Clear Sky Science · en
Hot corrosion and cyclic oxidation behavior of CoMoCrSi + Cr₃C₂ composite coatings on MDN 420 steel by HVOF spray process
Protecting Metals in Harsh Heat
Modern engines, power plants, and industrial boilers run so hot and in such corrosive conditions that their metal parts can literally burn away over time. Replacing these components is costly and risky. This study explores a special protective coating, sprayed onto a common high-strength steel, that aims to keep those parts from rusting, cracking, and crumbling when exposed to intense heat and aggressive salt deposits.
Why Hot Metal Falls Apart
In many turbines, boilers, and chemical plants, metal parts see both high temperatures and corrosive chemicals. Oxygen in the air slowly forms oxide layers, while molten salts—produced, for example, from fuel impurities containing sodium and vanadium—can attack these oxides and open fast pathways for further damage. Bare steel exposed to such conditions gains weight as thick, flaky oxide scales grow, then loses material as these scales break off. Over time, this cycle of growth and spalling leads to thinning, cracking, and failure of critical components.
A Tough New Jacket for Steel
The researchers focused on MDN 420 steel, a martensitic stainless steel used in demanding applications, and covered it with a composite coating made from cobalt, molybdenum, chromium, and silicon, reinforced with hard chromium-carbide particles. Using a high-velocity oxy-fuel (HVOF) spray gun, they fired this powder at supersonic speeds onto steel plates, building up a dense layer about two-tenths of a millimeter thick. Careful control of the spray process produced a coating with low porosity and a surface roughness suitable for resisting wear. Microscopic examination showed a compact structure with well-distributed hard particles and only small cracks and pores, while hardness tests revealed that the coated surface was more than three times harder than the underlying steel.
How the Coating Fights Heat and Salt
To see how well the coating worked, both coated and uncoated steel samples were repeatedly heated to 700 °C and cooled, either in air (to test oxidation) or in a mixture of molten sodium sulfate and vanadium oxide (to test hot corrosion). In air, the coating promoted the growth of thin, continuous layers of chromium oxide and silica at its surface. These layers acted like a tight skin that slowed the inward diffusion of oxygen and sealed micro-gaps between spray splats. As a result, coated samples gained far less weight than bare steel, meaning much less oxide buildup, and their rate of oxidation remained low and steady over 50 cycles.
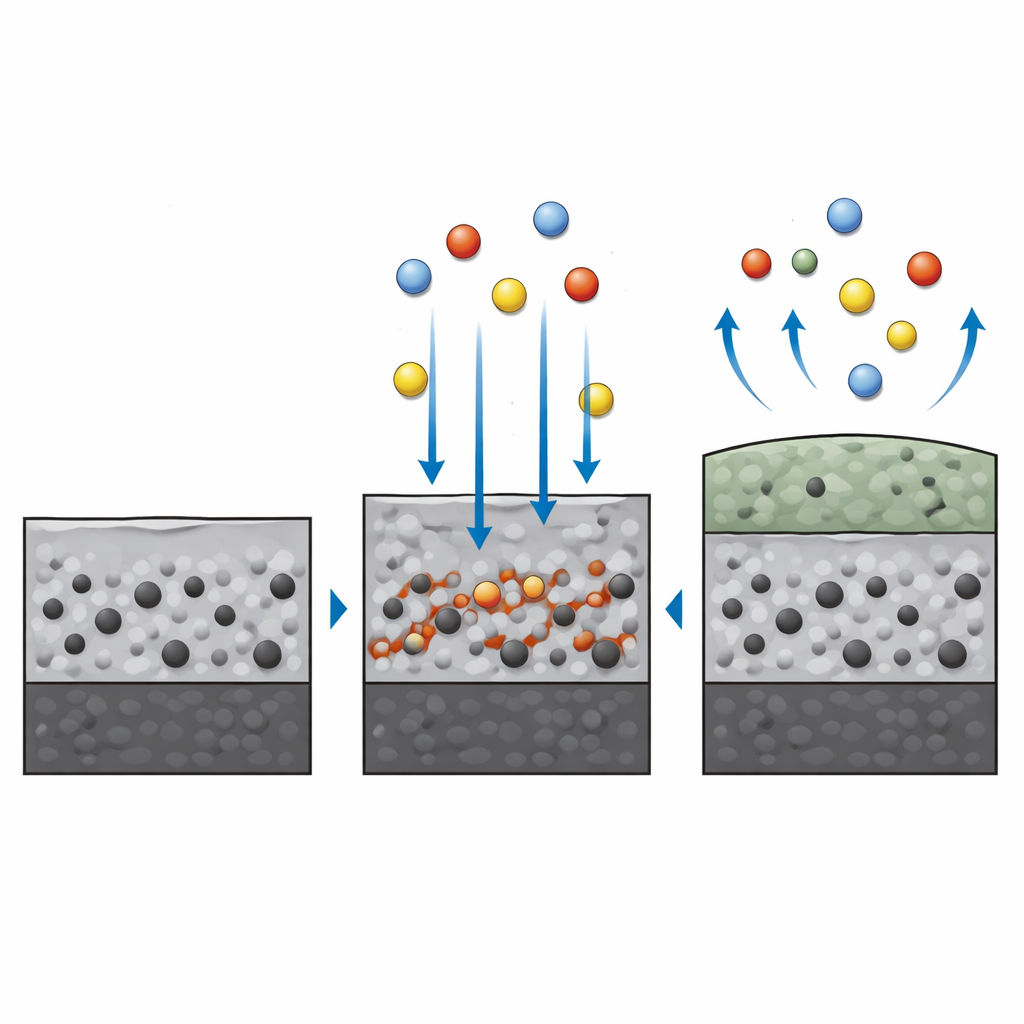
What Happens in a Salt Bath
The molten-salt tests were far more aggressive, mimicking conditions in fuel-fired boilers and turbines where sodium and vanadium compounds can melt and wet hot metal surfaces. Here, the coating still outperformed bare steel: weight gains and corrosion rates were dramatically lower. However, detailed analyses showed that the salt did not simply sit on the surface. It reacted with oxides of molybdenum and chromium within the coating to form complex salt-oxide compounds. These new phases, such as sodium molybdate and sodium vanadate, created porous, easily broken scales. Cracks and pits formed as volatile molybdenum oxides evaporated and the protective skin locally detached, exposing fresh material to further attack.
Balancing Strength and Long Life
Overall, the coating substantially reduced both oxidation and hot corrosion rates compared with bare MDN 420 steel—its measured rate constants were several times lower. The combination of a tough cobalt-based matrix, hard carbide particles, and self-forming protective oxides allows the coated steel to withstand high temperatures and corrosive salts for longer periods. Still, the study also reveals the limits of this protection: where salt-induced reactions and volatile oxides create pores and weak scales, damage gradually accumulates. For a layperson, the takeaway is that smartly designed coatings can act like fireproof, chemical-resistant armor for hot metal parts, buying much more operating time before failure—but that this armor must be further refined, for example by sealing pores or adjusting composition, to stand up to the harshest salt-laden environments.
Citation: S, S., Prasad, C.D., Kumaraswamy, G.N. et al. Hot corrosion and cyclic oxidation behavior of CoMoCrSi + Cr₃C₂ composite coatings on MDN 420 steel by HVOF spray process. Sci Rep 16, 10677 (2026). https://doi.org/10.1038/s41598-026-45658-z
Keywords: high temperature coatings, hot corrosion, oxidation resistance, thermal spray, stainless steel protection