords: rare earth recycling, green nanomaterials, water purification, zirconium oxide adsorbent, nuclear waste treatment