Clear Sky Science · en
Endometrial regeneration with mesenchymal stem cells and exosomes: an experimental rat model of intrauterine adhesions
Why damaged womb tissue matters
When the inner lining of the womb heals with scar tissue instead of healthy tissue, it can narrow or block the cavity, disturb menstrual cycles, and make pregnancy difficult. This condition, called intrauterine adhesions or Asherman syndrome, is often treated by surgically cutting the scar bands, but many women still struggle with poor fertility afterward. This study in rats explores whether living stem cells and tiny packages they release, called exosomes, could guide the lining to heal in a more natural way and reduce long‑lasting scarring.
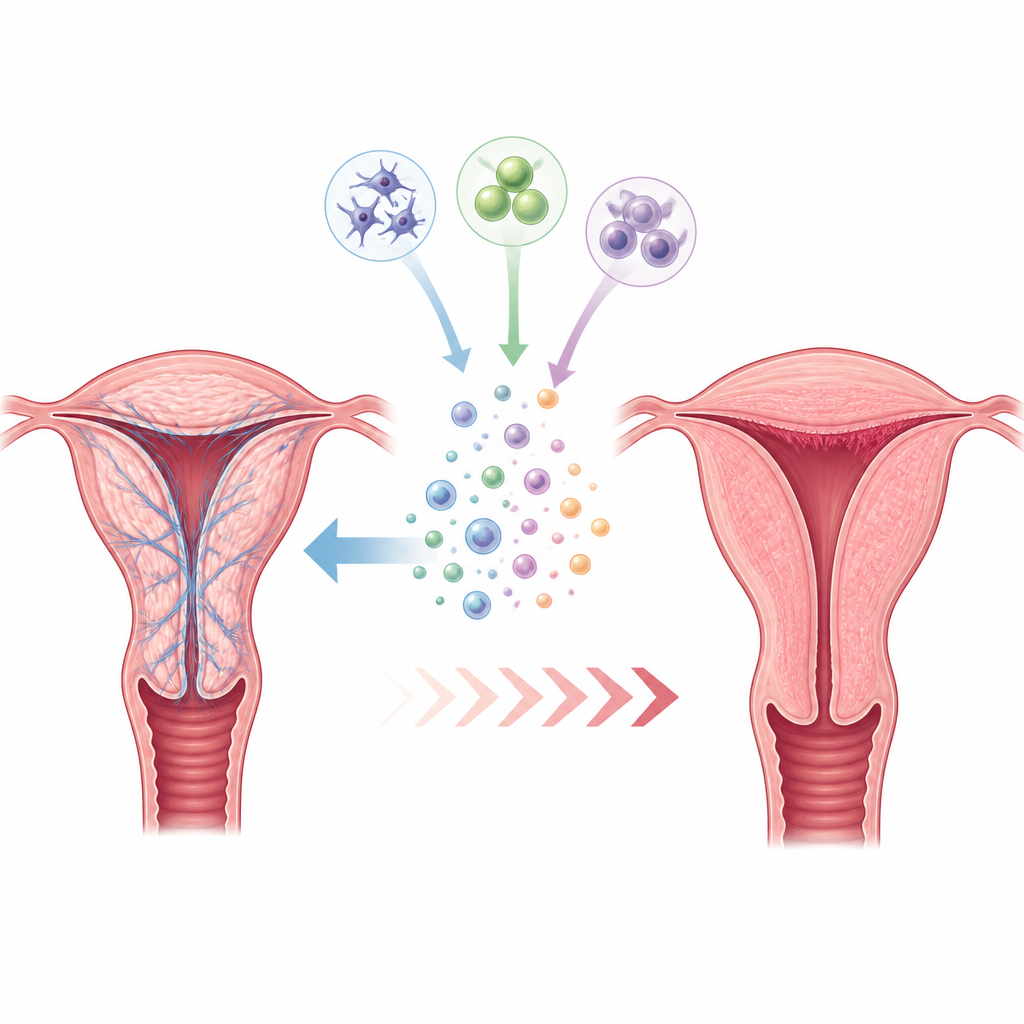
A closer look at scarring inside the womb
Intrauterine adhesions usually arise after infection or surgical trauma that harms the deep layer of the womb lining. Instead of regrowing smoothly, the walls stick together through fibrous bands that can partly or completely close the cavity. Even when these bands are cut during surgery and the space is reopened, many patients face recurring scars and ongoing menstrual and fertility problems. Current aftercare, such as estrogen treatment or devices that temporarily hold the walls apart, helps physically but does not fully restart the biology of true tissue repair.
Why stem cells and their messages are of interest
Mesenchymal stem cells are adult cells that can calm inflammation and support repair in many tissues. Growing evidence suggests that their main benefit comes less from turning into new cells and more from the chemical signals they send out. Among these signals are exosomes, microscopic bubbles loaded with proteins and genetic messages that can dial down inflammation, reduce excess collagen, encourage new blood vessels, and guide healthy rebuilding. Because exosomes are cell‑free and easier to standardize, they may offer a safer, more practical way to harness stem cell benefits.
How the rat study was designed
Researchers created womb scarring in female rats by briefly bathing one uterine horn with high‑strength alcohol, then waiting two weeks for adhesions to form. The animals were split into seven groups: untouched controls, scarred but untreated controls, and five treatment groups. Treatments used either stem cells or exosomes from two sources, umbilical cord tissue and fat tissue, placed directly into the uterine cavity. A separate group received umbilical‑cord exosomes through a vein to mimic a whole‑body treatment. Two weeks later, the team examined tissue slices under the microscope, measured the thickness of the lining and wall, and scored inflammation, new blood vessels, and fibrosis on simple scales.
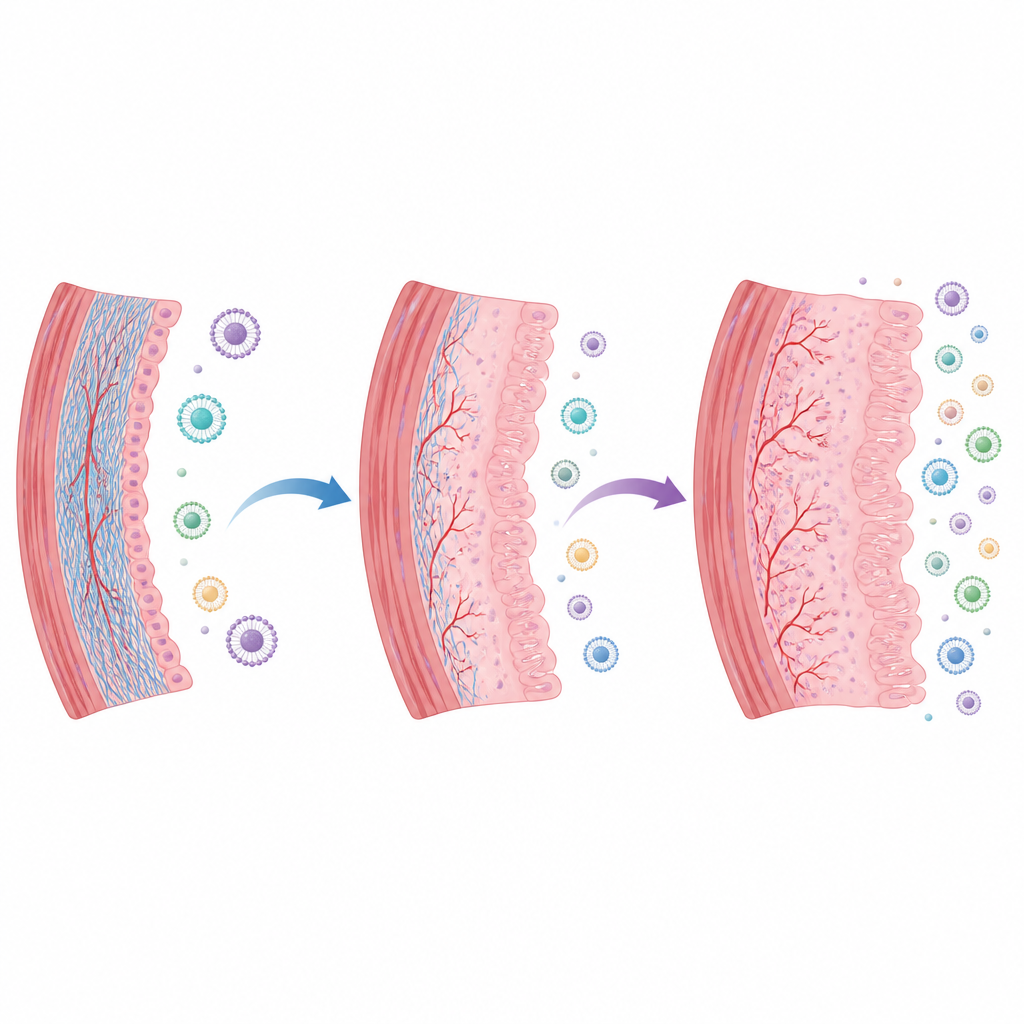
What the treatments changed in the tissue
Untreated scarred horns showed severe narrowing, a thin and patchy surface layer, fewer glands, and heavy collagen deposits. All treated groups looked better: the uterine wall was thicker, the cavity more open, and the lining more column‑shaped with more glands. Special stains showed less collagen in treated horns than in scarred controls, though the exact degree varied. When scores of inflammation and scarring were compared, exosome‑treated groups generally did better than those given whole stem cells. Exosomes from fat tissue showed the lowest levels of both inflammation and fibrosis, while umbilical‑cord exosomes, whether placed locally or given through a vein, also improved the scarring pattern to a similar extent.
What this could mean for future care
To a lay reader, the main message is that tiny repair signals taken from stem cells helped scarred womb tissue in rats look and behave more like healthy tissue, especially by calming inflammation and limiting hard scar buildup. Exosome‑based treatments, particularly those from fat tissue, gave the most favorable healing pattern in this early work, and giving umbilical‑cord exosomes through a vein worked about as well as putting them directly into the womb. The study is still preclinical, involved few animals, and did not test fertility, so it cannot yet guide medical care. But it suggests that future therapies based on stem cell‑derived exosomes may one day complement surgery to restore a more functional womb lining in women with intrauterine adhesions.
Citation: Atay, F., Atay, A.O., Akdemir, A. et al. Endometrial regeneration with mesenchymal stem cells and exosomes: an experimental rat model of intrauterine adhesions. Sci Rep 16, 15016 (2026). https://doi.org/10.1038/s41598-026-45939-7
Keywords: intrauterine adhesions, endometrial repair, mesenchymal stem cells, exosomes, fibrosis