Clear Sky Science · en
Sustainable heteroatom doped biochar for methylene blue adsorption with structure function insights
Turning Farm Waste into Clean Water Tools
Colorful dyes that give our clothes and products their bright shades can become a serious problem once they wash down the drain. Many resist natural breakdown and can harm aquatic life and human health. This study explores a simple but powerful idea: using charred agricultural waste, tuned at the microscopic level, as a sponge to pull a common blue dye out of water. By transforming crop residues into smart, low‑cost filters, the work points toward more sustainable ways to clean polluted water while putting farm leftovers to good use. 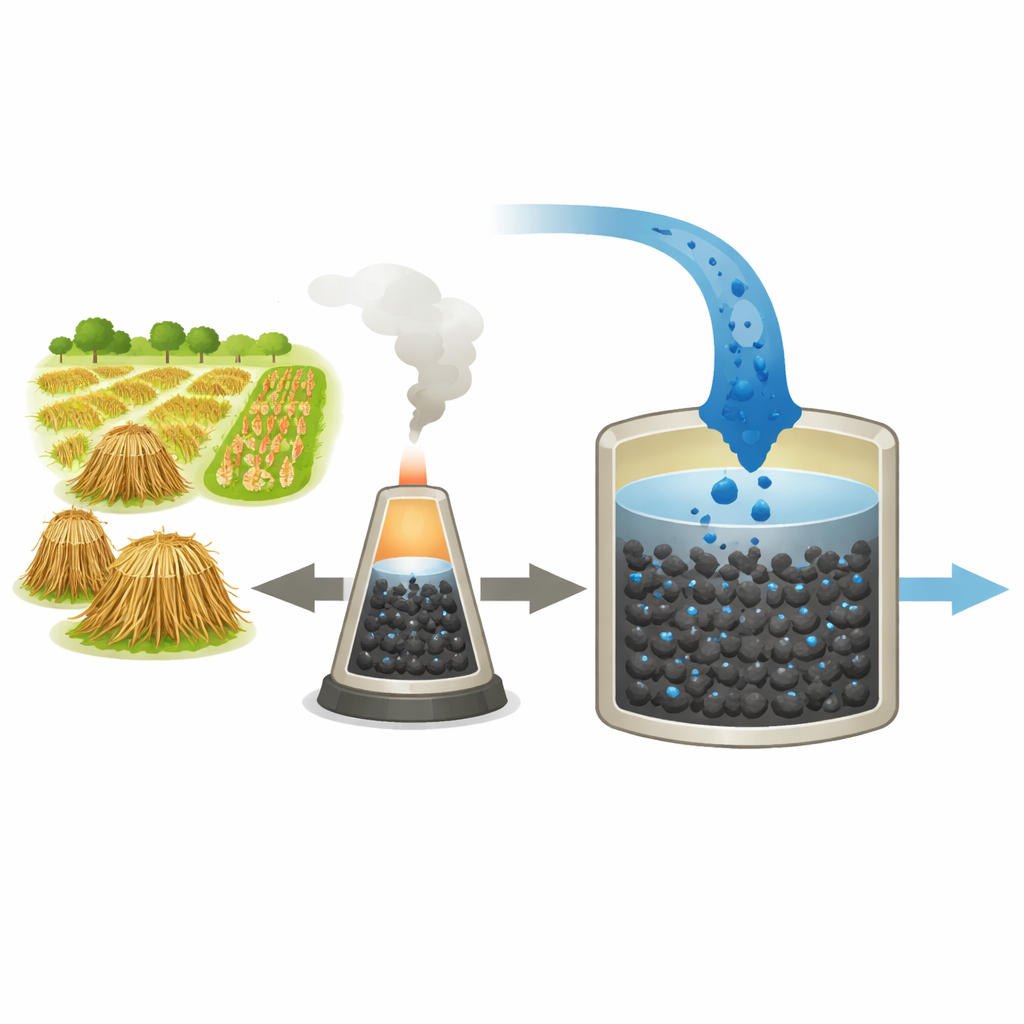
From Crop Stalks to Porous Char
The researchers started with four abundant farm by‑products from Egypt’s Nile Delta: rice straw, date palm fiber, sugarcane bagasse, and giant reed. When these materials are heated in low oxygen, they turn into biochar—a carbon‑rich, porous solid. Not all crop residues perform equally, though. Rice straw biochar stood out, offering the highest capacity to hold onto methylene blue, a bright cationic dye often used as a stand‑in for real textile pollutants. Its success stems from its chemistry: rich in lignin and mineral ash, rice straw forms a more stable, carbon‑dense char with a naturally more porous structure than the other residues. This made rice straw the prime candidate for further upgrading.
Upgrading the Sponge: Adding Helpful Atoms
To see how far they could push performance, the team modified rice straw biochar using two different approaches. One route used a nitrogen‑containing compound and heat in a sealed, water‑filled vessel (hydrothermal treatment) or in a household microwave to introduce nitrogen atoms into the carbon network. These nitrogen species create extra basic spots on the surface that can attract positively charged dye molecules and strengthen subtle stacking interactions between aromatic rings in the dye and the carbon surface. A second route soaked the biochar in phosphoric acid before heating, carving out a sponge‑like network of pores and decorating the surface with acidic oxygen‑ and phosphorus‑based groups. Across detailed tests, hydrothermal nitrogen doping and phosphoric‑acid activation emerged as the clear winners, far outperforming both unmodified biochar and the simpler microwave treatment. 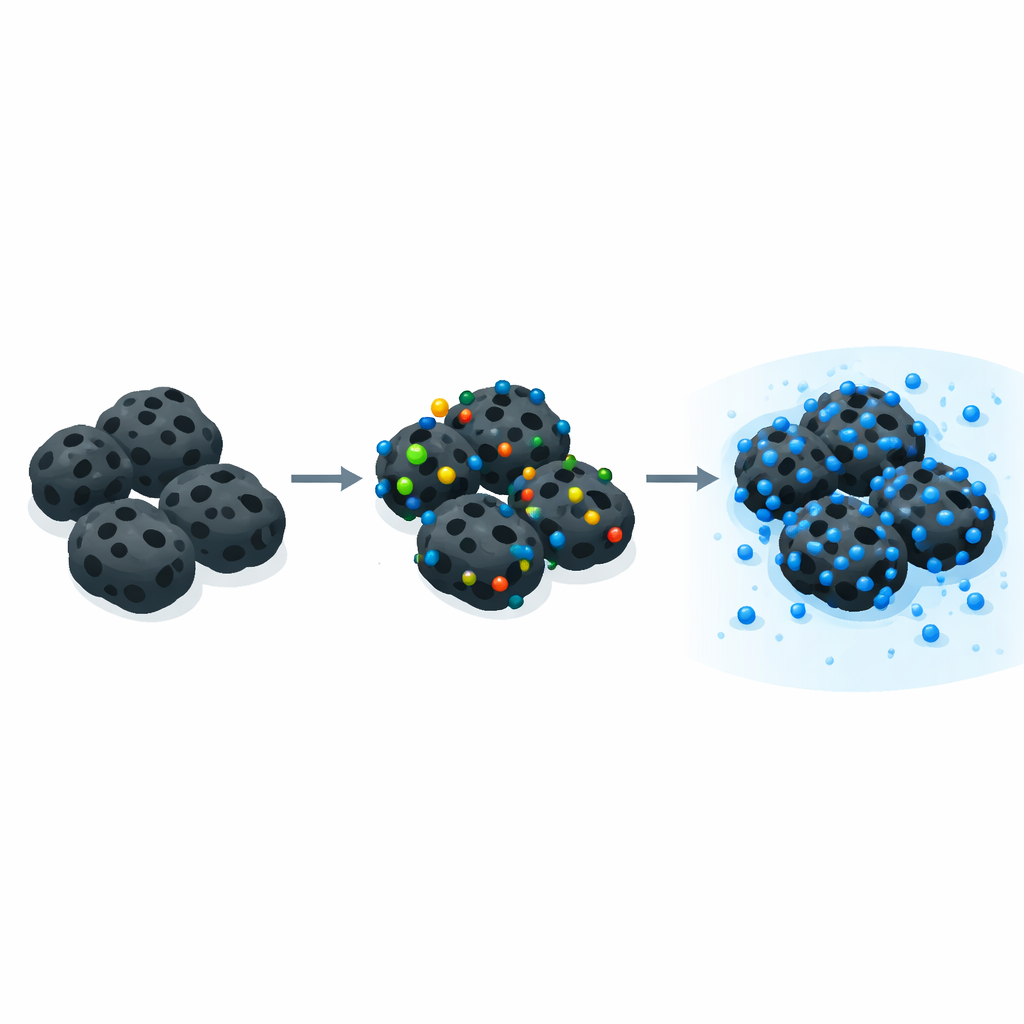
How the Modified Biochar Grabs Dye
Laboratory experiments traced how methylene blue moves from water onto these engineered surfaces. Measurements showed that dye molecules pack into a single, organized layer on the biochar, and that the uptake over time follows a pattern consistent with strong, specific interactions rather than mere weak sticking. Several forces work together: electrostatic attraction between charged dye molecules and oppositely charged sites on the surface; stacking of the dye’s flat aromatic rings against the carbon sheets; hydrogen bonds; and complexation with certain oxygen‑ and nitrogen‑bearing groups. The best nitrogen‑doped rice straw reached an adsorption capacity of about 137 milligrams of dye per gram of biochar, while the phosphoric‑acid‑activated version reached about 146 milligrams per gram—values that match or exceed many other plant‑based adsorbents reported in the literature.
Fine‑Tuning Conditions for Real‑World Use
The team also explored how water conditions shape performance, since real waste streams vary in pH, pollutant load, and temperature. Both top‑performing biochars worked better in more alkaline solutions, where their surfaces carry more negative charge and more strongly attract the positively charged dye. Raising the dye concentration increased how much each gram of biochar could hold until the surface approached saturation. Higher temperatures favored uptake as well, indicating an endothermic, spontaneous process that becomes more efficient with warmth. Importantly, the two leading materials responded somewhat differently: phosphoric‑acid‑activated biochar excelled at quickly stripping dye at lower doses and milder conditions, while nitrogen‑doped biochar matched its performance when pH and loading were optimized, benefiting from deeper micropores and a richer mix of surface sites.
What This Means for Cleaner Water
In plain terms, the study shows that farm waste—especially rice straw—can be turned into highly effective, tunable filters for dye‑contaminated water. By carefully adjusting how the char is made and which extra atoms are added, scientists can design surfaces that grab and hold large amounts of dye using a mix of charge attraction and molecular “Velcro‑like” sticking. The two leading strategies, nitrogen doping and phosphoric‑acid activation, reach similar top‑tier performance but differ in how they build porosity and surface chemistry. Together they offer a toolbox for creating low‑cost, robust biochar adsorbents that can help clean industrial wastewater while reducing open‑field burning and other disposal problems for agricultural residues.
Citation: Lotfy, V.F., Basta, A.H. Sustainable heteroatom doped biochar for methylene blue adsorption with structure function insights. Sci Rep 16, 13153 (2026). https://doi.org/10.1038/s41598-026-48042-z
Keywords: biochar, wastewater treatment, methylene blue, agricultural residues, adsorption