Clear Sky Science · en
In situ assembled MIL-101(Cr) composite functionalized with chitosan and tannic acid as an efficient adsorbent for Pb2+ removal from aqueous solutions
Why cleaning lead from water matters
Lead is a hidden threat in many water supplies around the world. It can seep into rivers and groundwater from factories, mines, and old pipes, and even tiny amounts can harm the brain, heart, and kidneys—especially in children. This study explores a new material that acts like a microscopic sponge, pulling lead out of water efficiently, reliably, and in a way that can be reused many times, offering a promising option for safer drinking water and cleaner ecosystems.
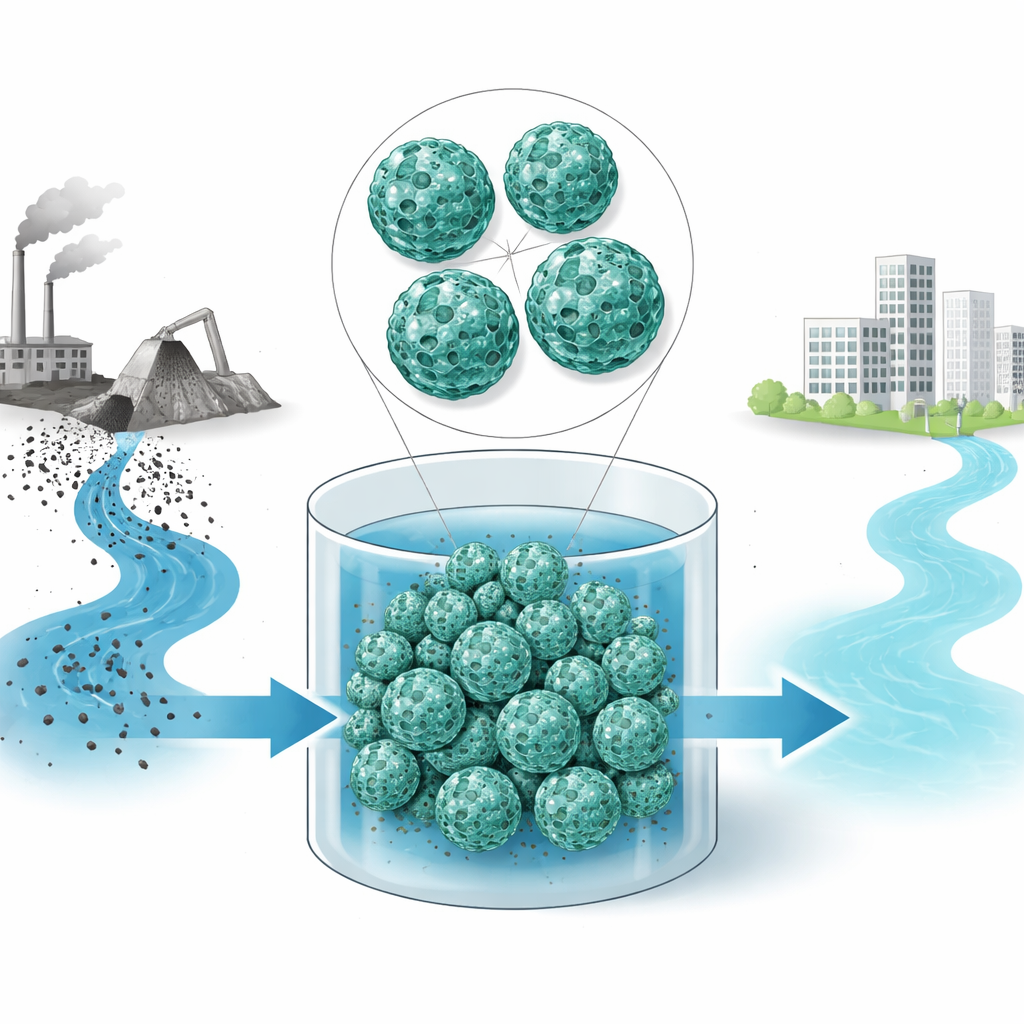
A new kind of smart cleaning sponge
The researchers built a hybrid material that combines ingredients from both chemistry labs and nature. Its backbone is a highly porous crystal called MIL-101(Cr), riddled with tiny cages and tunnels that provide a huge internal surface area. Onto this framework they assembled two plant- and shell-derived helpers: chitosan, a biopolymer obtained from crustacean shells, and tannic acid, a natural compound rich in metal-grabbing chemical groups. By letting these components form together directly in water, they created an intertwined network in which the crystal framework provides structure and space, while chitosan and tannic acid supply many sticky spots for grabbing lead.
How the material is made and what it looks like
To construct this composite, the team first grew the porous MIL-101(Cr) crystals in hot water, then prepared a smooth mixture of chitosan and tannic acid. They slowly combined the two so that the biopolymer mixture wrapped around and anchored onto the crystal particles. Microscopes revealed that the final material consists of the original well-shaped crystals embedded in a thin, rough coating of the natural polymers, without the crystals collapsing or clumping. Other tests showed that the composite keeps a very large surface area—almost nine football fields per gram—and contains both tiny and somewhat larger pores. This dual pore structure helps water and dissolved lead reach deep into the material while still providing plenty of internal nooks for capture.
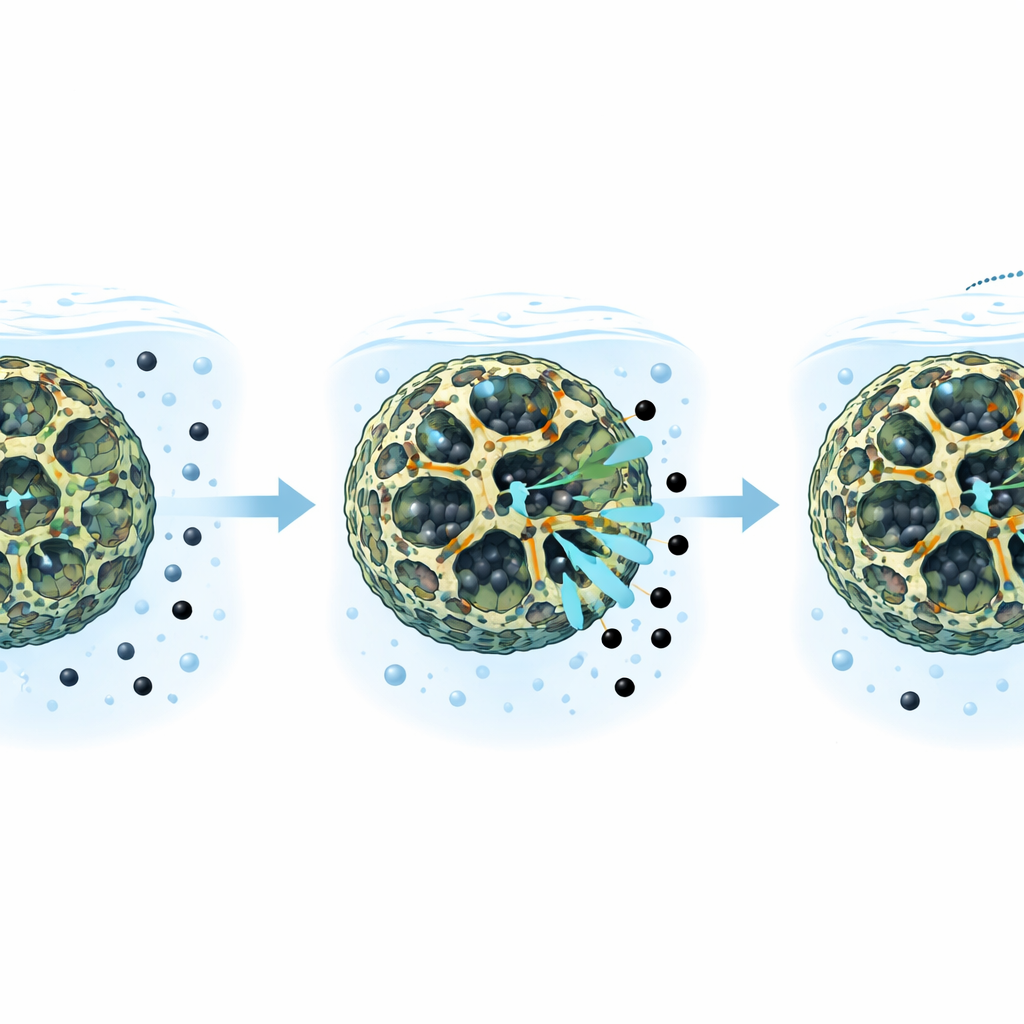
How well it removes lead from water
The team then tested how effectively their composite pulls lead out of water under different conditions. They found that it can hold over 300 milligrams of lead per gram of material and remove about 99% of lead from test solutions when the water has a mildly acidic to neutral pH, similar to many natural waters. Most of the lead is captured within the first couple of hours. Mathematical models that describe how substances stick to surfaces fit the data best when they assume that lead forms a single, ordered layer on the composite and that the binding involves relatively strong, almost lock-and-key style interactions rather than just weak attraction. The process worked slightly better at warmer temperatures, suggesting that a bit of added heat helps lead ions move into and attach inside the pores.
Working in realistic, mixed waters
Real wastewater is rarely pure; it contains many other salts and organic substances that can compete for the same binding sites. The researchers therefore added other common ions and colored molecules one at a time to see if they hindered lead capture. Some positively charged metals and dyes did reduce performance somewhat by competing for the same negatively charged spots on the material’s surface, but even under these more crowded conditions the composite still captured most of the lead. Importantly, the material withstood repeated cleaning: after soaking up lead, it could be rinsed in a mild acid bath to release the trapped metal and then reused. After six such cycles, it still removed about 90% as much lead as it did the first time, and only a very small amount of its chromium content leaked out, indicating that the framework remains largely intact.
What this means for safer water
Put simply, this new composite acts like a durable, reusable sponge that is especially good at locking onto lead in water. Its intricate internal structure gives it many places to hold the contaminant, while the natural components help attract and bind lead strongly. Because it combines high capacity, fast action, and long-term stability, and because it is made in water from relatively benign ingredients, the material stands out as a promising candidate for polishing contaminated water in treatment plants or other engineered systems. While further tests in real-world wastewaters and in continuous-flow setups are still needed, this work points toward practical, eco-friendly tools that could help communities reduce exposure to one of the most dangerous heavy metals.
Citation: Hemdan, M., Fayad, E., Binjawhar, D.N. et al. In situ assembled MIL-101(Cr) composite functionalized with chitosan and tannic acid as an efficient adsorbent for Pb2+ removal from aqueous solutions. Sci Rep 16, 9960 (2026). https://doi.org/10.1038/s41598-026-45525-x
Keywords: lead removal, water purification, adsorbent materials, metal-organic frameworks, chitosan composites