Clear Sky Science · en
Sustainable multifaceted HPLC approach for concurrent quantitation of an octa-mixture used in upper respiratory therapy with a five-dimensional sustainability assessment
Why this matters for everyday medicines
Cold and cough remedies seem simple from the outside, but behind every bottle and tablet is careful testing to make sure each ingredient is present in the right amount. This paper presents a new laboratory method that can check eight different substances at once in medicines used for upper respiratory problems, such as coughs, colds, and sinus infections. It also shows that this method is faster, cheaper, and kinder to the environment than many traditional approaches, making quality control more reliable and sustainable.
Many drugs in one common illness
Upper respiratory tract disorders, ranging from the common cold to more serious infections, affect millions of people worldwide and lead to huge numbers of missed work and school days. To ease symptoms like fever, cough, congestion, and sore throat, doctors and pharmacists rely on a mix of medicines: pain and fever reducers, cough suppressants, mucus thinners, allergy pills, antibiotics, and preservatives that keep liquid formulations stable. In real products, several of these are often combined in one syrup or capsule, especially for children. That convenience creates a testing challenge: pharmacies and manufacturers must be sure that every active ingredient, and even the preservatives, are present in safe and effective amounts.
A single test for an eight-drug cocktail
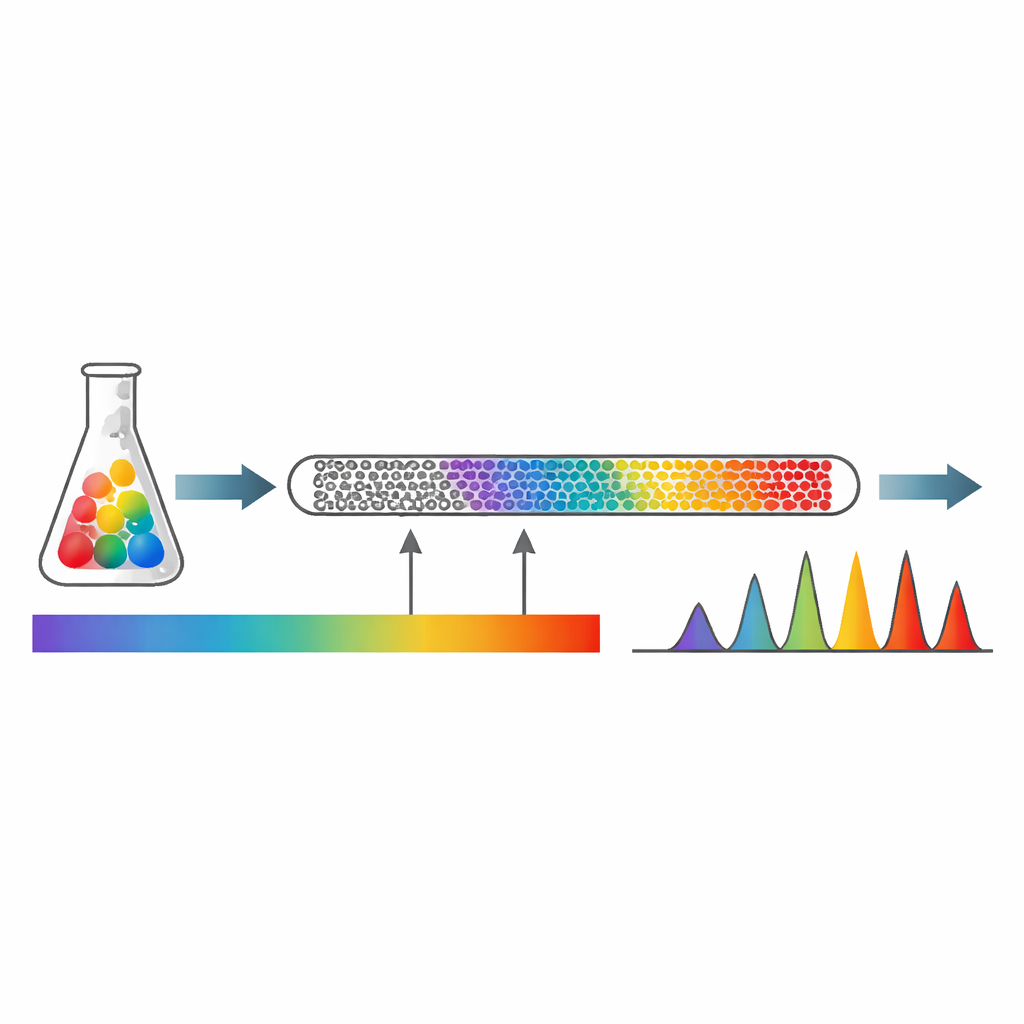
This study introduces a high‑performance liquid chromatography method with diode‑array detection—a sophisticated form of chemical “sieving” using pressurized liquid and light—to measure eight compounds at once. The team focused on drugs frequently used together in upper respiratory treatments: the bronchodilator albuterol, the mucus thinner erdosteine, the pain and fever reliever paracetamol, the antibiotic amoxicillin, the allergy medicine chlorpheniramine, the expectorant guaifenesin, and two common preservatives, methyl and propyl parabens. Using a specially chosen C18 separation column and a carefully designed mixture of water-based buffer and methanol that changes over time, they were able to separate all eight components clearly in less than 18 minutes from a single injection.
Making the method accurate and trustworthy
Designing the test was only the first step; proving that it works under real‑world conditions was just as important. The researchers adjusted many variables—type of column, solvent mixture, acidity, and flow rate—until they obtained sharp, symmetrical peaks for each compound with no overlap. They then validated the method according to international guidelines, checking that the signal increased in a straight‑line fashion with concentration over practical ranges, that very small amounts could be detected, and that repeated measurements on the same day and on different days gave nearly identical results. The differences between expected and measured values stayed below two percent, and the statistical indicators showed excellent precision and consistency.
From pure powders to real syrups and tablets
To show that the method works beyond ideal conditions, the team applied it to commercial products such as cough syrups, antibiotic capsules, and allergy tablets bought from local pharmacies. They used simple sample preparation steps: dissolving or diluting the medicine in methanol, sonicating when needed, and injecting the solution into the instrument. Each drug and preservative appeared at its characteristic time with no interference from sweeteners, colorants, or other inactive ingredients. By comparing the signals to standard solutions and by adding known extra amounts of each drug to the samples, they confirmed that the test recovers essentially 100% of each ingredient, demonstrating its suitability for routine quality control.
Measuring safety with a greener footprint
Beyond performance, the authors asked how “clean” and practical their method is. They used several modern assessment tools that score analytical methods on environmental impact, usability, and innovation. Because the new procedure uses modest volumes of relatively benign solvents (favoring methanol over more hazardous options), short run times, common equipment, and straightforward sample handling, it earned high scores on greenness (eco‑scale and AGREE), practicality (Blue Applicability Grade Index and Click Analytical Chemistry Index), and overall “whiteness,” which balances analytical quality, environmental impact, and day‑to‑day usability. A separate innovation index also flagged the method as a fresh, multifaceted approach within this field.
What this means for patients and the planet
In essence, the authors have created a single, reliable laboratory test that can check eight key components of cold and cough medications in one go, across a variety of real products. That makes it easier for manufacturers and regulators to ensure that combination therapies, especially those used in children, contain neither too little nor too much of each ingredient. At the same time, the method reduces solvent use, analysis time, and costs, supporting both public health and environmental goals. As similar multi‑purpose tests spread through quality control labs, patients can have greater confidence that the medicines they rely on for everyday respiratory illnesses are both safe and responsibly produced.
Citation: Khalil, H.A., Hafez, M.G., Elgammal, F.A.H. et al. Sustainable multifaceted HPLC approach for concurrent quantitation of an octa-mixture used in upper respiratory therapy with a five-dimensional sustainability assessment. Sci Rep 16, 12240 (2026). https://doi.org/10.1038/s41598-026-45971-7
Keywords: upper respiratory tract drugs, HPLC multi-analyte analysis, green analytical chemistry, pharmaceutical quality control, paracetamol and antibiotics