Clear Sky Science · en
Development of a novel external fixation device for inducing knee arthrofibrosis in rats
Why stiff knees matter
After injuries or surgeries, some people develop a stubborn stiffness in the knee that makes even simple movements painful or impossible. This condition, called joint contracture, can rob patients of their independence and is notoriously hard to treat. To test new therapies, scientists rely on animal models that mimic what happens in human joints—but building a realistic, safe model in small animals like rats has been surprisingly difficult. This study introduces a clever new way to stiffen rat knees without surgery, using a moldable plastic brace, opening the door to better research on how and why joints become locked.
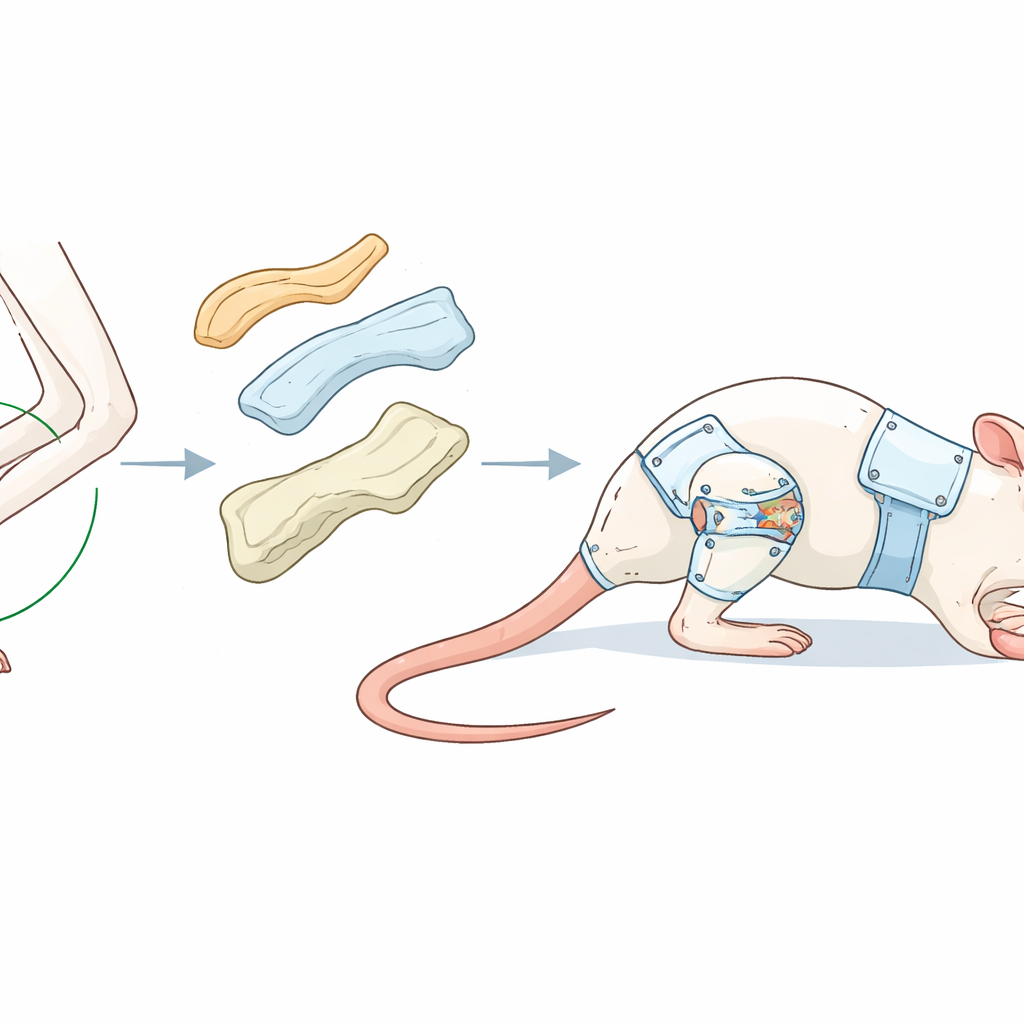
A new way to hold the knee still
The authors focused on arthrofibrosis, a form of joint scarring that develops when soft tissues around a joint thicken and tighten. In people, this often follows long periods of immobilization in a cast or brace. Existing animal models in rats usually involve surgery inside or around the joint, which creates its own trauma and inflammation and does not closely match the slow, non-injury pathway often seen in clinics. The team instead designed an entirely external device made from a thermoplastic polymer resin—small pellets that soften in hot water and harden again at room temperature—so it can be custom-shaped around each animal’s limb.
How the custom brace works
Under light anesthesia, the softened plastic was molded into a three-part "thigh–lower leg–trunk" frame that locked the rat’s knee in a bent position of about 135 degrees while leaving the ankle free to move. This semi-open structure hugged the irregular shape of the rat’s hindlimb without completely encasing it, reducing the risks of skin damage and poor circulation that plagued earlier cast-based methods. Once cooled, the brace became rigid and hard for the animals to chew through, yet could later be removed quickly by reheating small areas. The researchers checked the limbs frequently for swelling or signs of injury and found that, in most cases, a single application held for weeks with few complications.
What happens inside a stiffening joint
To see how this prolonged immobilization affected the knee, the scientists followed groups of rats for 1, 2, 4, or 6 weeks and compared them with a sham group that did not undergo long-term bracing. They measured how far the knee could be passively straightened and separated stiffness coming from muscles (myogenic) versus the joint structures themselves (arthrogenic) by surgically cutting the muscles at the end of the experiment. Early on, within the first two weeks, most of the loss of motion was due to changes in the muscles. Over time, however, the joint capsule and surrounding tissues became the main culprits, and total stiffness reached a plateau after about four weeks.
Peering at scars and cells
The team then examined the knees under the microscope. As immobilization time increased, the thin, loose capsule tissue at the back of the joint thickened markedly, with more cells, extra small blood vessels, and a denser web of collagen fibers—the structural protein that makes scar tissue tough. Special staining showed that collagen content rose steadily and leveled off by week four, mirroring the changes in movement. At the molecular level, cells in the joint lining ramped up production of two well-known fibrosis markers, α-SMA and COL1A1, at both gene and protein levels, again peaking around four weeks. Interestingly, by six weeks there were hints of slight remodeling, with a modest decrease in collagen fraction and a partial return of fat cells, suggesting the scarring process may evolve from an active to a more settled, chronic stage.
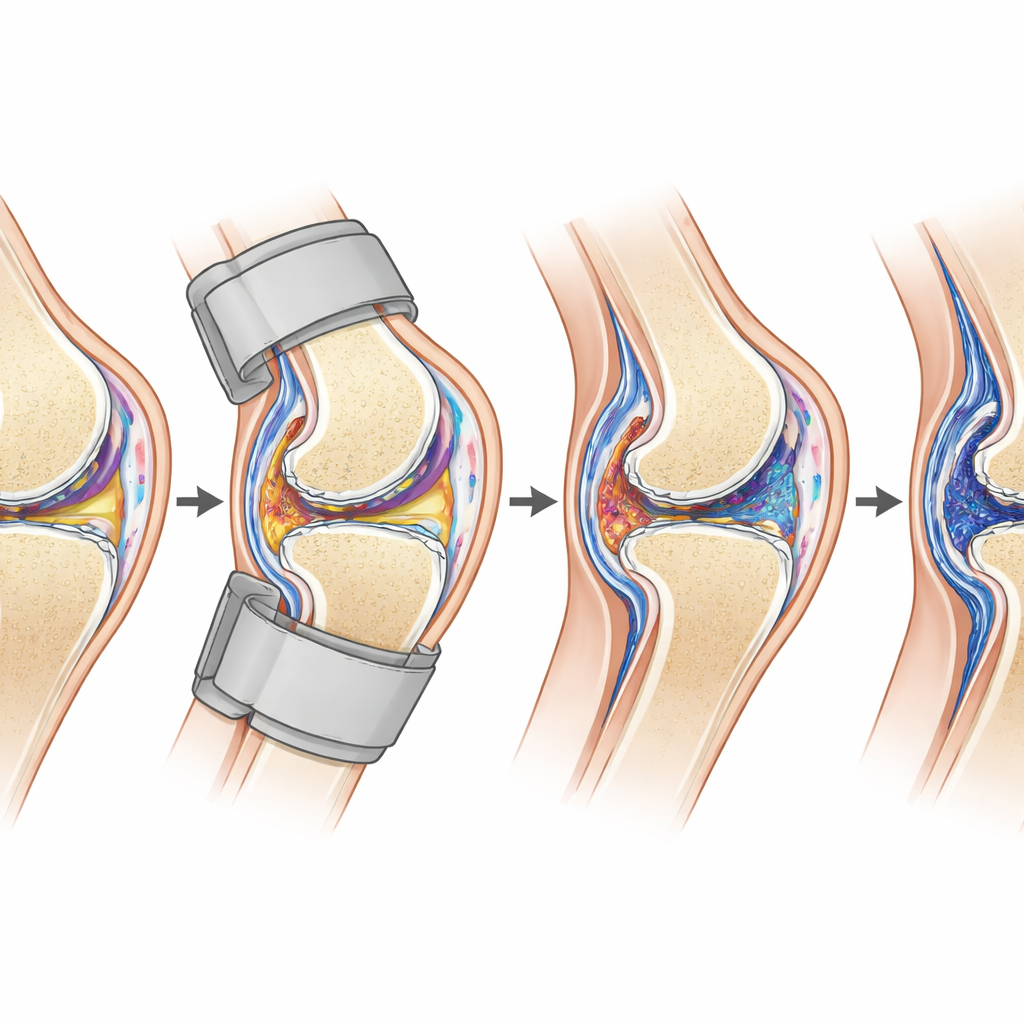
Safety checks beyond the knee
Because the brace material sits against the animal’s skin for weeks, the researchers also checked whether it harmed other organs. They looked at tissue slices from the heart, liver, spleen, lungs, and kidneys and ran standard blood chemistry tests for liver and kidney function. Across all time points, organ structure remained normal and blood values stayed within healthy ranges, indicating that the thermoplastic resin itself was biocompatible and did not introduce hidden toxicity or systemic stress.
What this means for future treatments
In everyday terms, this work shows that a simple heat-molded plastic brace can reliably make rat knees stiff in a way that closely resembles real-world, immobilization-related joint scarring—without the confounding effects of surgery. The model produces predictable loss of motion, thickened joint tissue, and molecular signatures of fibrosis, yet keeps the rest of the body unharmed. This makes it a powerful tool for testing new drugs, physical therapies, or devices aimed at preventing or reversing arthrofibrosis, ultimately helping researchers move closer to treatments that can spare patients from long-lasting knee stiffness.
Citation: Jia, J., Li, W. & Pan, Y. Development of a novel external fixation device for inducing knee arthrofibrosis in rats. Sci Rep 16, 11721 (2026). https://doi.org/10.1038/s41598-026-46668-7
Keywords: knee stiffness, arthrofibrosis, joint immobilization, rat model, fibrosis