Clear Sky Science · en
Cytotoxic and apoptotic effects of kaempferol 3-O-rhamnoside from Schima wallichii in HepG2 cells
How a forest tree may help fight liver cancer
Many modern medicines began as molecules found in wild plants. This study explores a compound from the Asian tree Schima wallichii that can push liver cancer cells to self-destruct, offering clues for how traditional remedies might inspire future cancer treatments.
A healing tree from Asian forests
Schima wallichii is an evergreen tree that grows across the Himalayas, China, Southeast Asia, and Vietnam. Local communities have long used its leaves and bark to treat fever, infections, and skin problems. Scientists already knew the plant contained antioxidant and antimicrobial substances, but its potential against cancer, especially liver cancer, had not been fully explored.
From leaves to a single active ingredient
Researchers collected Schima wallichii leaves in central Vietnam and prepared different extracts using common solvents. These extracts were tested on three human cancer cell types: lung, breast, and liver. A moderately polar fraction from the leaves was the most effective, especially against liver cancer cells known as HepG2. From this fraction, the team purified one major flavonoid, kaempferol-3-O-rhamnoside, a plant-based molecule related to pigments found in many fruits and vegetables. This compound showed much stronger growth-blocking effects on liver and breast cancer cells than the crude extracts, hinting that it is a key actor behind the plant’s anticancer activity.
Making cancer cells stop dividing and die
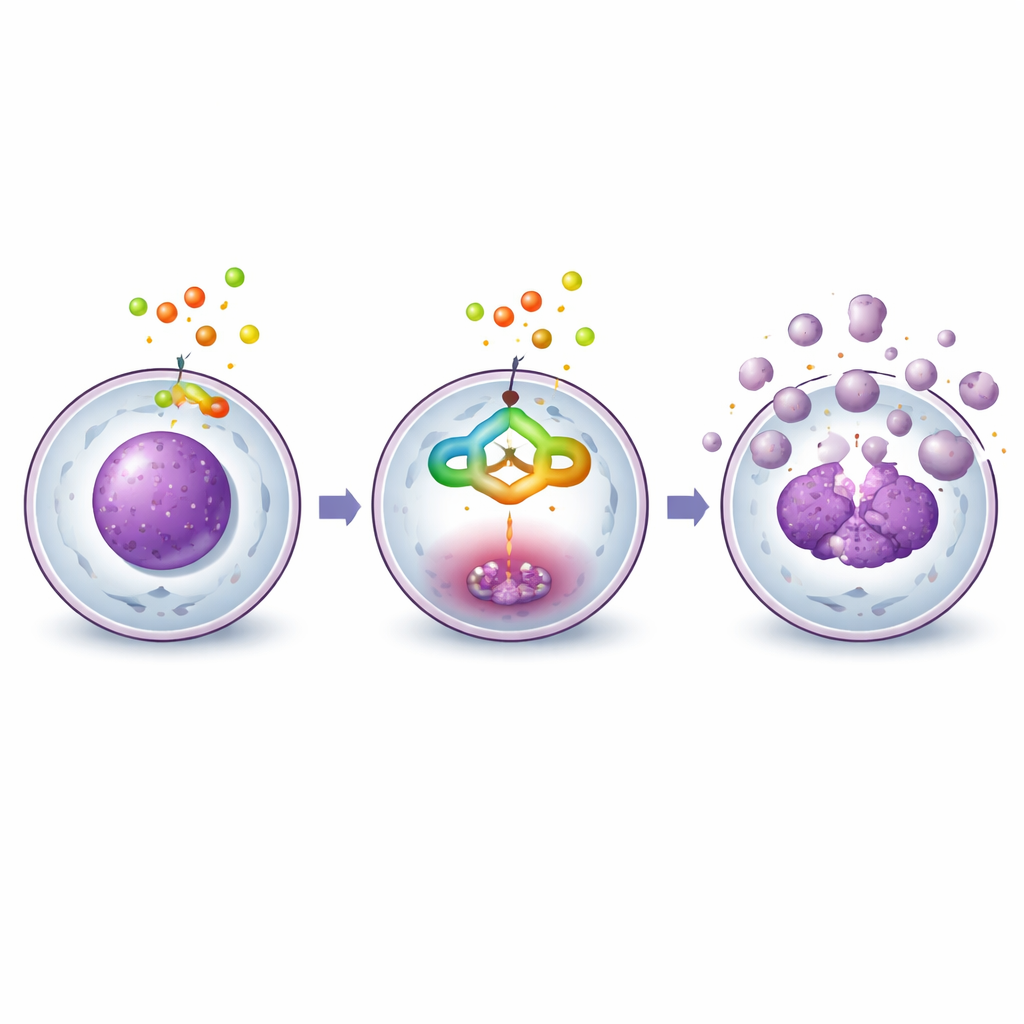
When liver cancer cells were exposed to the purified compound, their behavior changed in a clear, dose-dependent way. Under the microscope, untreated cells formed dense, healthy-looking layers. With increasing amounts of the compound, cells became round, shrank, detached from the surface, and finally broke into small pieces, all classic signs of programmed cell death, or apoptosis. Flow cytometry, a technique that measures DNA content in thousands of cells, showed that treated cells accumulated at the G2/M checkpoint, a control point just before cell division. This buildup means the compound blocks the normal cell cycle, preventing cancer cells from multiplying while nudging them toward self-destruction.
Switching on the cell’s internal self-destruct program
The team then examined key proteins that control apoptosis. They focused on caspase-3, an enzyme often called an executioner because it cuts many vital cell components during cell death. Biochemical tests showed that caspase-3 activity rose several-fold after treatment, and Western blots revealed that its inactive form was steadily converted into the active, cleaved form. Other related proteins, including upstream caspases and a DNA repair factor called PARP-1, also showed changes consistent with an internal death pathway being activated. Together, these results indicate that the compound does not simply poison the cells; instead, it engages the cells’ own built-in cleanup machinery.
Computer models support the lab findings
To better understand how the molecule might fit into these proteins, the researchers used computer docking and molecular dynamics simulations. These virtual experiments suggested that the compound can nestle snugly into the active pocket of caspase-3 and other apoptosis-related proteins, forming stable networks of hydrogen bonds and stacking interactions with specific amino acids. Over a simulated 100-nanosecond period in water, the complex between the compound and caspase-3 remained stable, with little structural wobble. Calculations of its electronic structure showed a balance between stability and reactivity, consistent with its ability to form multiple noncovalent contacts rather than reacting in a destructive way.
What this means for future therapies
In simple terms, the study shows that a natural molecule from Schima wallichii leaves can slow the growth of human liver cancer cells and push them to die by turning on their internal self-destruct system, rather than by blunt toxicity. While these results come from cell cultures and computer models, and not yet from animal or human studies, they provide a detailed picture of how this plant compound might work at the molecular level. This adds scientific support to traditional uses of the tree and points to kaempferol-3-O-rhamnoside as a promising starting point for designing safer, more targeted treatments for liver cancer in the future.
Citation: Lam, T.M.P., Tran, M.D., Nguyen, T.K. et al. Cytotoxic and apoptotic effects of kaempferol 3-O-rhamnoside from Schima wallichii in HepG2 cells. Sci Rep 16, 14656 (2026). https://doi.org/10.1038/s41598-026-48333-5
Keywords: Schima wallichii, liver cancer, apoptosis, natural compounds, caspase-3