Clear Sky Science · en
Genomic and biological characterization of lytic phages infecting Pseudomonas syringae associated with almond bacterial blast
Why tiny viruses matter for almond lovers
Almonds are one of the world’s most valuable nut crops, but in California’s orchards a microscopic battle is underway. A bacterial disease called almond bacterial blast can kill shoots, ruin blossoms, and strip yield from young trees. Traditional chemical sprays are losing their punch and raising environmental concerns. This study explores a very different ally: viruses that specifically infect and kill the problem bacteria, offering a potential green tool to protect orchards.
A growing disease threat in the orchard
The culprit behind almond bacterial blast is a strain of Pseudomonas syringae, a bacterium that attacks many fruits, vegetables, and tree crops. In California’s Central Valley, cold, wet springs have fueled recent outbreaks that caused up to 40% crop loss in some orchards. Growers mainly rely on copper-based sprays and a single antibiotic, but resistance to these products is spreading, and repeated use can harm beneficial microbes and leave residues in soil. The authors argue that to sustain almond production, growers will need biological tools that work with, rather than against, the orchard ecosystem.
Recruiting natural enemies of bacteria
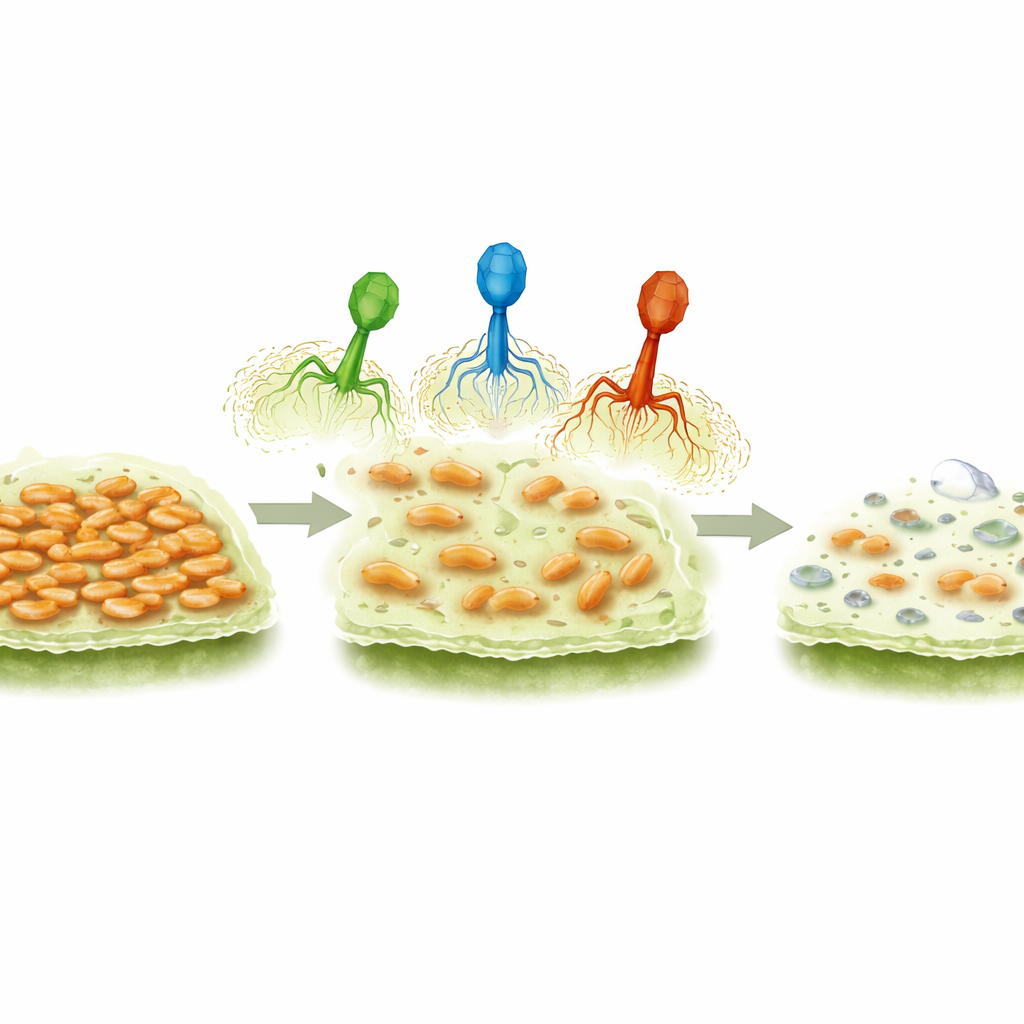
Bacteriophages—literally “bacteria eaters”—are viruses that infect only bacteria, leaving plants, animals, and people unharmed. The research team collected soil and sewage samples and fished out three phages that aggressively attack almond-associated P. syringae. Under the electron microscope, all three showed the classic look of a “tailed” virus: an angular head that holds DNA and a short tail used to latch onto bacteria. Despite this shared shape, the phages formed different patterns of clear spots, or plaques, when grown on bacterial lawns, hinting at distinct ways of interacting with their hosts.
How well the phages hunt and kill
The scientists tested how broadly each phage could infect a collection of 36 Pseudomonas strains from almonds and other crops. All three were highly effective against the main almond disease group, but were less successful on more distant relatives from crops like bean or radish, and did not infect unrelated Pseudomonas species at all. In liquid culture, the phages rapidly knocked down bacterial numbers, especially at higher starting doses, though some bacteria rebounded later—likely survivors that had become resistant. Importantly, when applied to slimy bacterial layers called biofilms, which help P. syringae cling to plant surfaces, the phages stripped away 60–96% of this buildup. One phage, called Mission, consistently did the best job, matching its distinctive halo-like plaques that suggest it can dissolve sticky protective coatings around cells.
Built for cold, tough environments
Because field performance depends on weather, the team examined how temperature and acidity affected the phages. All three stayed active for at least a day at cool to mild temperatures, matching conditions when almond bacterial blast is most severe. One phage held up better at warmer temperatures, while another tolerated a wider range of acidity, hinting that a mix of phages might better handle fluctuating orchard conditions. Genetic sequencing showed that each phage carries a compact, streamlined genome devoted to attacking and bursting its host, with no genes linked to antibiotic resistance or other harmful traits. Two of the phages closely resemble known virus types, while the third appears genetically distinct enough that it may represent a new branch on the phage family tree.
What this could mean for future treatments
This work does not yet test phage sprays directly on almond trees, but it provides a detailed blueprint of three promising candidates: what they infect, how fast they kill, how stable they are, and how their genes are organized. To a lay reader, the key message is that there are naturally occurring viruses that home in on the bacteria responsible for almond bacterial blast, punch holes in both free-floating cells and protective biofilms, and appear safe from a genetic standpoint. With further testing in orchards and careful design of phage mixtures, these microscopic hunters could one day help farmers protect almonds while reducing reliance on chemical pesticides.
Citation: Hoang, C.V., Fan, J., Lee, H. et al. Genomic and biological characterization of lytic phages infecting Pseudomonas syringae associated with almond bacterial blast. Sci Rep 16, 11657 (2026). https://doi.org/10.1038/s41598-026-47496-5
Keywords: almond disease, bacteriophages, biological control, Pseudomonas syringae, plant health