Clear Sky Science · en
Optical diagnostics of low-pressure RF-DBD Ar/CH₄ plasma: mapping electron temperature and density versus power, pressure, and gas flow
Why tiny sparks in gas could matter for clean energy
Hydrogen is widely discussed as a clean fuel of the future, but making it without adding more carbon dioxide to the air is a major challenge. This study looks at a special kind of glowing gas, or plasma, that can break methane apart into hydrogen under gentle conditions. By carefully mapping how this plasma behaves, the researchers aim to give engineers a clearer recipe for tuning reactors that turn methane into useful products while limiting unwanted carbon buildup.
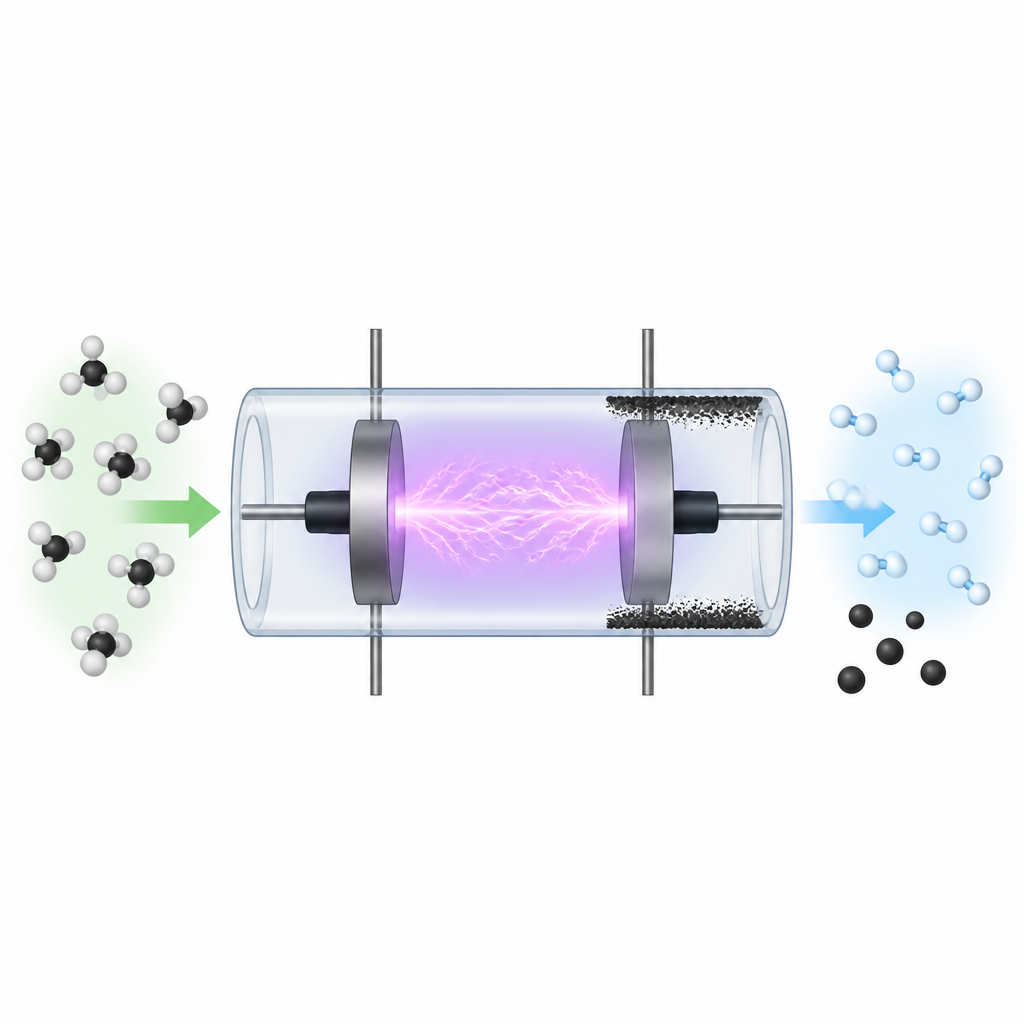
A glow in a glass tube
The team worked with a simple, well controlled setup: a glass tube pumped down to low pressure, filled with argon gas alone or with a mix of argon and methane. Around this tube they placed metal parts that feed in radio waves, which cause the gas inside to light up in a soft purple glow. This glowing state is plasma, where many of the gas atoms and molecules are electrically charged. The researchers then changed three main knobs on the system – how hard they powered the discharge, how much gas flowed through, and how much pressure was in the tube – and watched how the glow and the products responded.
Reading the light from the plasma
Rather than poking the plasma with a metal probe, which can disturb it, the team relied on the light the plasma gives off. Each type of atom or molecule shines at very specific colors, like a fingerprint. By measuring the brightness and exact colors of this light with an optical spectrometer, they could back out two key internal properties: how hot the electrons are and how many of them are present in a given volume. They also paid special attention to the red hydrogen line called Hα, which signals the presence of hydrogen atoms formed when methane breaks apart. These measurements let them build maps showing how electron temperature and electron density change with power, pressure, and gas flow.
How tuning the knobs changes the glow
In pure argon, raising the gas pressure from 0.5 to 1.0 Torr at modest power made the electrons slightly hotter but reduced how many there were. Turning up the power had the opposite effect: electrons cooled a bit on average while their number rose, reflecting more frequent collisions that create new charged particles. When methane was added, the picture shifted. The electrons became hotter overall, reaching about one and a quarter times the energy seen in argon alone, while their numbers tended to drop. That is because a share of the electrons are lost during the growth of carbon rich material, so the remaining electrons must carry more energy to keep the discharge going. Changing the mix of argon and methane further reshaped this balance by altering how long reactive fragments stayed in the plasma.
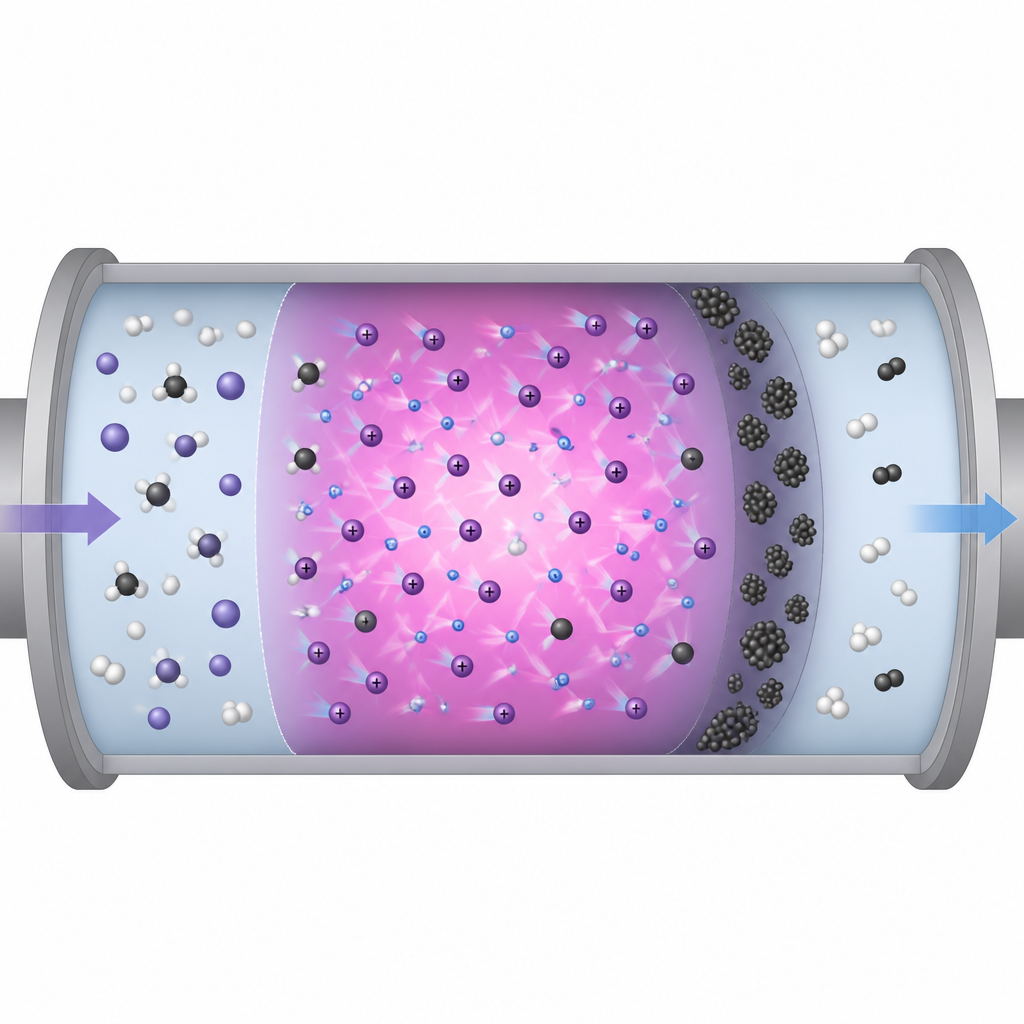
Watching carbon build up on walls
The same plasma that frees hydrogen from methane also creates carbon based fragments that can stick to nearby surfaces. To see what kind of material formed, the scientists examined the metal electrode and the inner wall of the glass tube after the experiments. Using an electron microscope, they found thin, cracked films coating the electrode and small clumps of particles on the glass, all a few micrometers across. Raman spectroscopy, which reads the way light scatters from bonds in a solid, showed two broad peaks typical of amorphous carbon. This means the deposits lack the neat, ordered structure of graphite and instead contain many defects and mixed bonding.
What this means for future hydrogen reactors
By tying together the inner state of the plasma, the strength of the hydrogen light signal, and the nature of the carbon deposits, the study provides a practical guide for those hoping to design plasma based hydrogen sources. It shows that small changes in pressure, power, and gas mix can nudge the plasma toward either more hydrogen release or more carbon buildup on the walls. Clear maps of electron temperature and density under different settings give a starting point for choosing operating conditions that favor efficient methane conversion while managing unwanted deposits, an important step toward reliable plasma reactors for cleaner energy applications.
Citation: Yelubayev, D.Y., Ongaibergenov, Z.Y., Utegenov, A.U. et al. Optical diagnostics of low-pressure RF-DBD Ar/CH₄ plasma: mapping electron temperature and density versus power, pressure, and gas flow. Sci Rep 16, 15129 (2026). https://doi.org/10.1038/s41598-026-45929-9
Keywords: plasma hydrogen, methane conversion, dielectric barrier discharge, electron temperature, amorphous carbon