Clear Sky Science · en
Functionalization of Polyvinyl pyrrolidone/Polyvinyl alcohol blends doped with zinc sulfate and irradiated with electron beam
Stronger Films for Future Energy Devices
Modern batteries and supercapacitors rely on thin films that can safely move charged particles while withstanding heat and mechanical stress. This study explores how to fine‑tune such films made from two common, low‑toxicity plastics—polyvinyl alcohol (PVA) and polyvinyl pyrrolidone (PVP)—by adding zinc compounds and exposing them to a carefully controlled electron beam. The result is a tougher, more orderly material that conducts electricity better, pointing toward safer, high‑performance energy storage components. 
Blending Everyday Polymers with Smart Additives
PVA and PVP are already used in products ranging from eye drops and tablets to food packaging and hydrogels. They mix well because one carries many "water‑loving" hydroxyl groups while the other carries carbonyl groups that can form strong hydrogen bonds with them. In this work, the researchers dissolved PVA and PVP in water, blended them in a 40:60 ratio, and added glycerol and a small amount of acetic acid to introduce additional bonding sites and flexibility. They then dissolved zinc sulfate into this mixture, cast it into thin films, and allowed it to dry, creating a soft, gel‑like solid that holds zinc ions throughout its structure.
Shaping Nanoparticles with an Electron Beam
The key twist in the study is the use of an intense electron beam, applied at several dose levels, to “activate” the films. When high‑energy electrons pass through the moist polymer, they split water molecules and generate a burst of highly reactive fragments and mobile electrons. These species, helped by glycerol and acetic acid, progressively transform dissolved zinc ions into tiny particles of zinc oxide and zinc sulfide. At the same time, the energetic treatment causes parts of the polymer chains to link together and become more ordered. Microscopy shows that the zinc‑based nanoparticles are evenly dispersed and grow slightly in size as the dose increases, filling the pores of the film without clumping. 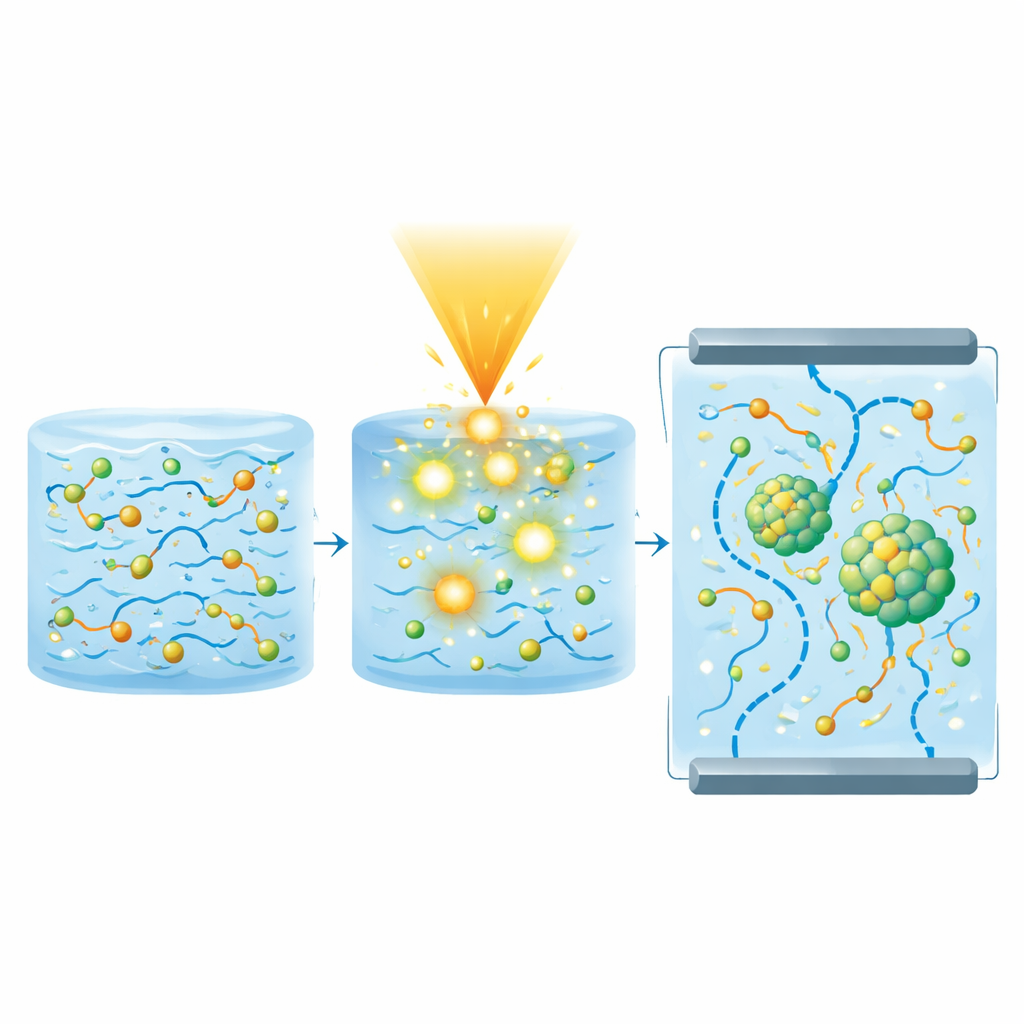
From Soft Blends to Stable, Ordered Films
Multiple measurement techniques reveal how this radiation treatment changes the material from the inside out. X‑ray diffraction shows that the films evolve from mostly disordered to noticeably more crystalline, with crystallinity rising from about 6% to more than 27% as the dose is raised. Infrared spectroscopy confirms stronger interactions between the two polymers and a growing number of “free” zinc‑related ions that can move under an electric field. Electron microscopy and elemental analysis confirm that zinc, oxygen, and sulfur are all present, forming a mix of zinc oxide and zinc sulfide within the polymer framework. Thermal tests show that the glass transition and melting temperatures shift upward, and that more energy is needed to start breaking the material down, all signs of improved heat resistance.
Better Charge Transport for Energy Applications
The authors also probed how easily charges move through the films. By placing the material between metal electrodes and sweeping over a range of frequencies and temperatures, they built up a picture of how ions respond. The electrical response curves show shrinking semicircles and growing low‑frequency tails as temperature and radiation dose increase, indicating that ions move more freely and that the bulk resistance falls. Dielectric measurements reveal that the films store and release electrical energy more effectively at higher doses, especially at 40 kGy, where the balance between crystalline order and flexible, disordered regions appears optimal. Analysis of subtle relaxation processes shows that charged species hop through the polymer network along pathways created and refined by the zinc nanoparticles and radiation‑induced structural changes.
What This Means for Real‑World Devices
Taken together, the results show that a relatively simple recipe—mixing two safe polymers with a zinc salt and then exposing the dried film to an electron beam—can turn a soft blend into a robust, finely structured solid that tolerates heat and moves ions efficiently. For a lay reader, the message is that the researchers have found a way to “cook” a plastic‑based gel so that it develops internal nanoscopic scaffolding, making it both tougher and better at shuttling charge. Such materials are strong candidates for the thin solid layers inside future supercapacitors and other energy‑storage devices, potentially improving performance and safety without relying on flammable liquids or rare ingredients.
Citation: Abdelmaksoud, H., Salah, M., Zakaria, K.M. et al. Functionalization of Polyvinyl pyrrolidone/Polyvinyl alcohol blends doped with zinc sulfate and irradiated with electron beam. Sci Rep 16, 12348 (2026). https://doi.org/10.1038/s41598-026-45515-z
Keywords: polymer nanocomposite, zinc nanoparticles, electron beam irradiation, solid electrolyte, supercapacitor materials