Clear Sky Science · en
Hydrometallurgical recovery of high-purity molybdenum from spent iron-molybdate catalysts via ammoniacal leaching and anti-solvent crystallization
Why old catalysts matter for new technology
Molybdenum is a metal you may never have heard of, yet it quietly underpins everyday technologies—from sturdy steels in cars and buildings to chemicals used in plastics and fuels. Much of the world depends on imports of this metal, leaving supply vulnerable to political and economic shocks. At the same time, factories discard large amounts of molybdenum locked inside used-up catalysts. This study shows how those spent materials can be turned from hazardous waste into a clean, high-value resource using a low‑energy, room‑temperature process.
Turning waste pellets into a metal source
Industrial plants that make formaldehyde, an important chemical building block, rely on iron–molybdate catalysts: hard, gray pellets containing more than 50 percent molybdenum by weight. Once these catalysts lose their activity, they are usually treated as troublesome waste despite their rich metal content. The researchers started by grinding these spent pellets into a fine powder and carefully analyzing their composition. They confirmed that molybdenum and iron are the main ingredients, arranged in crystalline compounds that are stable and not easy to dissolve in water. This set the stage for designing a targeted chemical route that would pull molybdenum into solution while leaving most of the iron behind.
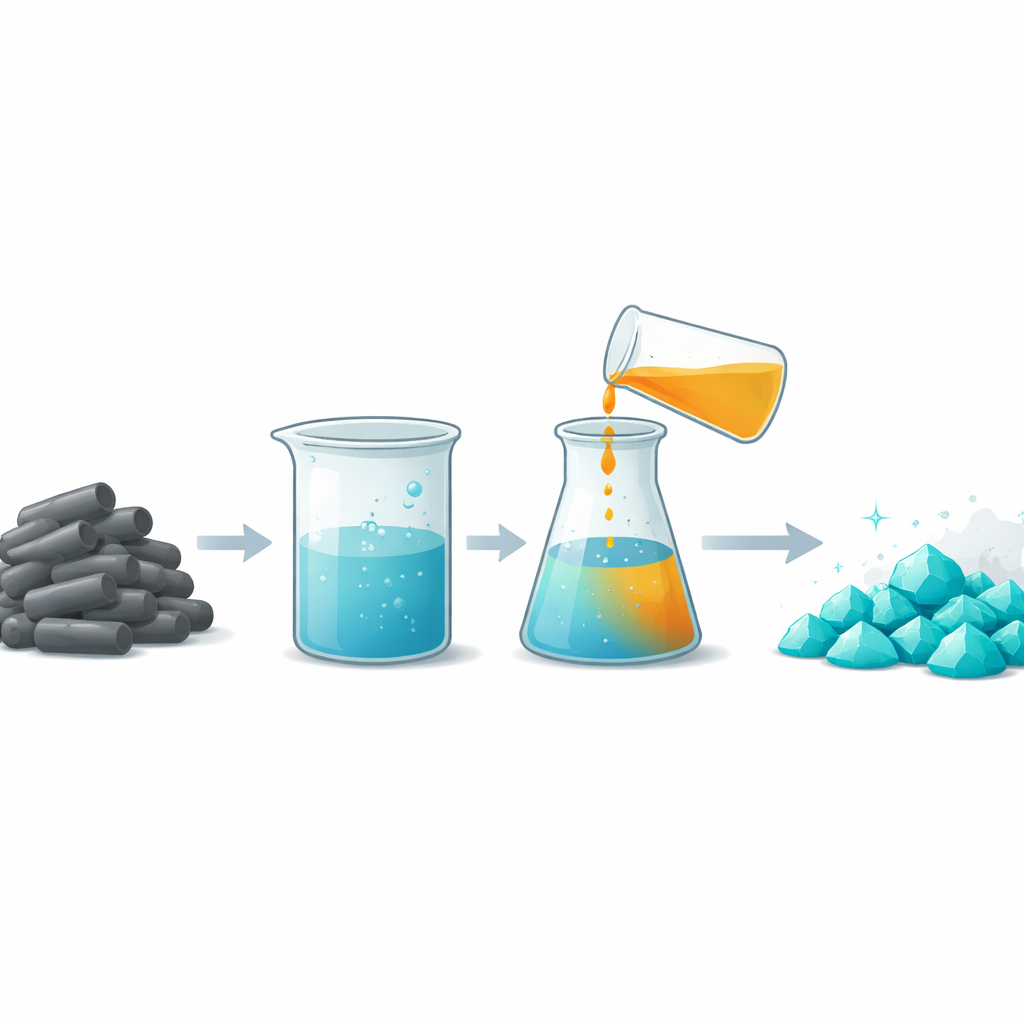
Gentle chemistry that saves reagent and energy
The first step in the new process is called leaching, in which the powdered catalyst is mixed with an alkaline solution so that molybdenum can move into the liquid phase. Using ammonia alone, earlier methods needed fairly strong solutions and still could not dissolve all of the molybdenum efficiently. In contrast, this study uses a smart combination of ammonia and an ammonium salt that together act as a buffer, keeping the solution in a narrow, favorable range of acidity and electrical potential. Under these mild, room‑temperature conditions, about 92 percent of the molybdenum dissolves even though the total amount of ammonia is less than half of what traditional recipes require. Iron behaves differently: it tends to form solid hydroxides and stay in the residue, so less than 5 percent follows molybdenum into the liquid.
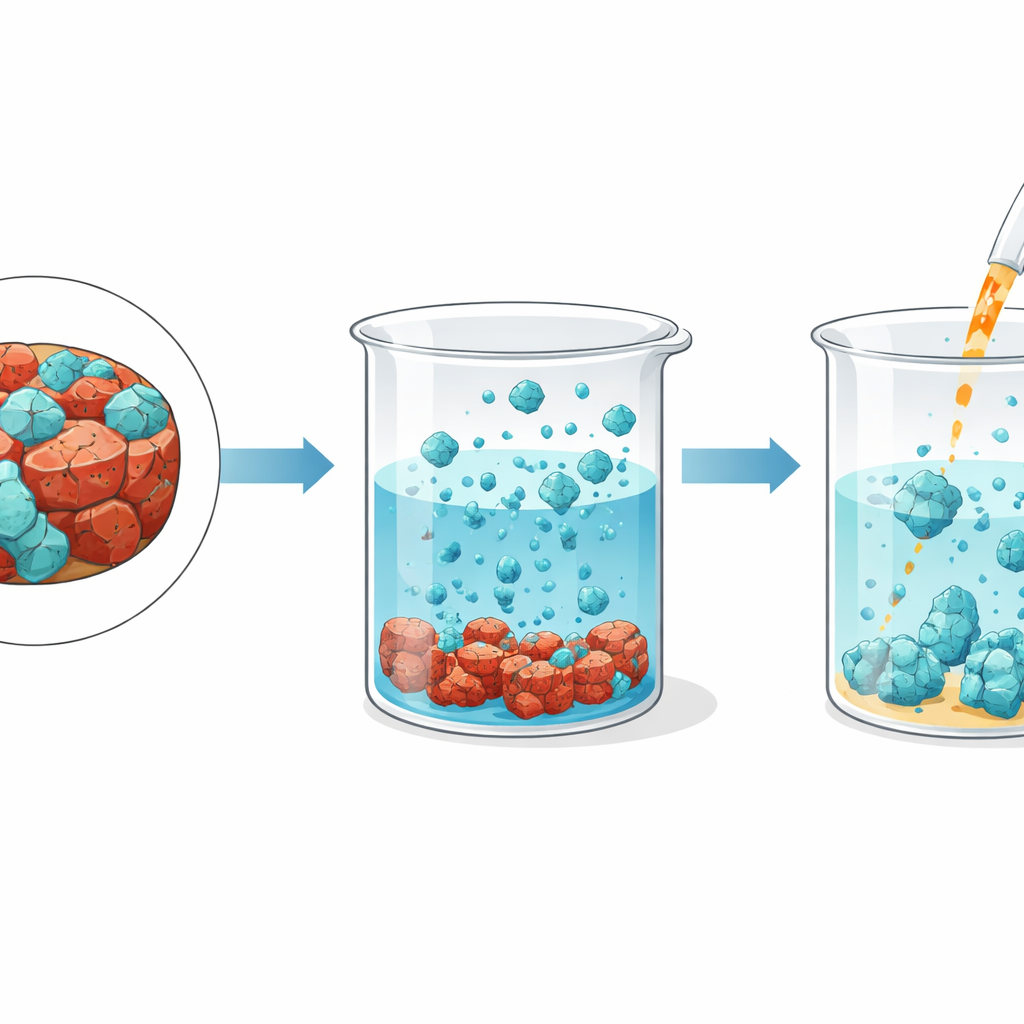
Letting iron quietly fall out
An interesting twist comes from how traces of iron remain temporarily dissolved. Some iron atoms form weak complexes with ammonia molecules, lingering in the solution in a kind of unstable balance. The team found that simply letting the leach solution sit undisturbed for several hours allows this balance to shift. The iron slowly converts into solid particles that settle to the bottom and can be removed by filtration, leaving a clear liquid rich in molybdenum and almost free of iron. This slow “self‑cleaning” step avoids the need for extra chemicals or heating, further simplifying the process and keeping energy use low.
Crystallizing pure product with a splash of alcohol
The next challenge is to pull molybdenum back out of the liquid as a solid product that can be reused. Rather than evaporating large amounts of water with heat, the researchers use a technique called anti‑solvent crystallization. They add ethanol, a common organic solvent, to the ammonium–molybdate solution. Because ethanol has a lower ability to stabilize charged particles than water, its presence reduces how well molybdenum species stay dissolved. As a result, they come together and crystallize as small, well‑formed particles of ammonium heptamolybdate, a standard commercial molybdenum compound. By tuning how much ethanol is added, how fast the mixture is stirred, and how long it is mixed, the team achieves about 95 percent recovery of molybdenum in crystals with extremely high purity.
From lab recipe to resource security
In everyday terms, this work shows how careful control of simple ingredients—ammonia, an ammonium salt, and ethanol—can transform spent catalyst pellets into a near‑perfect molybdenum product, all at room temperature and with modest chemical use. The approach largely avoids energy‑hungry heating steps and minimizes unwanted by‑products, making it both economically and environmentally attractive. If scaled up, such recycling schemes could turn existing waste streams into a strategic secondary source of molybdenum, easing dependence on a few mining regions and helping to secure supplies for the technologies that modern society relies on.
Citation: Farhan, M., Srivastava, R.R. & Ilyas, S. Hydrometallurgical recovery of high-purity molybdenum from spent iron-molybdate catalysts via ammoniacal leaching and anti-solvent crystallization. Sci Rep 16, 12039 (2026). https://doi.org/10.1038/s41598-026-47825-8
Keywords: molybdenum recycling, spent catalysts, hydrometallurgy, metal recovery, anti-solvent crystallization