Clear Sky Science · en
Tailoring the microstructure, optical, and magnetic characteristics of Co0.6Zn0.4Fe2O4 nanoferrites through Ni²⁺–Al³⁺ co-doping
Why tiny magnets matter
From faster electronics to better medical scanners, many modern technologies rely on tiny magnetic particles called nanoferrites. This study explores how carefully swapping some of the metal atoms inside these particles lets scientists fine-tune how they respond to light and magnetic fields. By learning to “dial in” these properties without changing the basic structure, researchers move closer to custom-made materials for sensors, high‑frequency circuits, and shielding sensitive devices from electromagnetic noise.
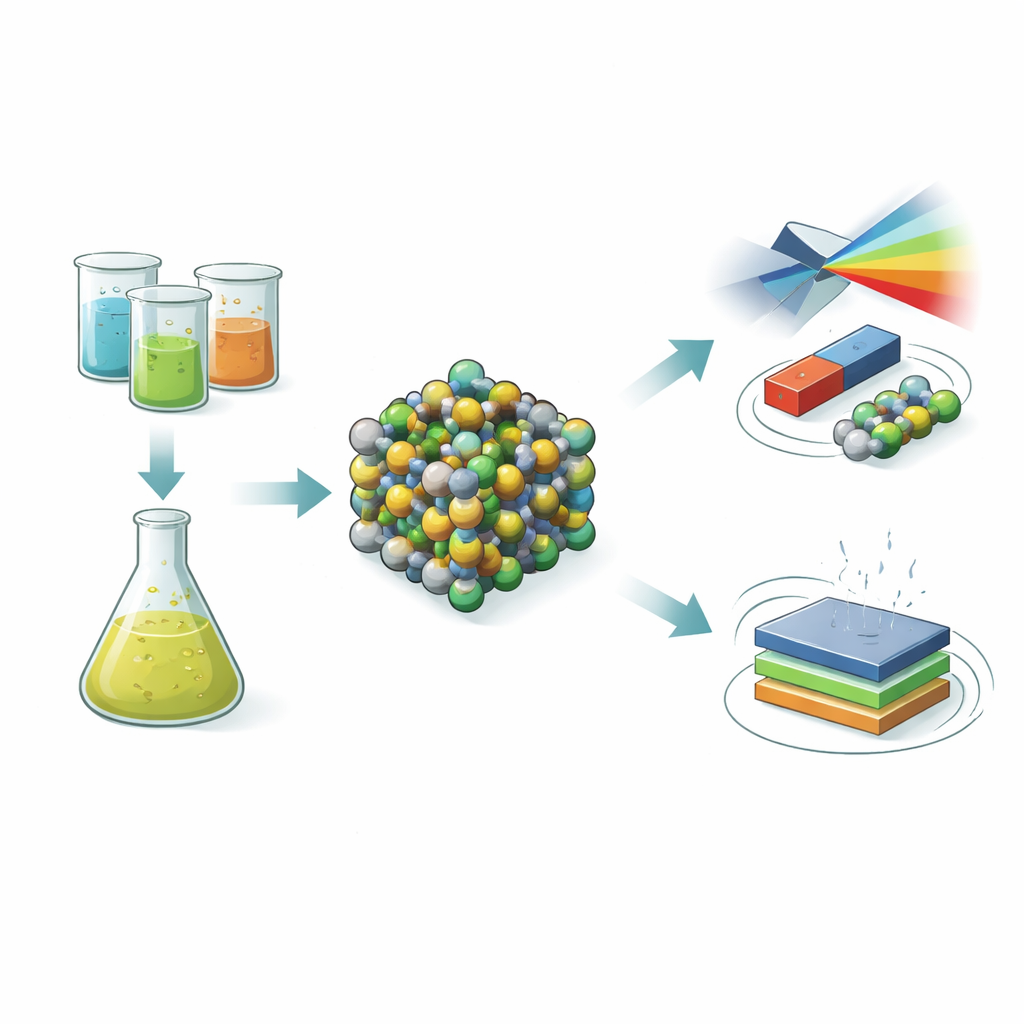
Building tiny mixed-metal particles
The researchers focused on a well-known family of materials called spinel ferrites, where iron atoms share a crystal framework with other metals such as cobalt and zinc. They started with cobalt–zinc ferrite nanoparticles and then gradually replaced a portion of the cobalt and zinc with nickel and aluminum. This process, known as co‑doping, was done using a simple wet-chemistry route called co‑precipitation, which lets them control particle size and composition at the nanoscale. After mixing metal salt solutions, adjusting the acidity, heating, washing, and annealing, the team obtained a series of powders in which the amount of nickel and aluminum increased step by step.
Checking the crystal framework and size
To see what had changed inside the particles, the team used a suite of structural tools. X‑ray diffraction confirmed that all samples kept the same basic cubic spinel framework, meaning the overall crystal “scaffold” remained intact even as metals were swapped. However, the unit cell—the repeating building block of the crystal—expanded slightly, and the average crystallite size shrank from about 16 to 11 nanometers as more nickel and aluminum were added. Electron microscopy images showed mostly spherical or cubic nanoparticles that became more uniformly cubic and slightly smaller with co‑doping. Calculations based on the diffraction data revealed increasing internal strain and defect density, signs that the atomic lattice was becoming more distorted as different-sized ions were introduced.
Watching how light and bonds respond
The optical behavior of the particles was tested using ultraviolet–visible spectroscopy. As nickel and aluminum content increased, the main absorption edge shifted toward shorter wavelengths—a blue shift—meaning the material’s bandgap energy grew larger. This trend is consistent with smaller particle size and a cleaner, less disordered electronic structure. A parameter called Urbach energy, which tracks disorder in the energy levels, decreased, supporting this picture of improved crystalline order. Infrared and Raman measurements, which probe how atoms vibrate, showed two key metal–oxygen vibration bands shifting to higher frequencies and becoming stronger. This indicates stiffer, shorter bonds and subtle rearrangement of metal ions between different sites in the crystal framework, backed up by X‑ray photoelectron spectroscopy that directly detected a mix of iron charge states and the preferred locations of the various metals.
Tuning soft magnetism for devices
The team then examined how these structural and optical changes affected magnetism. All samples behaved as soft magnets: they reached high saturation magnetization values (around 51–58 electromagnetic units per gram) but could be flipped with relatively small applied fields. As nickel and aluminum levels rose, the magnetization, remanent magnetization, and coercive field all decreased, and particles became smaller. Detailed modeling of how magnetization approached saturation showed that the magnetocrystalline anisotropy—a measure of how strongly spins are “locked” to certain directions in the lattice—dropped noticeably with co‑doping. This weakening arises mainly because nickel and especially non‑magnetic aluminum dilute cobalt-rich sites that normally provide strong directional locking, while added lattice defects and surface spin tilting further soften the magnetic response. The end result is a material that is magnetically easier to switch while still retaining substantial magnetization.
What this means for future technologies
Overall, the study shows that by carefully co‑doping cobalt–zinc ferrite nanoparticles with nickel and aluminum, scientists can subtly stretch the crystal lattice, shrink particle size, stiffen atomic bonds, widen the optical bandgap, and reduce magnetic hardness—all without destroying the underlying structure. For non-specialists, the key message is that swapping a small fraction of atoms acts like turning multiple control knobs at once: brightness response, magnetic softness, and structural stability can all be tuned together. Such finely engineered nanoferrites are promising building blocks for spin-based electronics, high‑frequency inductors, magnetic sensors, and coatings that quietly soak up unwanted electromagnetic interference.
Citation: Rekaby, M., Ahmed, M., Awad, R. et al. Tailoring the microstructure, optical, and magnetic characteristics of Co0.6Zn0.4Fe2O4 nanoferrites through Ni²⁺–Al³⁺ co-doping. Sci Rep 16, 14046 (2026). https://doi.org/10.1038/s41598-026-46866-3
Keywords: spinel ferrite nanoparticles, magnetic nanomaterials, cation doping, optical bandgap tuning, electromagnetic shielding