Clear Sky Science · en
Optimization and evaluation of gastro-expandable film of Eudragit S100 and ethylcellulose by using the design of experiment
Why a stomach‑floating film matters
Many people with digestive troubles take medicines that work best if they stay in the stomach for several hours. Standard pills often leave the stomach too quickly, which can limit how much drug the body actually absorbs. This study explores a thin, foldable film loaded with the drug itopride hydrochloride that unfolds and floats inside the stomach, staying in place long enough to release the medicine slowly and more effectively.
A foldable sheet that turns into a stomach raft
The researchers set out to build a “gastro‑expandable” system: a flat, polymer film that can be folded and packed inside a small capsule. After swallowing, the capsule shell dissolves and the film swells, unfolds, and becomes large enough that it is not easily swept out of the stomach. Two key ingredients give the film its structure and behavior: ethylcellulose, a common film‑forming material, and Eudragit S100, a polymer that helps the sheet maintain integrity in acidic stomach conditions. A third component, triethyl citrate, acts as a softener so the film bends without cracking. The goal was to combine these ingredients in just the right proportions so the film would unfold quickly, stay intact and floating for hours, and steadily release almost all of its itopride payload.
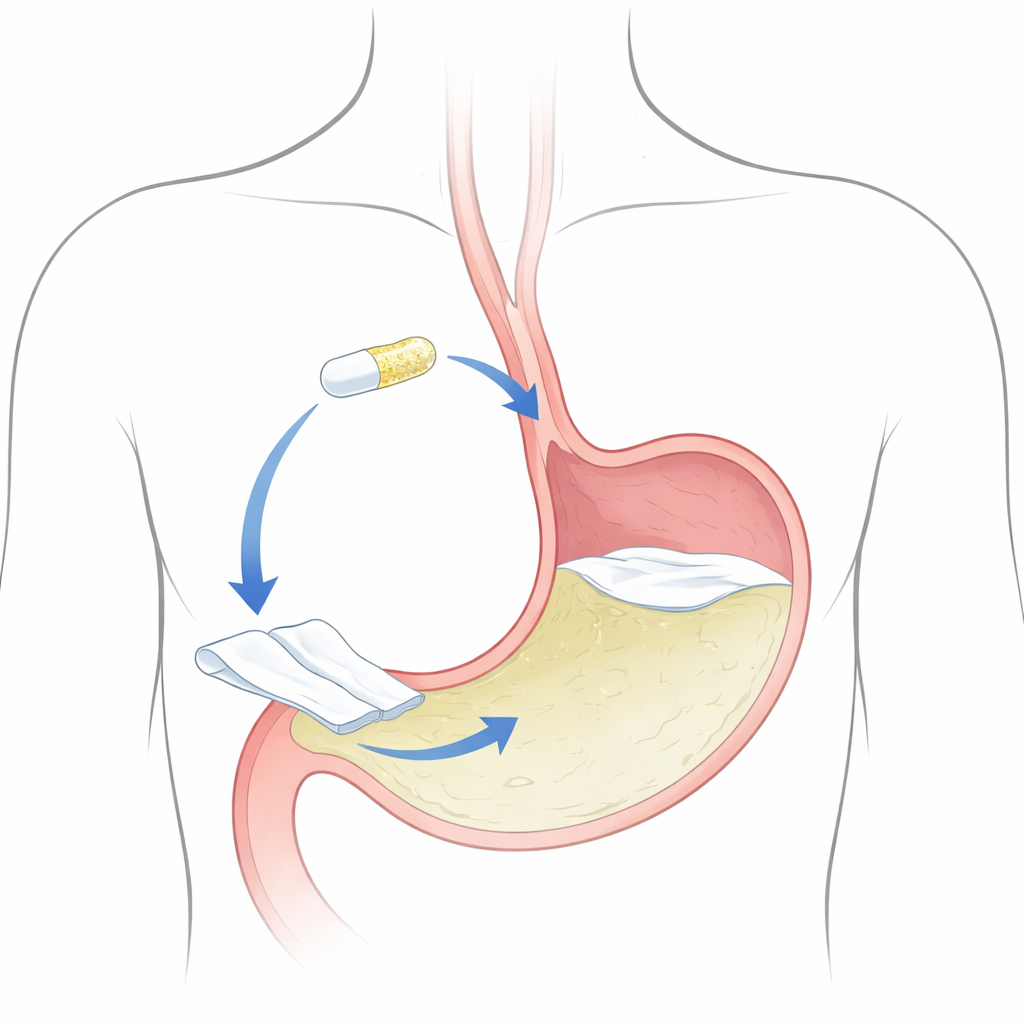
Designing the ideal floating film
Rather than relying on trial and error alone, the team used a structured statistical approach called a central composite design to plan and analyze their experiments. They varied the amounts of ethylcellulose and triethyl citrate, while keeping both the dose of itopride and the amount of Eudragit S100 constant. For each formulation, they measured how many times the film strip could be folded before breaking (a measure of toughness), how long it floated on simulated gastric fluid, and how much drug it released over time. Advanced tools such as infrared spectroscopy, thermal analysis, X‑ray diffraction, and electron microscopy confirmed that the drug remained chemically stable, became evenly dispersed in an amorphous (non‑crystalline) form within the film, and that the film surface was smooth and defect‑free.
Staying afloat and releasing drug over hours
From these experiments, the researchers identified an optimized film made with relatively high ethylcellulose and triethyl citrate levels. This version showed strong mechanical endurance, withstanding over a hundred folds without breaking, and began to float in stomach‑like fluid in under a minute. Once unfolded, it remained buoyant for more than eight hours, a time frame that covers typical stomach residence during fasting and early digestion. In laboratory release tests, the film delivered nearly all of its itopride content (about 99%) over 12 hours. Analysis of the release pattern suggested that the drug left the film through a mix of slow diffusion and gradual erosion of the sheet, supporting a smooth, sustained delivery rather than a rapid burst.
What happens in living animals
To see how this design performs in a living body, the optimized film was tested in rabbits and compared with a marketed sustained‑release capsule of itopride. X‑ray imaging using a contrast‑loaded placebo film showed that the folded sheet unfolded and stayed in the stomach for more than eight hours, matching the laboratory flotation results. In separate tests measuring drug levels in the blood, the film produced a slightly higher peak concentration and, more importantly, kept itopride in circulation for longer. Key measures such as total drug exposure over time, half‑life, and mean residence time were all improved compared with the commercial product, even though the standard in‑vitro dissolution curves of the two products were not identical.
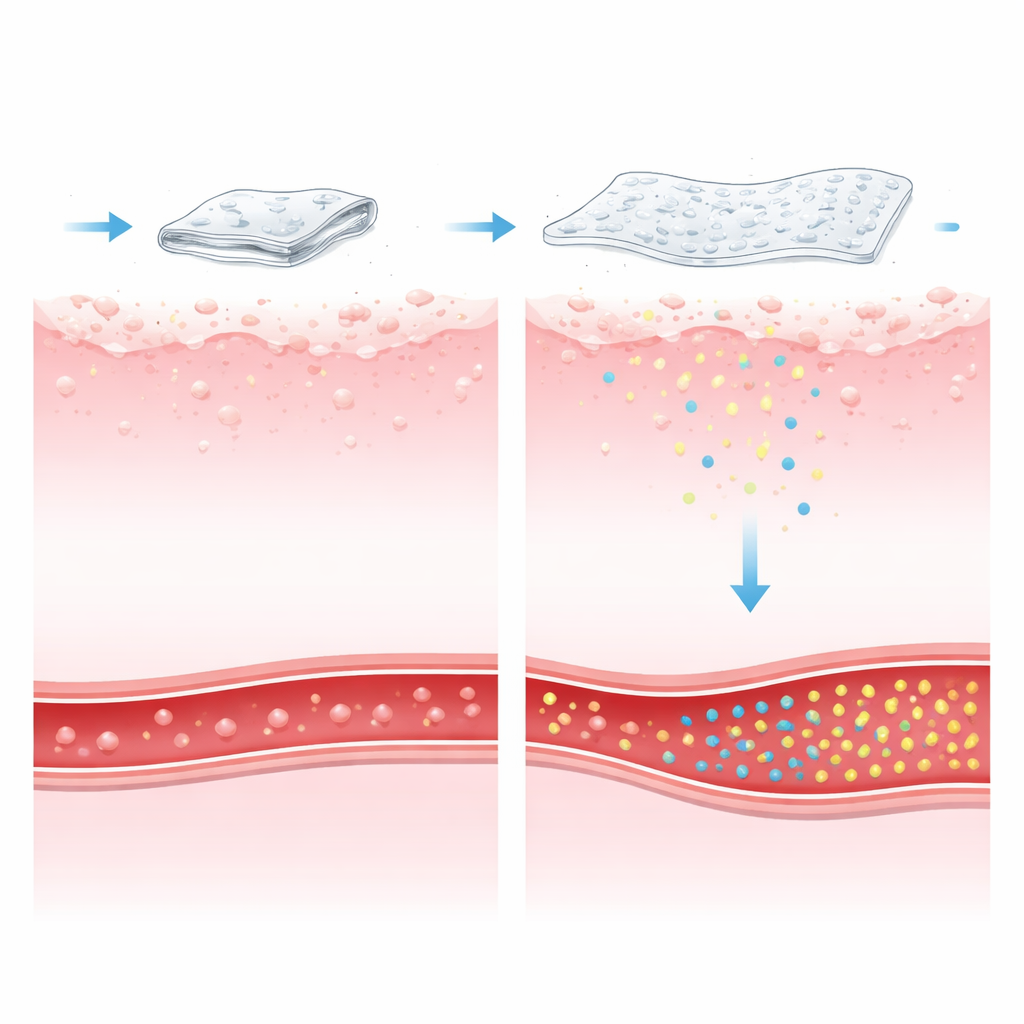
A new way to keep stomach medicines where they work
In simple terms, this work shows that a cleverly engineered, stomach‑floating film can keep a pro‑motility drug like itopride in the right place for long enough to do its job, while matching or slightly improving overall absorption compared with an existing sustained‑release capsule. The film is strong, unfolds quickly, floats for hours, and remains stable during storage. For patients who need extended control of symptoms such as reflux and slow stomach emptying, such gastro‑retentive films could offer more reliable relief and potentially fewer doses, pointing toward a practical alternative to conventional long‑acting tablets and capsules.
Citation: Dass, R., Rani, P., Verma, V. et al. Optimization and evaluation of gastro-expandable film of Eudragit S100 and ethylcellulose by using the design of experiment. Sci Rep 16, 10735 (2026). https://doi.org/10.1038/s41598-026-45540-y
Keywords: gastroretentive drug delivery, floating stomach film, itopride, sustained release, experimental design