Clear Sky Science · en
Bio-functionalities of nitrogen based carbon dots from chitosan via in-situ incorporation with nano-copper
Turning Shellfish Waste into Tiny Disease Fighters
Imagine if leftovers from shrimp shells could be turned into tiny particles that fight germs, soothe inflammation, and mop up harmful molecules in the body. This study shows how scientists did something very close to that: they transformed chitosan, a natural material from shellfish, into glowing nano-sized “dots” and carefully added copper to them. The result is a simple, low-toxicity material that kills bacteria and fungi, acts as an antioxidant, calms inflammation, and may one day help treat infections and chronic diseases.
From Natural Polymer to Tiny Light-Emitting Dots
Chitosan is a biodegradable substance already used in wound dressings and other medical products. The researchers first tweaked chitosan chemically to make it easier to break down and rebuild. Then they heated it in water under pressure to form nitrogen-containing carbon dots—nanoparticles only a few billionths of a meter across that naturally glow under ultraviolet light. These dots are easy to make, resist light damage, and can be linked to biological molecules, which makes them attractive for medical uses ranging from imaging to drug delivery.
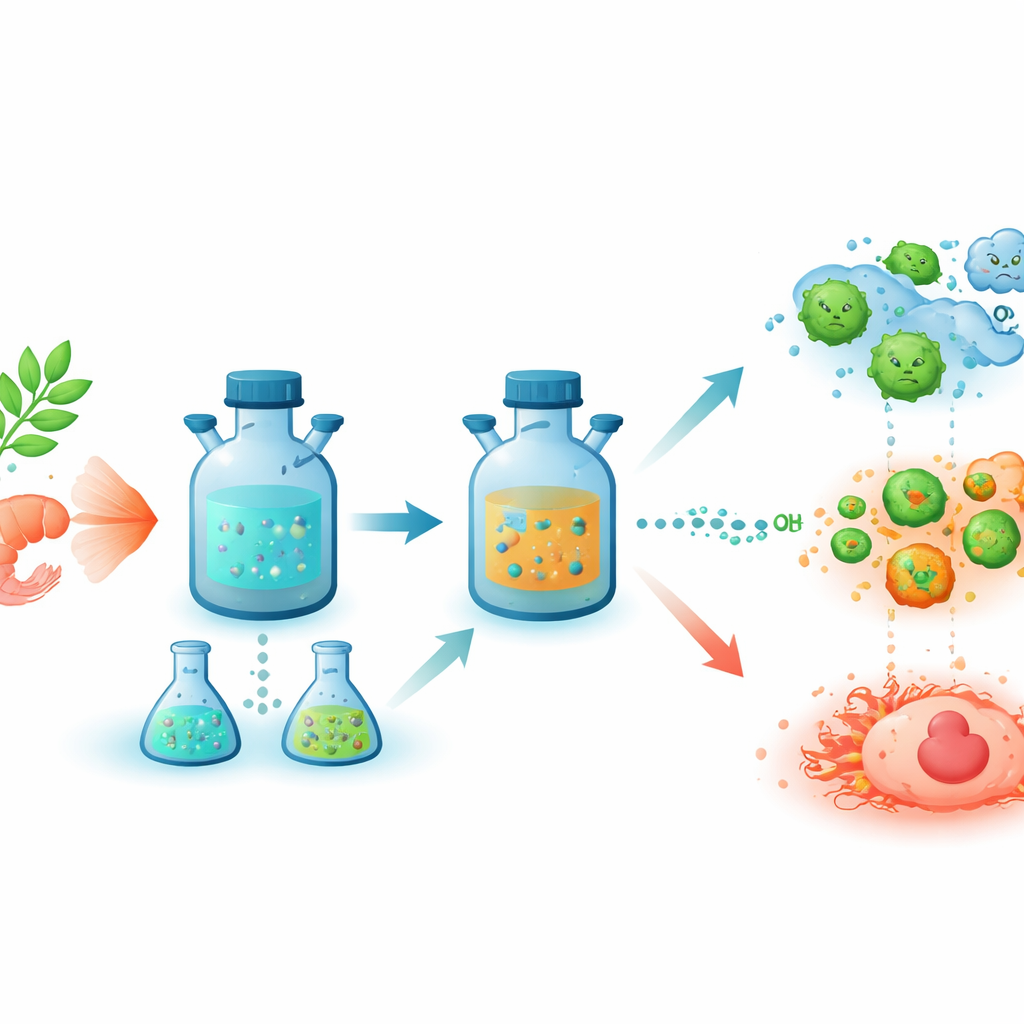
Adding Copper Without Harsh Chemicals
The team’s main twist was to build copper directly into these carbon dots without using any extra chemical reducing agents, an important step toward greener manufacturing. They mixed the freshly made carbon dots with one of three common copper salts—copper sulfate, copper nitrate, or copper acetate—and heated them again in water. Under these conditions, copper nanoparticles formed and became anchored onto the surface of the dots, creating three versions of copper-loaded particles. Although all three ended up with similar amounts of copper overall, their sizes and internal structures differed depending on which copper salt was used, showing that the exact starting ingredient subtly steers how the final nanomaterial grows.
How Copper Changes Size, Light, and Stability
Careful imaging revealed that the original nitrogen-doped carbon dots averaged about 10 nanometers in size, while the copper-decorated versions swelled to roughly three to five times that diameter. The different copper salts produced particles with slightly different shapes and degrees of uniformity, and also changed how the dots handled electric charge in water—properties that affect how they move and cluster in liquids and in the body. Interestingly, adding copper dimmed the natural glow of the dots. This happens because copper atoms create extra pathways for excited electrons to lose energy without emitting light. At the same time, copper improved the dots’ resistance to damage under prolonged ultraviolet exposure, suggesting that the modified particles could remain functional longer in real-world uses.
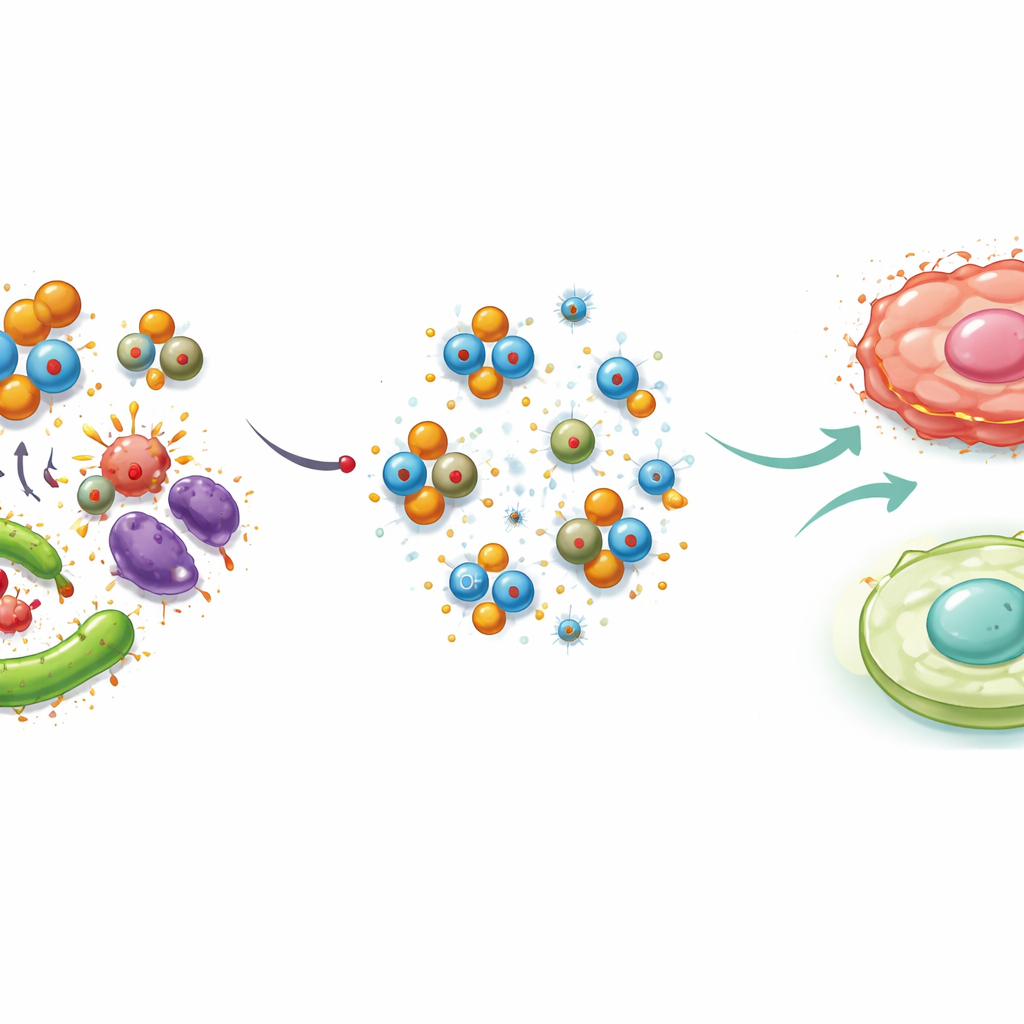
Fighting Germs, Oxidative Stress, and Inflammation
The most striking changes showed up when the particles were tested on living systems. Against two common bacteria—Staphylococcus aureus and Escherichia coli—and a fungal species, Candida albicans, the copper-doped dots killed far more microbes than undoped dots and in some cases approached the performance of standard drugs. They also required much lower doses to stop microbial growth. Similarly, in a standard test of antioxidant power, the copper-loaded dots neutralized nearly three times as many damaging free radicals as the original dots, with the nitrate-based version performing best. In inflammation tests using immune cells grown in dishes, copper-doped dots significantly improved cell survival compared with undoped dots, again with the nitrate-based particles giving the strongest protective effect.
Safety Signals and Future Promise
To check whether this added power came with extra danger, the researchers tested the particles on tiny aquatic organisms called brine shrimp. Even at concentrations higher than those needed to kill microbes, none of the copper-doped dots caused observable harm or death in the shrimp, suggesting low short-term toxicity under the tested conditions. Taken together, the results indicate that carefully incorporating copper into carbon dots made from a natural biopolymer can create a single, water-based nanomaterial that disinfects, reduces oxidative stress, and calms inflammation, all while appearing safe in early tests. Although more studies are needed, including tests against cancer cells and more detailed safety work, these copper–carbon dots hint at a future where waste-derived nanomaterials help protect our bodies from infection and chronic damage.
Citation: Emam, H.E., Rimdusit, S. & Ahmed, H.B. Bio-functionalities of nitrogen based carbon dots from chitosan via in-situ incorporation with nano-copper. Sci Rep 16, 13275 (2026). https://doi.org/10.1038/s41598-026-47664-7
Keywords: carbon quantum dots, chitosan, copper nanoparticles, antimicrobial nanomaterials, antioxidant and anti-inflammatory therapy