Clear Sky Science · en
The effect of lateral π-extension on azulene-based molecules on surface studied by LT-STM
Why tiny tilted rings matter
Electronics are shrinking toward the scale of individual molecules, where the exact shape of a molecule can decide how it sticks to a surface and how easily it carries charge. This study looks at exotic, non‑hexagonal carbon rings built from the blue‑colored dye azulene and asks a simple question with big implications: if you slightly widen these molecules sideways, can you control how they arrange themselves and how reactive they become on metal surfaces?
Two near‑twins with a subtle twist
The researchers compared two closely related molecules: CPAT, made from three connected azulene units, and CPAT‑Ph, which adds three extra ring “wings” around the same core. Both were gently evaporated onto ultra‑clean gold and copper crystals in a vacuum and then examined with low‑temperature scanning tunnelling microscopy, a technique that can image and probe single molecules. Because CPAT and CPAT‑Ph share the same central framework but differ in their side rings, they form a good test case for how lateral extension changes sticking strength, motion, and electronic behavior on a surface.
Order versus motion on gold
On a gold surface, CPAT molecules behave like well‑trained dancers. They lock into specific sites on the gold reconstruction and spontaneously sort into tidy, single‑handed (homochiral) domains, where every molecule has the same left‑ or right‑handed twist. High‑resolution images show that this separation arises from subtle noncovalent forces between molecules, such as dipole interactions and steric fit, rather than from strong bonding. In striking contrast, the bulkier CPAT‑Ph molecules refuse to form such ordered patterns. They appear more weakly attached and more mobile, clustering in a disordered fashion across the gold without clear domains or a preferred twist, showing that the added side rings loosen the constraints imposed by the surface.
Shifting energy levels without stronger bonding
To see how these structural changes affect electrons, the team measured local spectra on individual molecules and on small assemblies. For CPAT, they identified characteristic occupied and empty energy levels, with images of the electron density revealing that certain states are concentrated on the outer azulene units and side groups. Importantly, these patterns hardly change when molecules are part of a chain, indicating that neighboring molecules do not strongly mix their electronic states. CPAT‑Ph shows a similar overall pattern, but its key occupied state shifts closer to the metal’s Fermi level, a sign that the laterally extended version couples differently to the surface. Yet even in this case, the electronic states remain largely confined to each molecule, confirming that lateral extension mainly tunes how the molecule talks to the metal, not how strongly it talks to its neighbors.
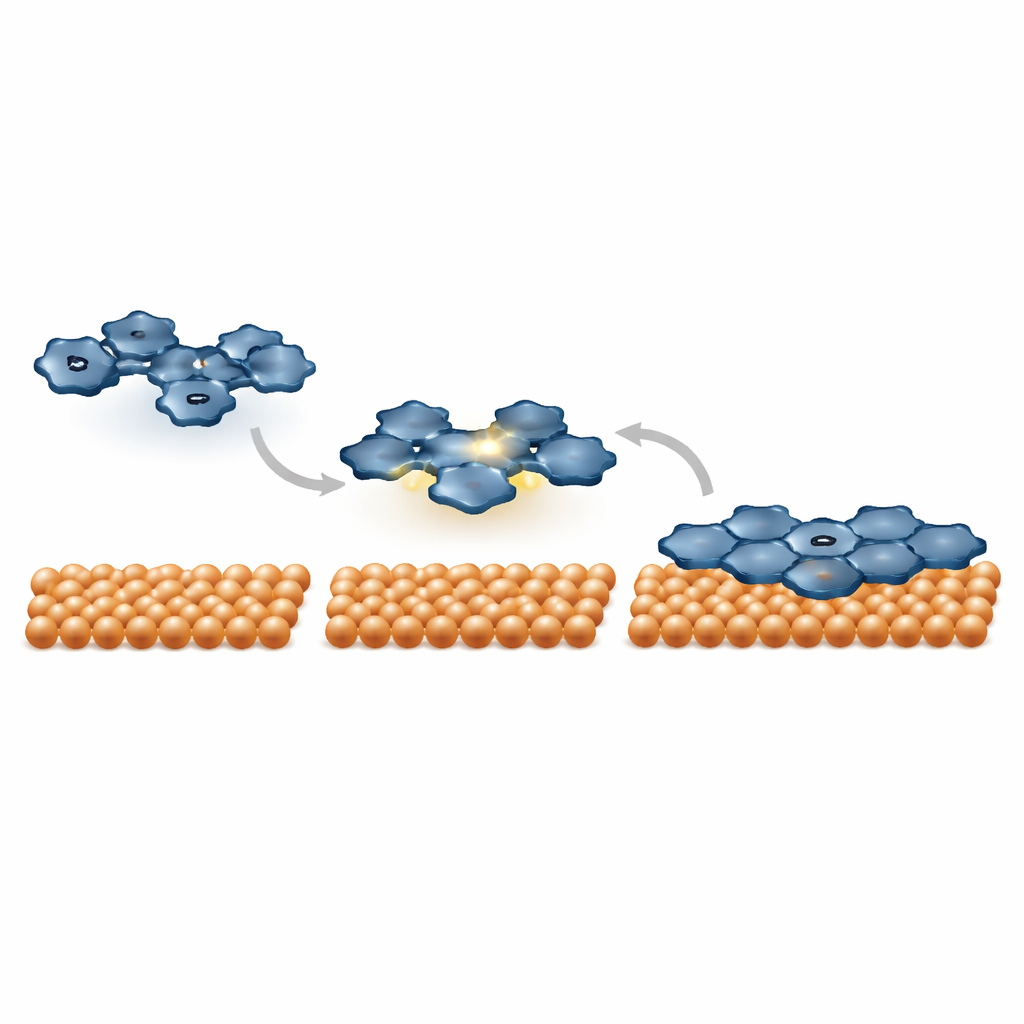
Heating up to build new carbon frameworks
When the samples are gently heated, both types of molecules on gold start to link in a rather disordered way, and some CPAT units even desorb, but no well‑defined new carbon networks emerge. The situation changes on a more reactive copper surface. There, CPAT‑Ph first adopts a slightly bent shape, indicating stronger interaction through its central ring. Upon further heating, the molecules both join together and flatten out as some of their rings fuse. High‑resolution images reveal a patchwork of fused five‑, six‑, seven‑, and eight‑membered carbon rings, signalling that several competing internal ring‑closing routes are active. These partially fused products are intermediate steps on the way to so‑called nanographenes, tiny graphene‑like fragments with deliberately distorted ring patterns.
What this means for future nano‑building
Overall, the work shows that simply extending a molecule sideways can act as a practical design knob: it weakens or strengthens how the molecule adheres to different metal surfaces, shifts its energy levels relative to the metal, and can open up or close off pathways to more complex carbon structures under heat. For future molecular electronics and surface‑grown carbon materials, this means that careful choice of side rings on non‑hexagonal frameworks like azulene offers a way to steer self‑assembly, preserve useful molecular states, and trigger on‑surface chemical reactions precisely where and when they are needed.
Citation: Sarkar, S., Khera, N., Au-Yeung, K.H. et al. The effect of lateral π-extension on azulene-based molecules on surface studied by LT-STM. Sci Rep 16, 11226 (2026). https://doi.org/10.1038/s41598-026-46150-4
Keywords: azulene molecules, scanning tunnelling microscopy, molecular self-assembly, nanographene synthesis, surface chemistry