Clear Sky Science · en
Magnetic amino-functionalized hyper-crosslinked resin as a reusable adsorbent for methylene blue
Why cleaning dyed water matters
Bright blue dyes that color our clothes and products can quietly pollute rivers and lakes when factory wastewater is not fully treated. One common dye, methylene blue, is tough to break down and can harm living organisms even at low levels. This study explores a new material that can grab this dye out of water, be pulled out with a magnet, and then be used again, offering a simple and reusable way to help keep water cleaner.
A sponge built from tiny rigid cages
The researchers started from a special plastic called a hyper crosslinked resin. Unlike everyday plastics, this material is full of permanent tiny pores, like a rigid sponge made of countless cages and tunnels. These microscopic spaces create a huge internal surface where dye molecules can stick. The team chemically added amino groups to the resin to make its surface more welcoming to water and more attractive to charged dye molecules. This step turned an originally water hating plastic into one that can interact strongly with methylene blue.
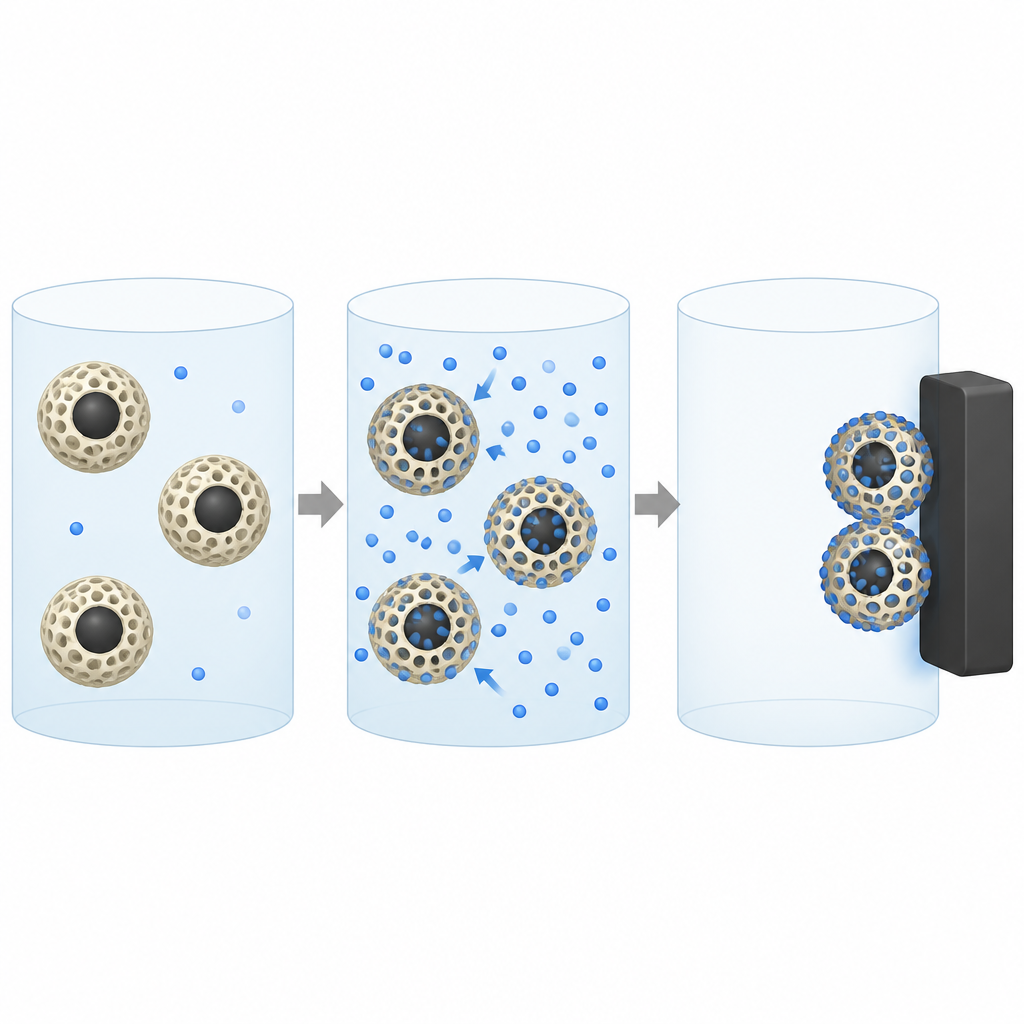
Adding magnetism for easy cleanup
To make the material easy to collect after it does its job, the scientists grew iron oxide nanoparticles, known as magnetite, directly inside the porous resin. The resulting composite, called Fe3O4@XDV NH2, behaves like a powder made of tiny porous beads with magnetic cores. Detailed imaging and structural tests showed that the iron oxide particles are small, well distributed, and do not destroy most of the pore system. Magnetic measurements confirmed that the beads respond strongly to a magnetic field, so they can be rapidly gathered from water with a simple magnet instead of slow filtration or centrifugation.
How well the magnetic sponge captures dye
The team then tested how effectively the magnetic resin removes methylene blue from water under different conditions. They found that the process works best in mildly alkaline water, around pH 9, where the surface of the material becomes negatively charged and strongly attracts the positively charged dye. Under these conditions and at room temperature, the material showed high dye uptake, both in direct measurements and in models that describe how a thin layer of dye forms on the surface. The data fit a model in which a single coating of dye covers similar sites on the material, suggesting an orderly, one layer adsorption process.
Fast action, reusable performance
Time based tests showed that most of the dye removal happens within the first hour, with the rate controlled by how quickly dye molecules reach and occupy active sites in the pores. Further analysis indicated that the process is spontaneous and releases heat, so higher temperatures actually reduce performance. Importantly, the material still removed large fractions of dye in more realistic waters, including tap water and a laboratory made wastewater containing salts and other organic compounds. Even in these tougher mixtures, removal remained high, although competing substances slightly reduced efficiency compared with pure water.
Using and reusing the magnetic beads
For any practical water treatment, the cleaning material must be used repeatedly. The researchers tested different liquids to strip the dye from the loaded beads and found that an acidic solution works best. It changes the surface charge, pushing the dye molecules off the resin so the beads can be used again. After six cycles of dye capture and acid washing, the material still retained about 89 percent of its original capacity, showing good durability. When compared with other magnetic adsorbents reported in the literature, this magnetic resin offers a strong balance of capacity, ease of separation, and reusability.
What this means for cleaner water
In simple terms, the study presents a tiny magnetic sponge that can soak up a common blue dye from water, be quickly pulled out with a magnet, cleaned, and put back to work several times. While designed and tested in the lab, the material combines strong dye capture, stability, and easy handling in a way that could help future wastewater treatments deal more efficiently with colored pollutants from industry.
Citation: Cirillo, C., Iuliano, M., Modestino, M. et al. Magnetic amino-functionalized hyper-crosslinked resin as a reusable adsorbent for methylene blue. Sci Rep 16, 14933 (2026). https://doi.org/10.1038/s41598-026-45545-7
Keywords: methylene blue, wastewater treatment, magnetic adsorbent, hyper crosslinked polymer, dye removal