Clear Sky Science · en
Amelioration of gentamicin-induced acute kidney injury by trifluoperazine: in vivo mechanistic insights
Why this matters for everyday medicine
Many people in intensive care rely on strong antibiotics to survive life-threatening infections. One of the most widely used drugs, gentamicin, is highly effective against dangerous bacteria but can seriously damage the kidneys, sometimes within only a few days. This study explores an unexpected helper: trifluoperazine, a long‑used antipsychotic medicine, and asks whether it can be repurposed to shield the kidneys from gentamicin’s harmful side effects.
A powerful antibiotic with a hidden cost
Gentamicin is a workhorse drug in hospitals because it kills tough, gram‑negative bacteria quickly and cheaply, and can be combined with other antibiotics for a stronger effect. The downside is that up to one in five critically ill patients receiving gentamicin for more than three days may develop acute kidney injury, a sudden loss of kidney function that can progress to chronic disease. Gentamicin builds up in kidney cells, where it generates excessive “chemical rust” in the form of reactive oxygen species and disrupts the cells’ normal cleanup and repair processes. Over time, this chain reaction inflames and scars kidney tissue.
Testing an old drug in a new role
The researchers used a mouse model to see whether pretreating animals with trifluoperazine could blunt gentamicin’s kidney toxicity without harming the kidneys on its own. They chose doses that closely mirror those used in people, then compared four groups: untreated controls, gentamicin alone, trifluoperazine alone, and trifluoperazine given before and during gentamicin. They measured standard blood markers of kidney function, examined kidney tissue slices under the microscope, and probed key molecular signals linked to stress, inflammation, cell recycling, and cell death.
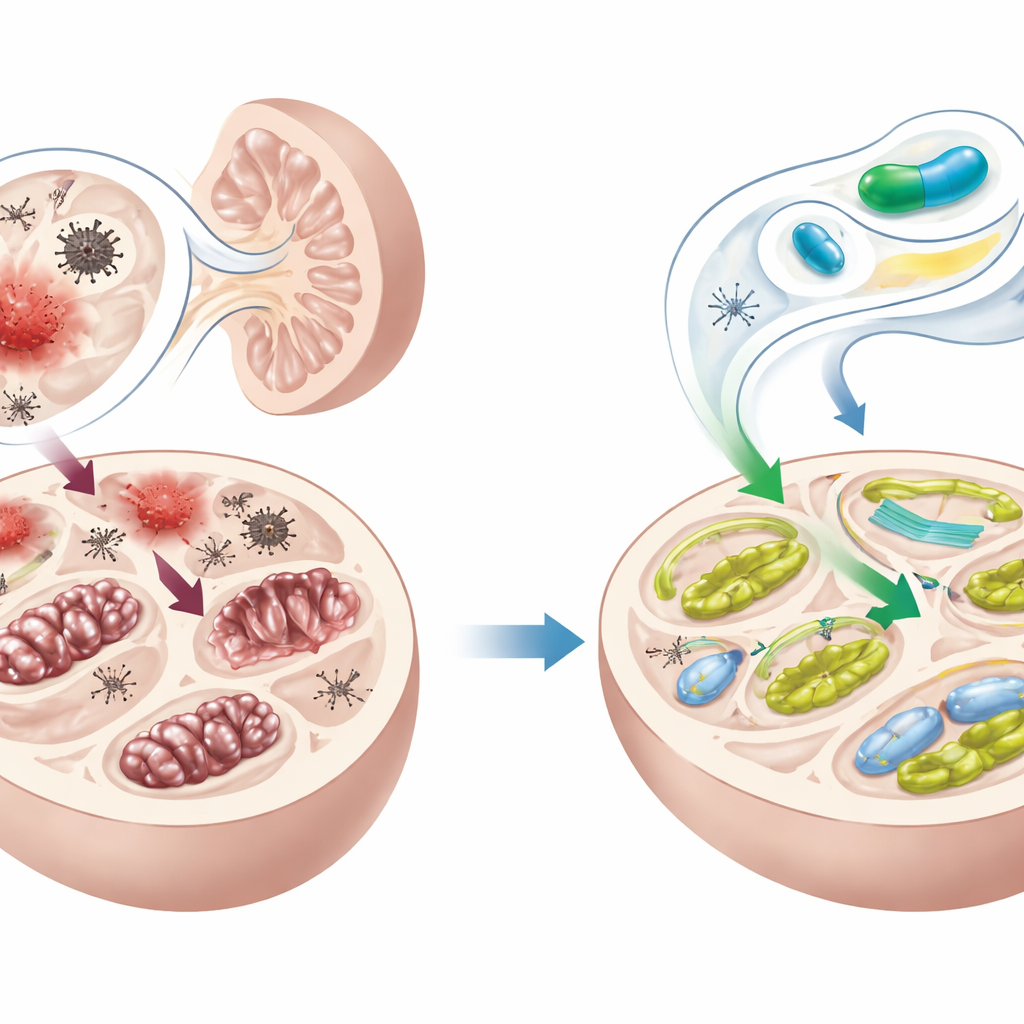
Protecting kidney function and structure
As expected, gentamicin alone caused clear kidney damage: blood levels of urea and creatinine rose, and microscopic examination showed distorted filtering units, swollen and degenerating tubules, leaky blood vessels, and bleeding in the surrounding tissue. In sharp contrast, kidneys from mice pretreated with trifluoperazine looked much closer to normal. Their blood tests improved, and detailed scoring of tissue injury showed far less tubular, glomerular, endothelial, and interstitial damage. Trifluoperazine alone did not worsen kidney function or structure, suggesting that at the tested dose it was safe for the kidneys.
Calming stress, inflammation, and cellular chaos
The team then asked what was happening inside the kidney cells. Gentamicin greatly increased markers of oxidative stress, indicating heavy damage to fats and other cellular components, and turned on “emergency” genes that respond to this stress. It also disrupted autophagy, the cell’s internal recycling system, causing waste‑handling proteins to pile up. At the same time, gentamicin activated inflammatory switches (such as NF‑κB and the NLRP3 inflammasome), stress‑linked signaling routes (ERK1/2), and executioner proteins that drive cell suicide. Trifluoperazine pretreatment softened each of these blows: it reduced oxidative damage, restored more normal autophagy, muted inflammatory and fibrotic signaling, dampened ERK1/2 and NLRP3 activity, and lowered markers of programmed cell death.
What this could mean for future treatments
Altogether, the findings suggest that trifluoperazine can act like a broad‑spectrum shield for the kidneys during gentamicin therapy, working on multiple fronts at once—reducing chemical stress, preserving the cell’s cleanup systems, and quieting inflammatory and death pathways. Although this work was done in mice and does not yet prove safety or benefit in humans, it opens the door to repurposing a familiar psychiatric drug as a kidney‑protective add‑on for patients who urgently need prolonged courses of powerful antibiotics.
Citation: Goda, A.E., Almosilhy, N.A. & El-Mahdy, N.A. Amelioration of gentamicin-induced acute kidney injury by trifluoperazine: in vivo mechanistic insights. Sci Rep 16, 12896 (2026). https://doi.org/10.1038/s41598-026-47243-w
Keywords: acute kidney injury, gentamicin, trifluoperazine, drug repurposing, nephrotoxicity