Clear Sky Science · en
Electrochemical behaviour of Na-ion and Na-S batteries under external magnetic fields
Why tiny magnets matter for future batteries
As our demand for affordable, large-scale energy storage grows, scientists are looking beyond today’s lithium batteries to cheaper, more abundant options. Sodium-based batteries are promising candidates, but they suffer from problems that shorten their life and threaten safety. This study explores a surprisingly simple helper—an external magnetic field—to see whether invisible magnetic forces can guide charged particles inside sodium batteries, making them safer, longer lasting, and more efficient without changing their internal chemistry.
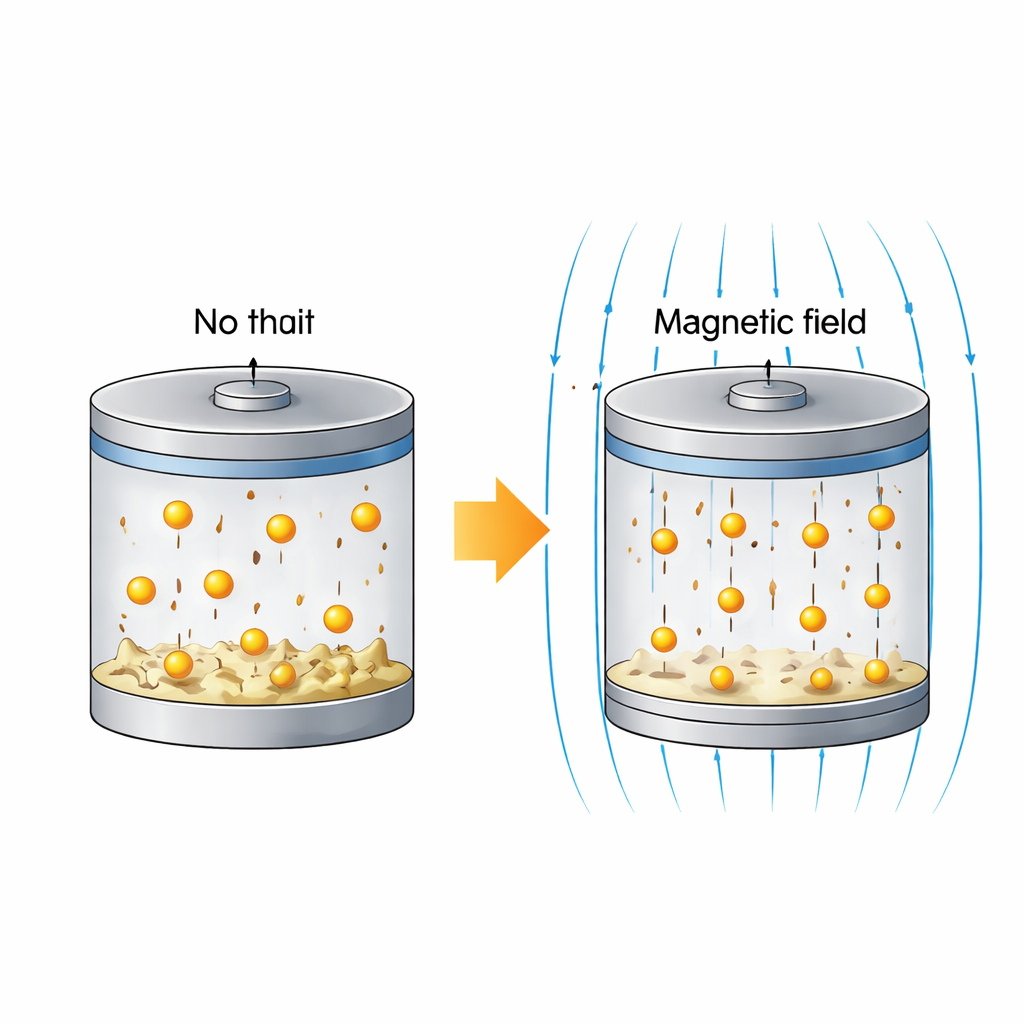
New tricks for sodium batteries
Sodium is chemically similar to lithium but far more plentiful and evenly distributed around the world, making sodium batteries attractive for grid storage and other large-scale uses. Yet when metallic sodium is charged and discharged, it tends to grow needle-like spikes, called dendrites, that can pierce the separator and cause short circuits. In sodium–sulfur cells, another problem arises: dissolved sulfur species can drift back and forth between the electrodes, wasting active material and slowly draining capacity. Most improvements so far have focused on redesigning battery materials. Here, the researchers asked a different question: can an external magnetic field, applied from outside the cell, subtly steer ion motion and improve performance even when no magnetic additives are present?
Testing batteries inside a magnetic field
To find out, the team built two kinds of small coin cells. The first was a simple sodium–sodium “symmetric” cell, ideal for watching how sodium metal itself behaves during repeated plating and stripping. The second was a sodium–sulfur half-cell using a sulfur–polyacrylonitrile composite cathode, a common design for room-temperature Na–S batteries. They then exposed these cells to static magnetic fields ranging from 50 to 450 millitesla using permanent magnets and a controlled solenoid setup, while identical control cells were cycled with no field. By recording voltage during charge–discharge, probing internal resistance with impedance measurements, and later dissecting the cells under an electron microscope, they could connect electrical behavior to physical changes inside the batteries.
Smoother sodium metal under magnet control
In the sodium–sodium cells, the presence of a magnetic field made the voltage curves more stable and reduced the extra voltage, or polarization, needed to move sodium ions. Control cells without the field developed distorted voltage shapes and abrupt changes that signal the growth of dendrites and the onset of “soft” short circuits, where filaments temporarily connect the electrodes. Impedance measurements showed that cells under a 250 millitesla field consistently had the lowest resistance for charge transfer at the sodium surface. These trends fit with magnetohydrodynamic effects: the field nudges moving ions and the surrounding liquid into gentle circulation, thinning the stagnant layer near the metal and evening out ion arrival. The result is more uniform sodium deposition and a delayed onset of harmful needle growth, even though the field cannot eliminate it entirely.
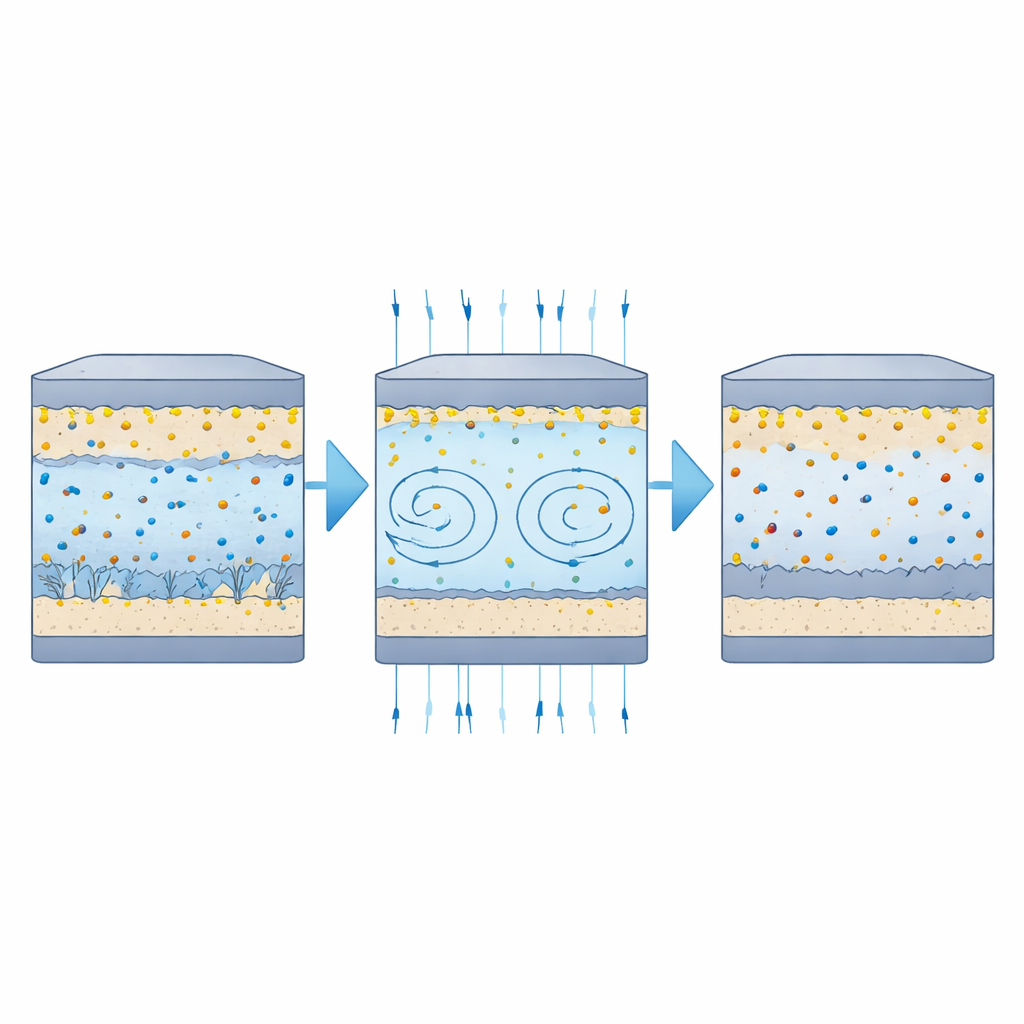
Keeping sulfur where it belongs
The sodium–sulfur cells told a similar story from the cathode side. When cycled at a moderate rate, both field-free and field-assisted cells lost capacity during the first few dozen cycles, reflecting the usual early losses of this chemistry. After that initial drop, however, the cells operated under a 250 millitesla field held on to more capacity—about 100 milliampere-hours per gram extra—and faded more slowly over 100 cycles. Their voltage curves showed smaller gaps between charge and discharge, hinting at easier ion transport and more reversible sulfur reactions. Across a wide range of current rates, the magnet-exposed cells consistently delivered slightly higher capacities. After testing, separators from the control cells were strongly stained, signaling that sulfur species had migrated and deposited far from the cathode, whereas separators from the magnet-assisted cells were noticeably cleaner. Microscopy also revealed fewer salt-rich spikes and more uniform surfaces in electrodes cycled with the field, consistent with stirred, more evenly distributed reaction products.
What this means for real-world batteries
Taken together, the experiments show that an external magnetic field—even without any special magnetic particles inside the battery—can measurably reshape how ions move and react in sodium-based cells. By gently stirring the electrolyte at the microscopic level, the field smooths out concentration hot spots, encourages more even sodium metal growth, and slows the wandering of sulfur species that sap capacity. The improvements are modest rather than miraculous, and magnetic fields do not erase all failure modes, but they offer a non-invasive design lever that could be combined with better materials and architectures. If refined and scaled, this approach may help future sodium batteries become safer, longer-lived workhorses for large-scale energy storage.
Citation: Alimbetova, G., Assan, N., Koishybay, S. et al. Electrochemical behaviour of Na-ion and Na-S batteries under external magnetic fields. Sci Rep 16, 10806 (2026). https://doi.org/10.1038/s41598-026-45275-w
Keywords: sodium batteries, magnetic fields, dendrite suppression, sodium–sulfur cells, ion transport