Clear Sky Science · en
Evolution of polypyrrole electrode during electropolymerization and its effect on energy storage performance
Storing More Energy in a Smaller Package
From electric cars to backup power for homes, we increasingly rely on devices that can deliver bursts of energy quickly without wearing out. Supercapacitors are promising candidates, but their performance depends heavily on the tiny structures inside their electrodes. This article explores how carefully controlling the growth time of a common conducting plastic, polypyrrole, can reshape those structures and significantly boost how much energy a supercapacitor can store and deliver.
Why This Plastic Matters for Future Power
Supercapacitors sit between batteries and regular capacitors: they charge and discharge much faster than batteries but usually store less energy. A key way to improve them is to engineer electrodes with a large surface area and easy pathways for ions from the electrolyte to move in and out. Polypyrrole, a conducting polymer made from the small molecule pyrrole, is attractive because it conducts electricity well, is stable in water-based environments, and can be formed directly on a support by passing an electric current. The authors focus on pure polypyrrole, rather than mixtures with metals or carbon, to answer a simple but previously underexplored question: as you grow this polymer layer longer and make it thicker, how exactly do its shape and internal structure change, and what does that do to energy storage?
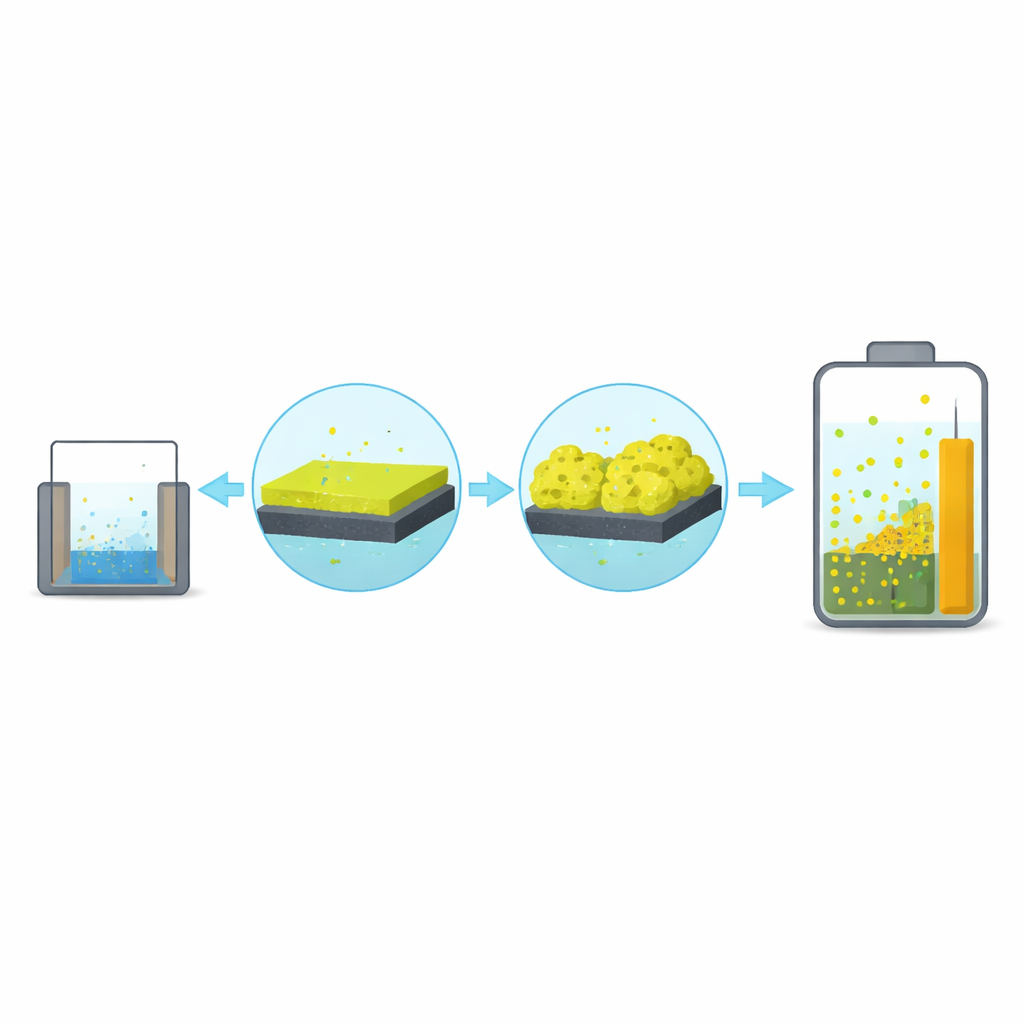
Watching the Electrode Grow Over Time
To investigate this, the team deposited polypyrrole onto graphite using an electrochemical process, varying the deposition time from the equivalent of 5 cycles up to 50 cycles. Electron microscope images show that the film does not simply become uniformly thicker. At short times, the surface is dotted with small, pebble-like particles about 1–2 micrometers across. With more cycles, these particles grow and merge into much larger, porous, cauliflower-like clusters, reaching sizes above 20 micrometers at 50 cycles. The total mass of polypyrrole on the electrode increases steeply with time, suggesting the film develops multiple layers and a highly textured surface that can expose more area to the electrolyte.
Peering Inside the Material’s Bonds
Alongside visual images, the researchers used light- and electron-based probes to track how the internal chemistry of polypyrrole evolves. Raman spectroscopy, which measures the vibrations of chemical bonds, showed that as deposition time increases, signatures of structural “defects” become more pronounced. These defects are not necessarily harmful; in conducting polymers, they often act as sites where charge can be stored and moved. X-ray photoelectron spectroscopy confirmed how different carbon and nitrogen bonding environments change, indicating a transition from simpler carbon–carbon bonding to more fully developed polypyrrole ring structures. Together, these measurements show that longer growth times produce thicker films with more electroactive sites and a more complex internal network for charges to travel through.
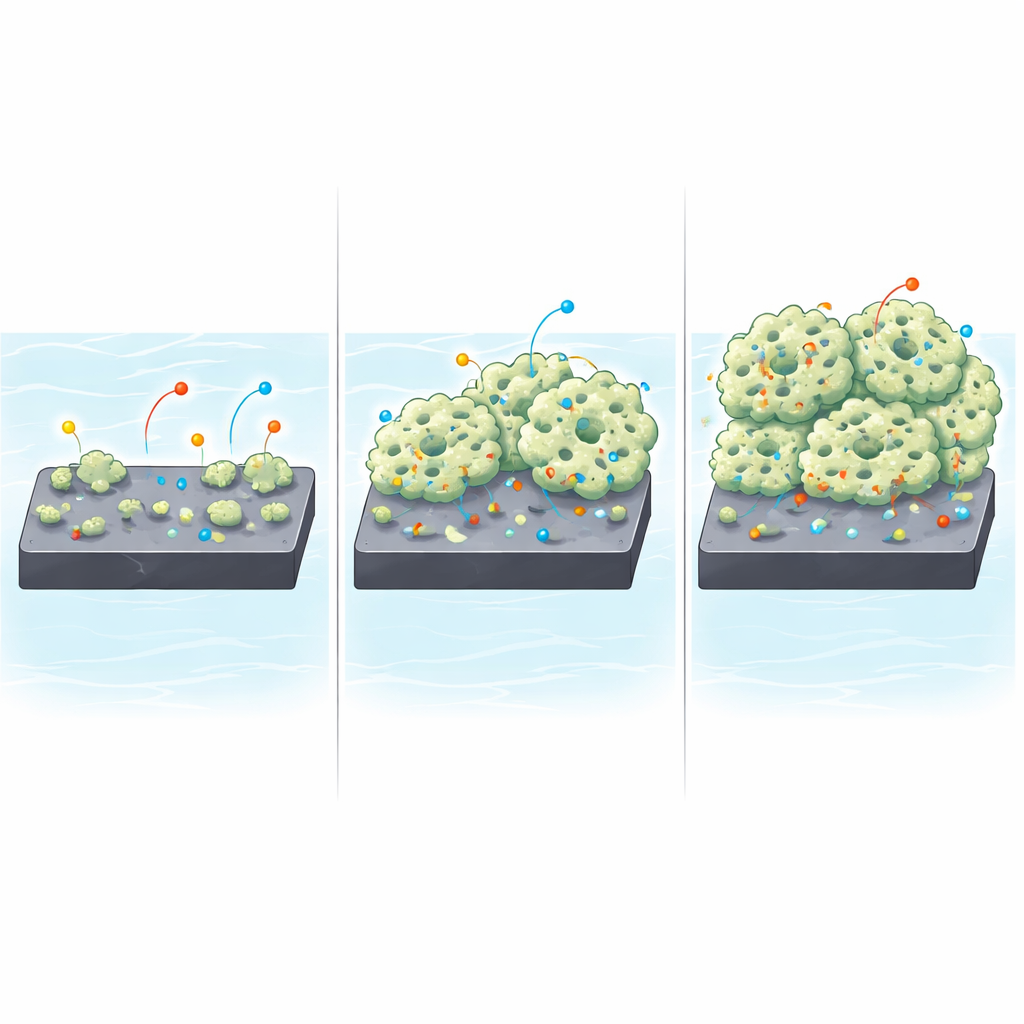
Balancing Easier Charge Storage with Harder Ion Travel
The true test of any electrode is how it behaves in an actual circuit. Using techniques that scan voltage back and forth and charge and discharge the electrodes at controlled rates, the team found that longer-grown polypyrrole films store significantly more charge per gram. In a three-electrode test cell, the 50-cycle film reached a specific capacitance of about 412 F/g, far surpassing thinner films. However, the same measurements also revealed trade-offs. Electrical impedance analysis showed that as the film thickens, the resistance associated with moving charge across the interface rises sharply, and ions must follow more tortuous paths through the porous structure. The diffusion behavior shifts from relatively straightforward ion motion in thin films to a regime known as Warburg diffusion in thicker ones, reflecting slower, more complex ion transport through the deep, cauliflower-like network.
From Lab Electrode to Working Device
To connect these findings to real-world use, the researchers built a symmetric “button cell” supercapacitor using two 50-cycle polypyrrole electrodes in a common salt solution. This simple device delivered an energy density of about 10 Wh per kilogram and a power density of about 146 W per kilogram at low current, values competitive with many polymer-based supercapacitors. At higher charging rates, the device could deliver more power but stored less total energy, reflecting the same diffusion limits seen in the electrode-level tests. Over 3000 charge–discharge cycles, the cell retained about 60% of its initial capacitance—moderate stability for a material known to swell and contract during operation.
What This Means for Better Supercapacitors
In everyday terms, this work shows that how long you let a polypyrrole film grow can make the difference between a modest and an impressive energy store. Longer growth creates heavier, more structured, cauliflower-like films with many more places to park charge, boosting capacitance and energy density. At the same time, ions have to work harder to reach all those sites, increasing resistance and gradually limiting performance at high power or over many cycles. Understanding and tuning this balance between accessible surface area and ion transport is key to designing the next generation of compact, fast-charging supercapacitors based on conducting plastics.
Citation: Pham, D., Gouafong, R., Irvin, J. et al. Evolution of polypyrrole electrode during electropolymerization and its effect on energy storage performance. Sci Rep 16, 11720 (2026). https://doi.org/10.1038/s41598-026-47559-7
Keywords: supercapacitors, polypyrrole, conducting polymers, electrode design, energy storage