Clear Sky Science · en
Fabrication of a ZGTPM nanocomposite membrane for the synergistic removal of tetracycline via size selective sieving and photocatalytic degradation
Why Cleaning Antibiotic-Tainted Water Matters
Traces of antibiotics are now routinely found in rivers, lakes, and even treated wastewater. One common drug, tetracycline, is used so widely in medicine and agriculture that it can build up in the environment, fostering drug-resistant bacteria and harming aquatic life. This paper describes a new type of water-cleaning membrane that not only filters tetracycline out of water but also helps break it down under light, offering a powerful tool for safer water and slowing the spread of antibiotic resistance.
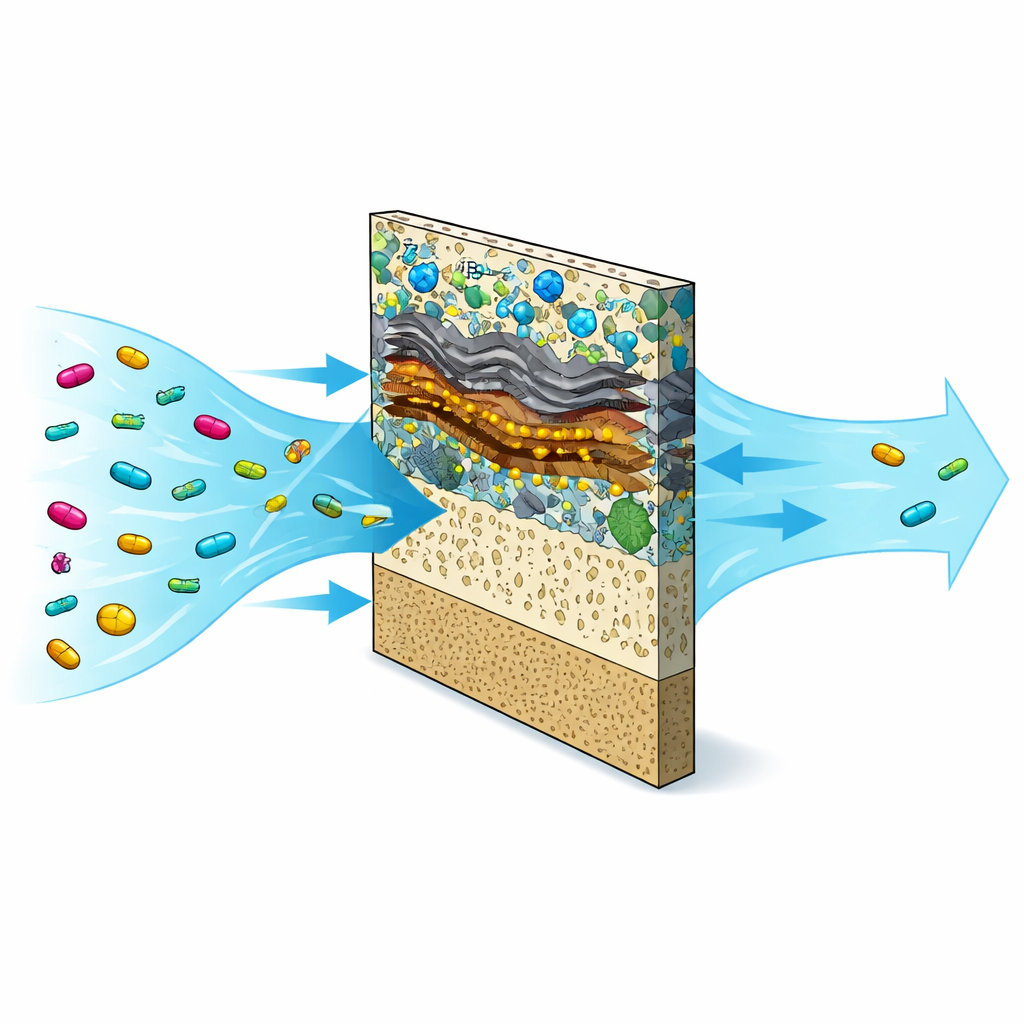
A Smart Filter Built from Advanced Materials
The researchers designed a composite membrane they call ZGTPM, built from several cutting-edge materials working together. At its core is a standard plastic support, but the active layer is packed with tiny porous crystals (ZIF-8), sheet-like carbon (graphene oxide), ultra-thin metal carbides (MXene), and nanosized titanium dioxide particles. Each ingredient brings a special ability: ZIF-8 offers countless tiny pores, graphene oxide and MXene make the surface highly water-loving and electrically conductive, and titanium dioxide nanoparticles act as miniature light-driven cleaners. Combined, these components create a thin film that is both highly permeable to water and strongly interactive with tetracycline molecules.
From Polluted Water to Clean Flow
When tetracycline-laden water is pushed through this membrane, two things happen at once. First, the antibiotic molecules are physically trapped and held on the membrane surface and within its pores, thanks to size-based sieving and sticky interactions such as hydrogen bonding and stacking between ring-shaped molecules and flat carbon sheets. This adsorption step alone removes about a third of the drug from the water in darkness. Second, when the membrane is illuminated with simulated sunlight, the titanium dioxide and MXene work together to transform light energy into reactive species that attack the captured antibiotic. In trials, the membrane removed more than 99.5% of tetracycline, with a measured adsorption capacity much higher than many existing filters. At the same time, pure water flowed through about 80% faster than through the unmodified base membrane, meaning the system cleans water efficiently without sacrificing throughput.

How Light Helps Destroy Antibiotic Molecules
Under light, the titanium dioxide nanoparticles in the membrane generate energetic electrons and “holes.” MXene and graphene oxide act like highways and sinks for these charges, helping to separate and guide them instead of letting them cancel each other out. This charge traffic leads to the creation of highly reactive oxygen-based molecules in the surrounding water. These species attack tetracycline that has already been concentrated on the membrane, breaking its complex structure into smaller fragments and partially mineralizing it into simpler compounds. Careful experiments in the dark and under light allowed the team to separate the roles of capture and destruction: about one third of the overall removal came from adsorption, while roughly two thirds came from light-driven breakdown. This synergy prevents the membrane’s surface from quickly becoming saturated and supports long-term cleaning performance.
Built to Last in Realistic Conditions
Beyond initial performance, the team tested how well the membrane stood up to repeated use and challenging water chemistry. After five full cycles of use and simple cleaning with alcohol and water, the membrane still removed over 97% of tetracycline, and its water flow remained high, suggesting limited clogging. Microscopy and spectroscopy showed that the structure and chemical groups remained stable, and tests for zinc and titanium in the treated water detected only minute amounts, well below safety limits. The membrane also handled a broad range of pH values, salt levels, and temperatures with only slight losses in efficiency. Even in real municipal wastewater spiked with tetracycline, it removed more than 93% of the antibiotic, despite competing organic matter and dissolved ions.
What This Means for Future Water Treatment
Taken together, these results show that the ZGTPM membrane can both catch and partly destroy a stubborn antibiotic under sunlight-like conditions, while staying robust and reusable. By combining porous crystals, carbon sheets, metal carbides, and light-activated nanoparticles in a single thin layer, the researchers created a compact, energy-efficient device that tackles pollution in multiple ways at once. With further optimization and scale-up, such multifunctional membranes could help wastewater plants and industrial facilities more effectively strip antibiotics from water, reducing ecological damage and the pressure that drives antibiotic resistance.
Citation: El-Sawaf, A., Nassar, A.A., Hammouda, G.A. et al. Fabrication of a ZGTPM nanocomposite membrane for the synergistic removal of tetracycline via size selective sieving and photocatalytic degradation. Sci Rep 16, 12582 (2026). https://doi.org/10.1038/s41598-026-47068-7
Keywords: antibiotic removal, photocatalytic membrane, tetracycline, water purification, nanocomposite materials