Clear Sky Science · en
PMMA/pPFPA membrane with low content of modified TiO2 nanoparticles for effective retention of pharmaceuticals from water
Why cleaning medicine-tainted water matters
Traces of everyday medicines such as painkillers and heart drugs are now regularly found in rivers, lakes, and even drinking water. Standard wastewater treatment plants were never designed to catch these tiny molecules, so many slip through and end up back in our taps. This study explores a new type of plastic filter, or membrane, that not only strains out pharmaceuticals from water but also helps break them down into harmless substances when exposed to ultraviolet (UV) light.
A smarter filter for stubborn pollutants
The researchers set out to build a membrane that combines three useful functions in one material: letting water pass, trapping drug molecules on its surface, and breaking them apart using light. They blended two polymers, PMMA and pPFPA, to form the main structure and added a small amount of specially modified titanium dioxide (TiO2) nanoparticles. Titanium dioxide is well known as a light-activated cleaner, but it often clumps or washes out of filters. Here, the particles were coated so they can form strong chemical bonds to the polymer, helping them stay fixed in place and work efficiently over time.

Tuning the membrane’s inner architecture
To fine-tune how water and pollutants move through the material, the team made two versions of the membrane. The first (M1) contained just the basic polymer blend and TiO2. The second (M2) also included two water-loving additives, PEG and PVP, which act as pore-forming agents during fabrication. Microscopy images revealed that M1 had relatively large, open pores, while M2 developed a denser, sponge-like structure with pores nearly an order of magnitude smaller and far more uniform. Measurements showed that the average pore diameter shrank by about 85%, and the surface became more water-attracting, making it easier for water to wet and flow through the membrane.
How the new membrane behaves in water
The team carefully measured the surface charge and wetting behavior of both membranes, because these properties control how pharmaceuticals interact with the material. Across typical water pH values, both membranes carried a negative charge, which tends to repel negatively charged drug molecules and reduce permanent fouling. The M2 membrane, thanks to PEG and PVP, was slightly less negative but more hydrophilic, taking up water more readily. Gas adsorption tests revealed that, beneath the visible structure, M2 contained a fine network of nanometer-scale pores with a higher surface area than M1. This combination of small, well-connected pores and water-loving chemistry gave M2 a good balance of water permeability and plenty of surface sites where pollutants can be captured.
Putting the membrane to the test
The researchers then challenged the M2 membrane with a mixture of three common drugs: diclofenac (a painkiller), ibuprofen, and metoprolol (a heart medication). In a flowing setup that mimics real filtration, the membrane removed less than a third of each drug by simple passage through the pores, reflecting their small size relative to the pore openings. However, when the membrane was left in still water, up to 70% of some pharmaceuticals adhered to its surface through sorption. The real breakthrough came when UV light was switched on. The bound TiO2 nanoparticles generated highly reactive radicals that attacked the drug molecules, breaking their rings and ultimately converting them into smaller, less harmful substances such as carbon dioxide and water.
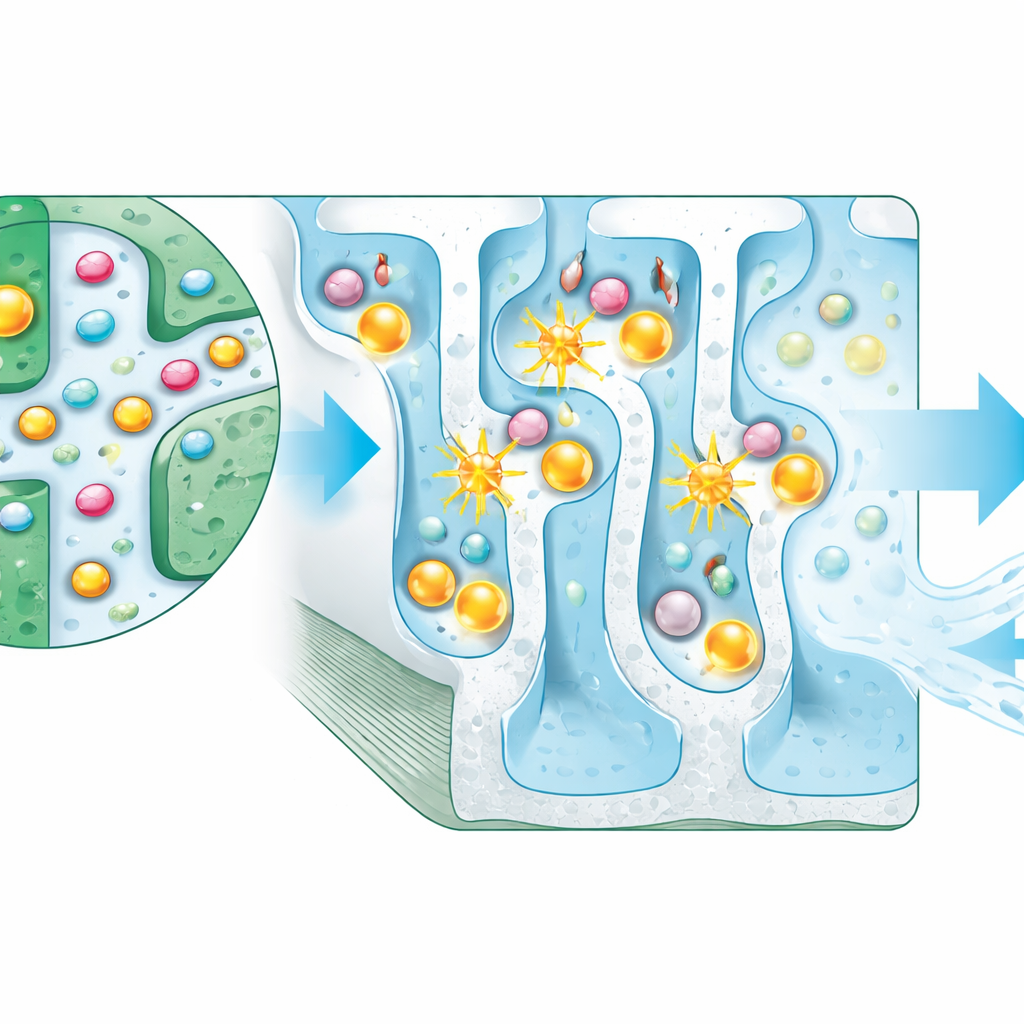
Light-driven cleanup from the inside out
During photocatalysis tests, the membrane achieved complete removal of all three pharmaceuticals within about two hours under UV illumination, far outperforming similar filters reported in earlier studies. Importantly, only a very small fraction of the titanium dioxide—less than 0.05%—was detected leaving the membrane, showing that the chemical bonding strategy effectively locks the nanoparticles in place. The experiments also teased apart the contributions of simple sieving, surface sorption, and light-driven breakdown, demonstrating that adsorption and photocatalysis, rather than size-based rejection alone, are the main engines of contaminant removal in this design.
What this means for future drinking water
Overall, the study presents a robust, light-activated membrane that can handle pharmaceutical pollution through a combination of physical trapping and on-surface destruction, even with very low amounts of catalyst. For a layperson, this means moving beyond filters that merely hold onto contaminants toward materials that help erase them. If scaled up and coupled to suitable UV sources, such membranes could enable compact water treatment steps that quietly scrub out leftover medicines before they reach our taps, offering a promising tool for cleaner and safer drinking water.
Citation: Pasichnyk, M., Schmitt, C., Plank, M. et al. PMMA/pPFPA membrane with low content of modified TiO2 nanoparticles for effective retention of pharmaceuticals from water. Sci Rep 16, 10506 (2026). https://doi.org/10.1038/s41598-026-45387-3
Keywords: pharmaceuticals in water, photocatalytic membranes, titanium dioxide nanoparticles, advanced water treatment, polymer nanocomposites