Clear Sky Science · en
Biomass-derived carbon quantum dots for the fabrication of a durable, self-cleaning, and corrosion-resistant superhydrophobic coating on steel
Why keeping steel clean and rust‑free matters
From bridges and ships to factory tanks and skyscrapers, modern life leans heavily on steel. Yet steel has a weakness: when water and salt reach its surface, rust soon follows, bringing high maintenance costs and safety risks. This study reports a new way to armor steel with an ultra water‑repelling, self‑cleaning coating made from plant waste. The work blends ideas from nature’s lotus leaf with green chemistry, aiming to keep metal surfaces dry, clean, and corrosion‑free without relying on persistent fluorinated chemicals.
Turning roadside trees into smart nano‑building blocks
The researchers started with an abundant landscaping tree, Conocarpus lancifolius, whose leaves are often discarded as waste. They converted these leaves into carbon quantum dots—tiny carbon particles only a few billionths of a meter across. These dots carry many oxygen‑ and nitrogen‑based chemical groups on their surface, which help them disperse evenly in water and interact strongly with metals. Using techniques such as infrared spectroscopy, X‑ray diffraction, electron microscopy, and surface chemical analysis, the team confirmed that the dots are small, mostly amorphous carbon particles enriched with these reactive groups. In other words, the leaves were successfully turned into a versatile nano‑ingredient that can be blended into protective coatings.
Building a lotus‑like skin on steel
To protect the steel, the team used an industrially familiar process called electrodeposition to lay down a thin nickel‑based layer, either with or without the carbon dots mixed in. Afterward, they dipped the rough nickel surface into a solution of stearic acid, a biodegradable fatty acid similar to those found in soaps and foods. This final step lowers the surface energy, encouraging water to bead up instead of spread out. The crucial difference emerges when the biomass‑derived carbon dots are present in the bath: they act as countless tiny nucleation points during metal deposition. Instead of growing a few large, smooth nickel grains, the process produces a dense forest of fine grains and nano‑scale bumps, creating the kind of multi‑level roughness that nature uses on lotus leaves to repel water.
How nano‑texture boosts water repellency and toughness
High‑resolution imaging of the finished coatings shows how dramatically the carbon dots reshape the surface. Without them, the steel is covered by relatively large, sparsely spaced mounds; with them, the surface becomes a tightly packed landscape of much smaller features and sharper peaks. Atomic force microscopy reveals that the overall roughness nearly doubles, and this change translates directly into performance: the water contact angle rises to about 167 degrees—meaning droplets are almost perfect spheres—and the tilt angle needed for a drop to roll off falls to about 1 degree. In tests, the dot‑enhanced coating kept its extreme water repellency after being dragged 900 millimeters over sandpaper, whereas the dot‑free version failed at around 400 millimeters. The enhanced coating also stayed super‑repellent in harsh solutions spanning the full pH range from strongly acidic to strongly alkaline.
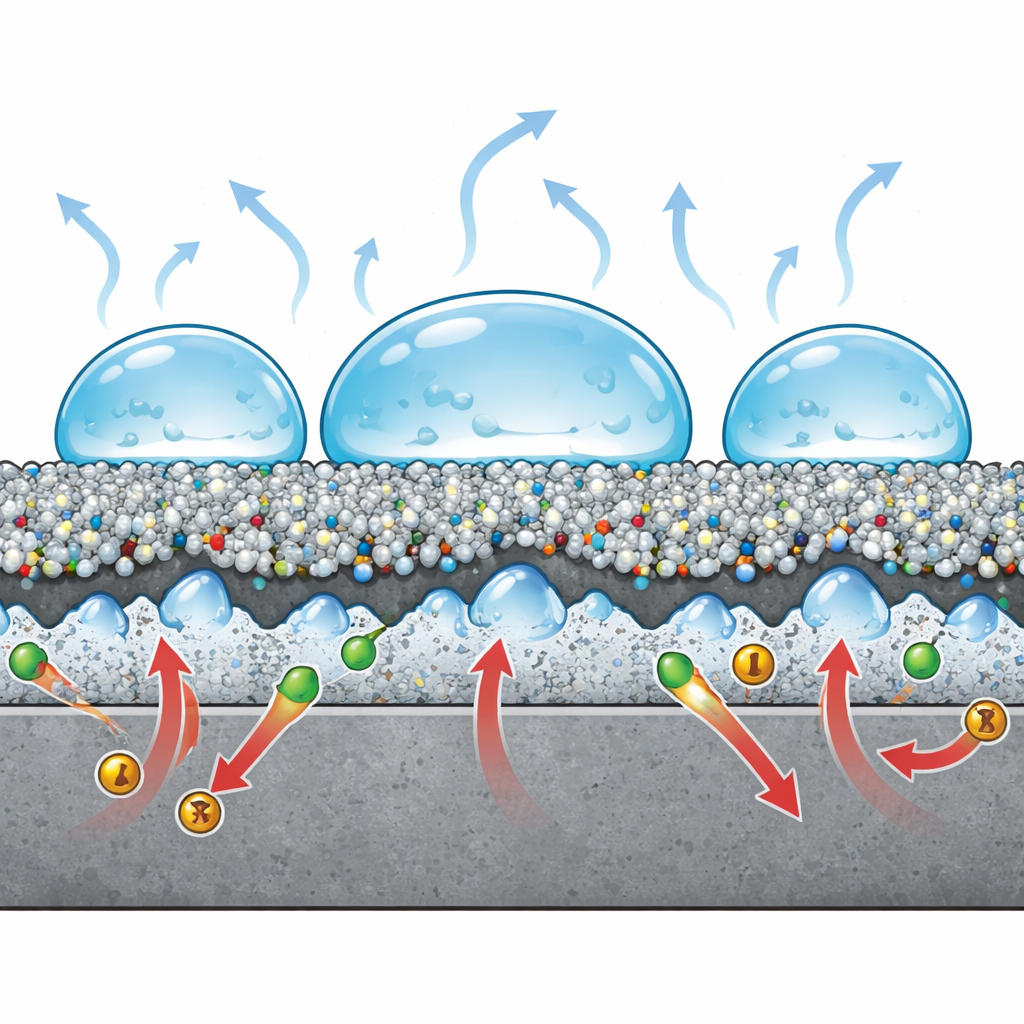
Blocking rust with air pockets and a tighter barrier
To see how well this lotus‑like skin shields steel from rust, the authors immersed coated and uncoated samples in salty water and measured how easily electrical current could pass—a proxy for corrosion. Both electrochemical impedance tests and controlled polarization scans showed that adding carbon dots greatly increases the resistance to corrosion while lowering the effective area exposed to the electrolyte. The textured coating traps air in its crevices, so droplets touch only a small fraction of the solid surface, making it harder for aggressive ions such as chloride to reach the metal. Chemical analysis after immersion found fewer chloride signals on the dot‑rich coating than on the control, supporting the idea that the new layer acts as both a physical and electrostatic barrier. Overall, the corrosion protection efficiency rose to about 93 percent, compared with roughly 79 percent for the similar coating lacking carbon dots.
What this means for real‑world surfaces
For a non‑specialist, the message is straightforward: by combining plant‑based nanomaterials with a standard metal‑plating step and a simple fatty‑acid treatment, the researchers created a tough, self‑cleaning, and strongly rust‑resistant skin for steel. Water and dirt roll off easily, the surface endures abrasion and harsh chemicals, and the need for problematic fluorinated additives is avoided. If scaled up, this approach could help protect infrastructure, marine equipment, and outdoor structures more sustainably, turning common green waste into a high‑value ingredient that keeps critical metal surfaces drier, cleaner, and safer for longer.
Citation: Mohamed, M.E., Abd-El-Nabey, B.A. & Ezzat, A. Biomass-derived carbon quantum dots for the fabrication of a durable, self-cleaning, and corrosion-resistant superhydrophobic coating on steel. Sci Rep 16, 13897 (2026). https://doi.org/10.1038/s41598-026-47261-8
Keywords: superhydrophobic coatings, corrosion protection, carbon quantum dots, biomass recycling, self-cleaning surfaces