Clear Sky Science · en
3-carbamoyl proxyl nitroxide attenuates CCl4-induced liver fibrosis in mice through antioxidant-inflammatory regulation of TLR4/NF-κB signaling pathway
Why this matters for everyday health
Scarring of the liver, known as fibrosis, is a silent process that can turn common problems such as fatty liver, viral hepatitis, or long-term alcohol use into life-threatening disease. Today there is still no drug approved specifically to halt or reverse liver scarring. This study explores a small antioxidant molecule, called 3-carbamoyl proxyl nitroxide (3-CP), that may protect the liver in two ways at once: by reducing damaging reactive molecules and by calming inflammatory signals that drive scarring. Using human liver cells and a mouse model, the researchers asked whether 3-CP could slow or even partially reverse liver fibrosis.
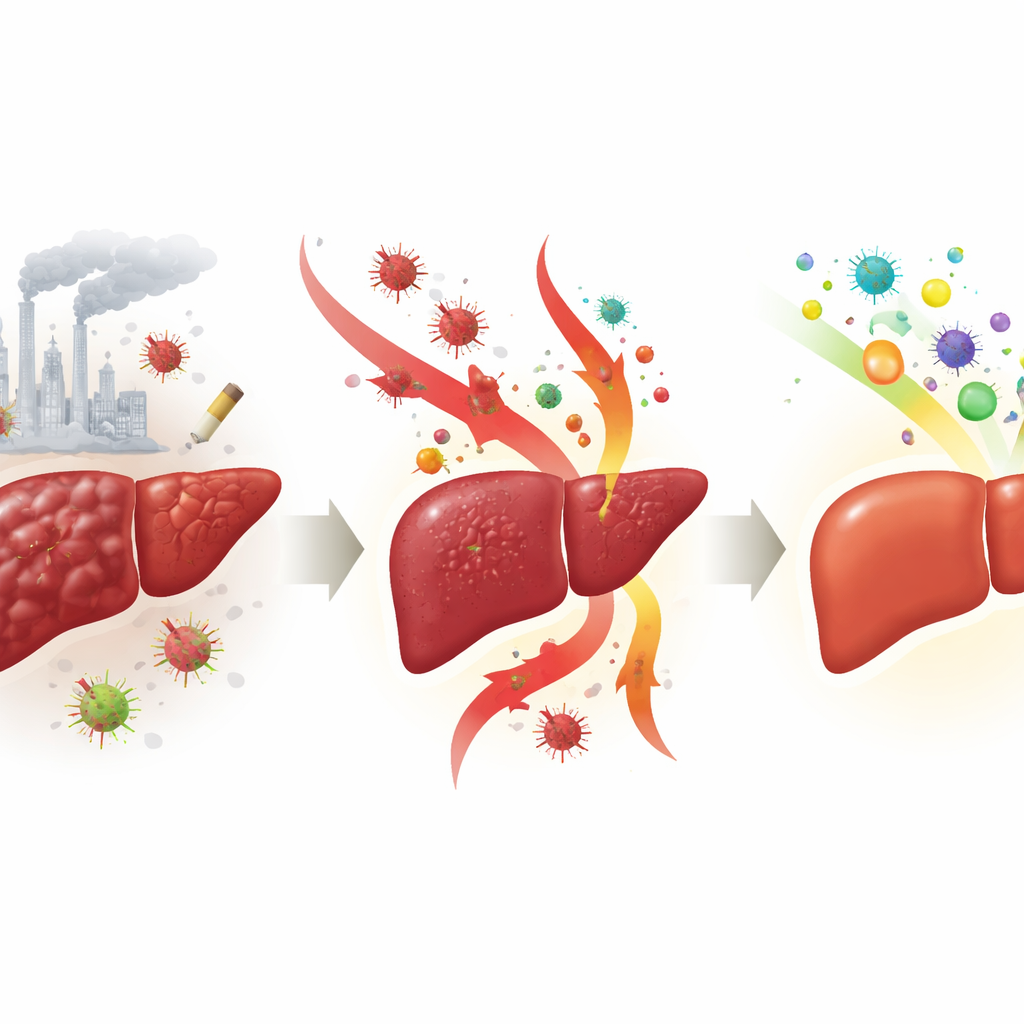
How liver scarring takes hold
Liver fibrosis develops when repeated injury outpaces the organ’s ability to repair itself. In this state, specialized support cells in the liver, called stellate cells, wake up from a quiet state and turn into scar-producing cells. They start laying down large amounts of structural proteins, including forms of collagen, that gradually stiffen and distort the tissue. At the same time, high levels of reactive oxygen species—unstable oxygen-containing molecules—and inflammatory messengers damage liver cells and attract immune cells, creating a self-sustaining cycle of harm. A key control hub in this cycle is a signal chain known as the TLR4/NF-κB pathway, which senses bacterial products in the blood and switches on inflammation-related genes.
A small radical with big protective potential
Nitroxide radicals such as 3-CP are unusual in that they are stable free radicals that can neutralize other reactive molecules again and again, acting like tiny catalytic sponges for oxidative stress. They also slip easily across cell membranes. Earlier work suggested that related nitroxides can ease lung scarring and other inflammatory conditions with good safety in early trials, but their effects in liver fibrosis were not well understood. Here, the authors tested 3-CP in cultured human stellate cells stimulated with bacterial components, and in mice whose livers were damaged with carbon tetrachloride, a classic toxin that reliably produces fibrosis. They focused not only on how much scar tissue formed, but also on oxidative stress, inflammation, and activity of the TLR4/NF-κB pathway.
Protecting liver cells and reducing scar formation
In the cell experiments, 3-CP slowed the growth and movement of activated stellate cells and encouraged them to undergo programmed cell death, without harming normal liver cells at similar doses. It also cut down the levels of reactive oxygen species and reduced the production of hallmark scarring proteins, including alpha-smooth muscle actin and type I collagen. In mice, treatment with 3-CP during toxin exposure led to healthier-looking livers with more normal structure, less collagen staining, and a roughly half-sized scarred area compared with untreated animals. Blood tests showed that markers of liver injury were lower, and measurements of inflammatory messengers such as IL-1β, IL-6, TNF-α, and TGF-β in the liver dropped by about one-third to one-half toward normal values.
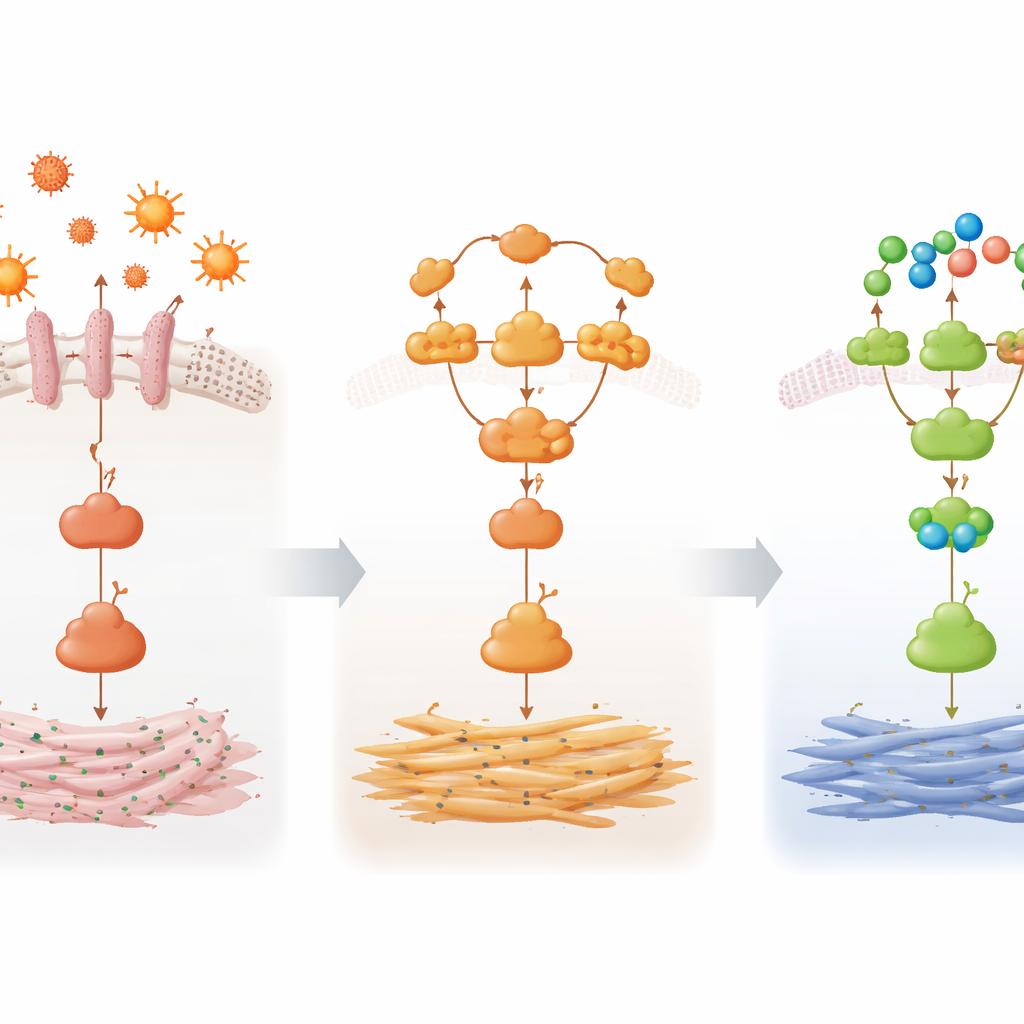
Quieting a harmful signal chain
To understand how 3-CP exerts these effects, the team examined key proteins in the TLR4/NF-κB pathway, which links bacterial products in the bloodstream to inflammatory gene activity inside liver cells. In the fibrotic mice, components of this pathway were switched on, with higher levels of receptor and adaptor proteins and more of the NF-κB subunit p65 in its activated, phosphorylated form. Treatment with 3-CP reduced the amounts of these activated forms, and microscopic imaging showed weaker signals from the activated proteins in liver tissue. Computer-based molecular docking suggested that 3-CP can fit into pockets on several of these pathway proteins, hinting that it might dampen signaling by interacting directly with them, although this will require further experimental proof.
What this could mean for future treatments
Taken together, the findings show that 3-CP lessens liver scarring in toxin-exposed mice while also protecting cultured human stellate cells from oxidative and inflammatory overdrive. It appears to do this by both scavenging reactive oxygen species and dialing down a major inflammatory switch in the liver. For a layperson, this means 3-CP acts a bit like a dual-action fire extinguisher, reducing both the sparks and the alarm signals that keep liver damage smoldering. The work does not yet prove that 3-CP will be safe or effective in people, and more studies are needed to confirm exactly how it interacts with the TLR4/NF-κB pathway and to test long-term safety. Still, the results position 3-CP as a promising lead compound in the search for medicines that can truly slow or reverse liver fibrosis.
Citation: Yao, R., Wang, R., Wang, Y. et al. 3-carbamoyl proxyl nitroxide attenuates CCl4-induced liver fibrosis in mice through antioxidant-inflammatory regulation of TLR4/NF-κB signaling pathway. Sci Rep 16, 10798 (2026). https://doi.org/10.1038/s41598-026-46137-1
Keywords: liver fibrosis, oxidative stress, inflammation, nitroxide antioxidant, NF-kappaB signaling