Clear Sky Science · en
Enhancing and optimization for efficient removal of nickel ions by modified activated carbon
Why cleaning metal from water matters
Nickel is widely used to make batteries, electronics, and shiny metal coatings—but when it leaks into rivers and groundwater, it becomes a long‑lived poison. It can damage organs, disrupt DNA, and build up in fish and crops that people later eat. Many factories struggle to remove dissolved nickel from their wastewater efficiently and cheaply. This study explores a low‑cost, reusable filter material made from activated carbon that has been specially treated to grab nickel ions more strongly, offering a practical way to protect drinking water and the environment.
Turning common carbon into a smarter sponge
The researchers started with commercial activated carbon, a porous charcoal already used in many filters. By treating it with a mixture known as a Fenton solution—hydrogen peroxide and an iron salt in acidic water—they etched and oxidized the surface. This process created what they call modified activated carbon, or MAC, which has a larger internal surface area and many more oxygen‑containing chemical groups. These groups act like tiny hooks that can latch onto dissolved metal ions. Tests using infrared light, gas adsorption, and electron microscopy confirmed that the new material developed a highly porous, sponge‑like structure with nanoscale features and abundant reactive sites.
Finding the best recipe for nickel capture
Simply having a good material is not enough; how it is used in water also matters. The team systematically varied four key conditions: how long the water stays in contact with MAC, how acidic it is (pH), how much MAC is present, and how concentrated the nickel is. Using a statistical approach called response surface methodology, they ran 54 carefully chosen experiments and built a mathematical model that predicts how much nickel can be removed under any combination of these settings. The model matched measurements very well and showed that the amount of MAC used per liter of water is the single most important factor, followed by the starting nickel concentration. The best conditions turned out to be very acidic water (pH 1), 150 minutes of contact, a relatively small MAC dose of 0.2 grams per liter, and a nickel concentration of 125 milligrams per liter.
How the nickel sticks inside the filter
To understand what happens at the microscopic level, the researchers tracked how fast nickel was taken up and how it distributed itself on the MAC surface. The time‑dependent data fit a type of kinetic model that points to “chemisorption,” meaning the nickel forms stronger, more specific bonds rather than just weak physical attraction. Equilibrium tests showed that the behavior is consistent with nickel forming a mostly single layer on many different kinds of sites inside the pores. A third line of analysis suggested that the bonding involves ion‑exchange and electrostatic interactions, where nickel ions trade places with other charged species on the carbon surface. Together, these findings paint a picture of a highly structured, chemical locking‑in of nickel rather than simple sticking by chance.
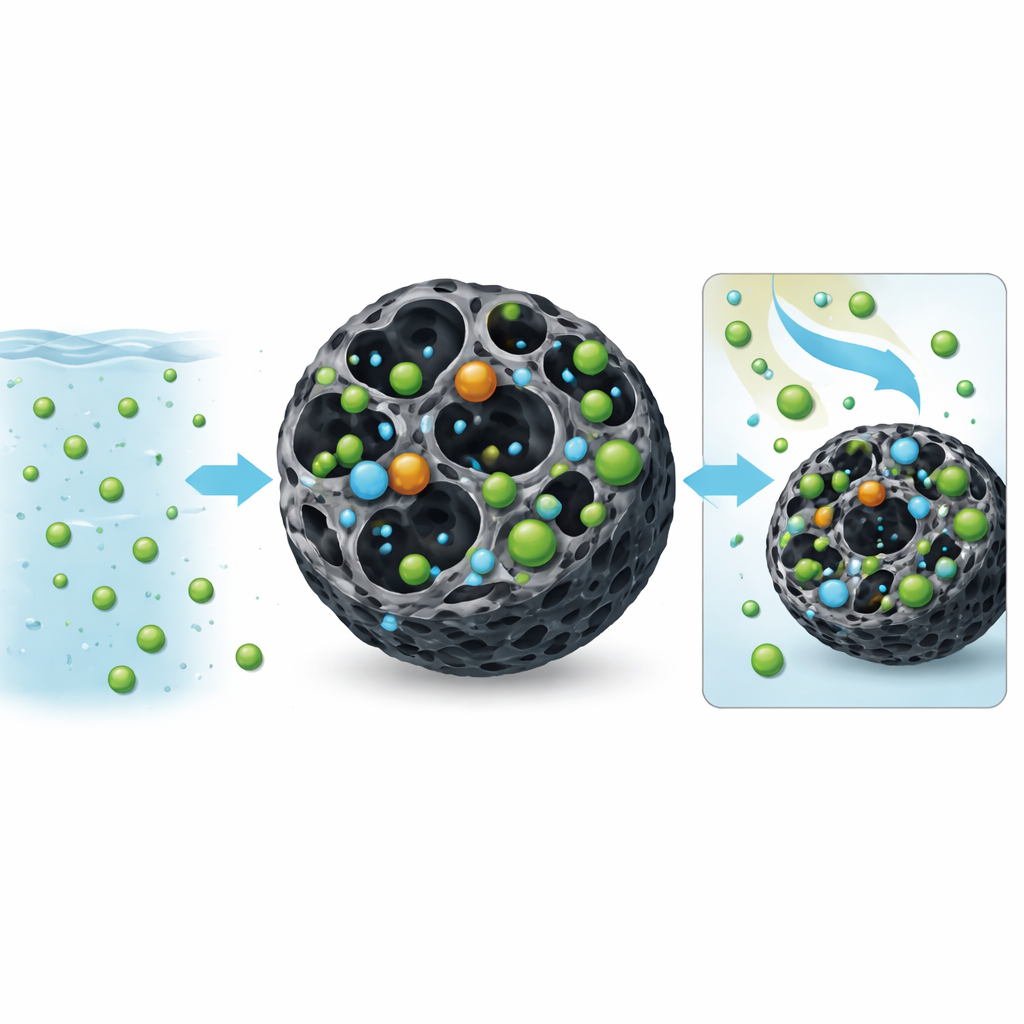
Reusing the filter and counting the cost
Any real‑world treatment must be affordable and reusable. The team repeatedly cycled the MAC through five rounds of nickel capture and acid rinsing. While performance declined slightly, the material still removed most of the nickel after several uses, retaining about 85% of its original capacity. A simple cost analysis, based on local prices for the carbon and chemicals plus electricity for stirring, showed that producing one kilogram of MAC costs under one US dollar—cheaper than many commercial activated carbons. That low price, combined with strong performance, makes it attractive for large‑scale wastewater plants.
What this means for safer water
In plain terms, the study shows that an inexpensive, modified form of charcoal can pull large amounts of toxic nickel out of water, again and again, using simple treatment steps. By fine‑tuning conditions like acidity, contact time, and how much material is used, engineers can reach very high removal levels with relatively small amounts of MAC. Because the material is cheap, robust in harsh acidic streams, and regenerable, it offers an appealing option for factories and municipalities seeking to cut heavy‑metal pollution before it reaches rivers, soil, and the food chain.
Citation: Abdel-Moniem, S.M., Mohammed, R., Ibrahim, H.S. et al. Enhancing and optimization for efficient removal of nickel ions by modified activated carbon. Sci Rep 16, 12700 (2026). https://doi.org/10.1038/s41598-026-46490-1
Keywords: nickel removal, activated carbon, wastewater treatment, heavy metals, water purification