Clear Sky Science · en
Effects of multistep adsorption isotherms on solute transport
Why slowing pollutants in soil matters
When chemicals like pesticides or industrial compounds seep into soil and groundwater, they rarely move as a simple smooth wave. Instead, they may travel in uneven surges, arriving at wells or rivers in a series of unexpected pulses. This study explores how a particular kind of sticking behavior on soil grains can split a single contaminant plume into several distinct fronts, helping scientists better predict when and where pollution will show up.
How chemicals stick to soil
Many dissolved substances do not stay entirely in the water that flows through pores in soil and rock. Some portion latches onto grain surfaces, a process called adsorption. Classic models assume a single smooth relationship between how much is in the water and how much sticks to the soil. In those simple cases, the contaminant plume is only slowed down as a whole. Its shape does not change much as it moves with the flowing groundwater.
When sticking happens in several stages
Real soils and real chemicals are often more complicated. Some compounds begin to use new types of attachment sites only after their concentration passes certain thresholds. This creates a multistep pattern of adsorption: one set of sites is active at low levels, more sites turn on at moderate levels, and still others at higher levels. In mathematical terms, the curve describing how much sticks versus how much remains dissolved bends back and forth, so its slope does not simply increase or decrease. The study focuses on this multistep behavior and asks how it reshapes the movement of pollutants through porous ground.
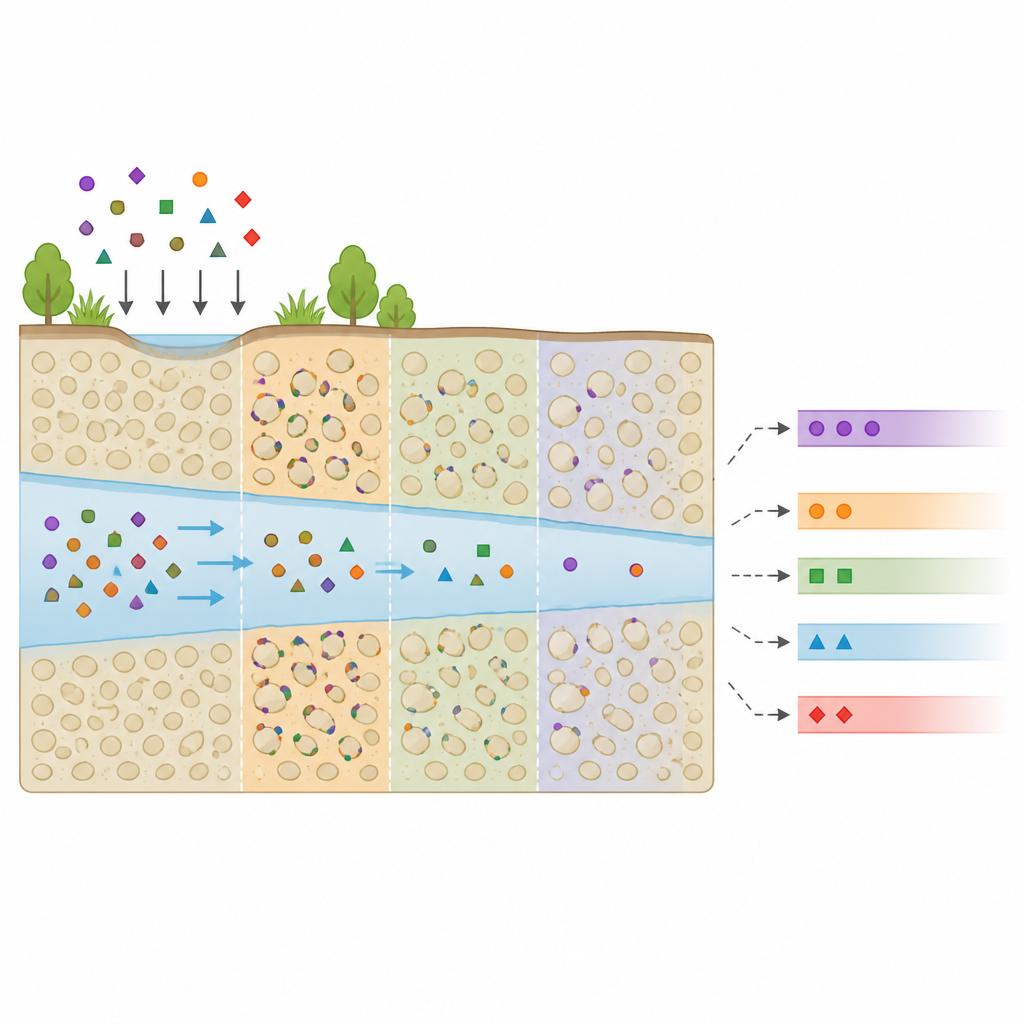
Watching a plume break into steps
The researcher first used a modified groundwater transport program to simulate chemical movement in a one dimensional flow of water through a uniform soil column. Dispersion and diffusion were turned off to isolate the effect of sticking alone. When a constant concentration source was applied at the inlet, the initially sharp front did not remain single. Instead, it split into a series of flat plateaus separated by sharp jumps, like a staircase laid along the flow direction. Each plateau matched one of the threshold concentrations where a new adsorption step became active, and each jump traveled at its own constant speed. Lower concentration fronts moved faster, while higher concentration fronts lagged behind.
Testing which soil and chemical traits matter
To understand what controls this stepwise pattern, the study varied each parameter of a three step model one at a time. Threshold concentrations set the heights of the plateaus: shifting a threshold simply moved the level of a step. Other parameters, which describe how strongly and how much a given step can adsorb, mainly controlled how fast each front traveled. Stronger or higher capacity sites slowed the associated concentration front more. In some combinations, two neighboring steps moved at the same speed and merged into a single front, while in others all three remained clearly separated. This showed that local changes in the adsorption curve mostly affect the neighboring part of the plume.
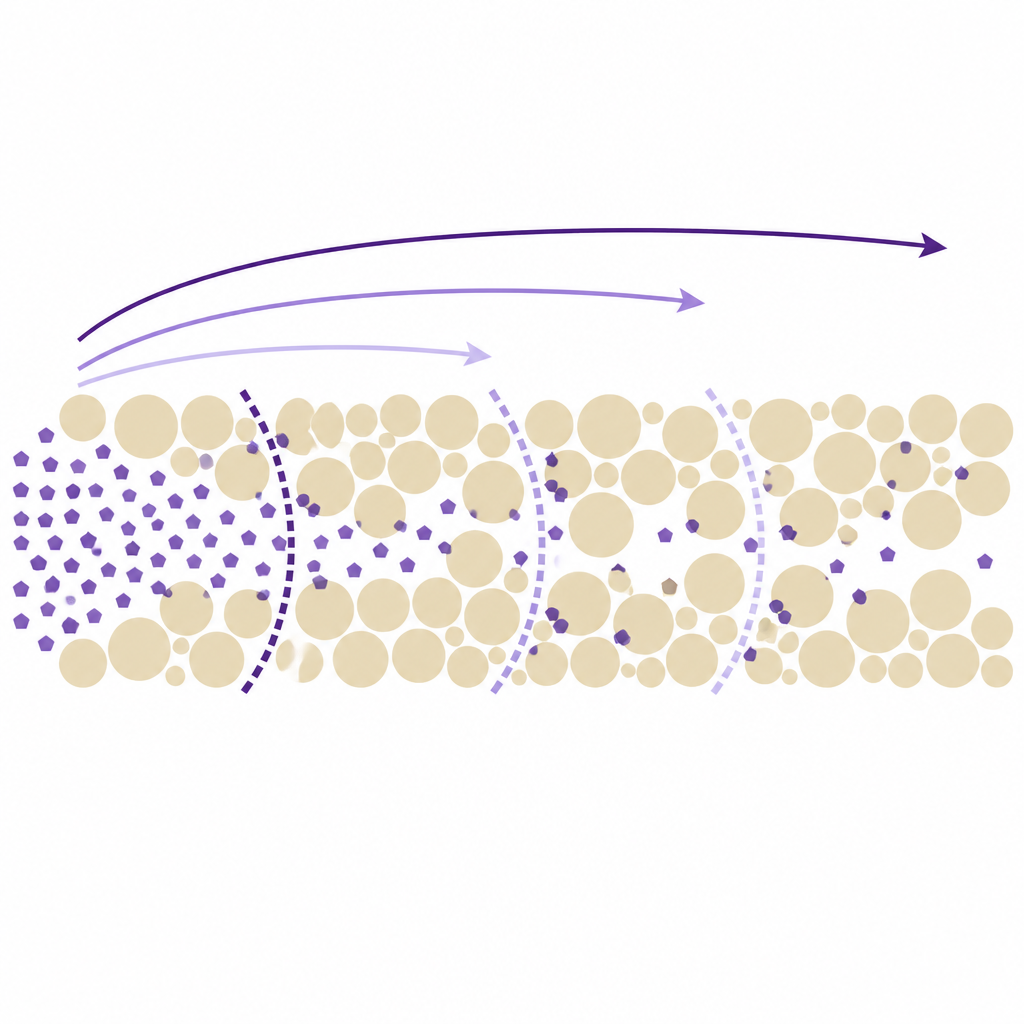
Linking plume speed to the shape of the curve
The numerical work was complemented by an analytic treatment, building on earlier theories of nonlinear adsorption. In this framework, the speed of any concentration front is tied to the average slope of the adsorption curve over the range of concentrations that front spans. A steeper average slope means stronger overall sticking and thus a slower front. Applying this idea to the multistep curve showed that distinct steps will form only if those average slopes increase from one step to the next, so that low concentration fronts always outrun higher concentration ones. When this condition is not satisfied, the steps coalesce. Across dozens of simulated cases, the simple analytic formulas matched the numerical results very closely.
What this means for pollution and cleanup
In plain terms, the study shows that certain combinations of soil properties and chemical behavior can turn a single contaminant cloud into a train of separate waves that arrive at different times. The heights of these waves depend on the concentration thresholds where new sticking mechanisms switch on, while their speeds are set by how strongly each stage of sticking holds the chemical back. Recognizing when a multistep pattern applies can improve models used for groundwater protection and cleanup planning, although it requires more detailed measurements. The work also offers a clear recipe for predicting whether multiple fronts will stay distinct or blend together, helping scientists anticipate complex pollution behavior in the subsurface.
Citation: Fekete, E. Effects of multistep adsorption isotherms on solute transport. Sci Rep 16, 14957 (2026). https://doi.org/10.1038/s41598-026-45699-4
Keywords: solute transport, adsorption isotherm, groundwater, porous media, pesticide migration