Clear Sky Science · en
Study on the deoxygenation characteristics of RP-5 aviation fuel via nitrogen-enriched air scrubbing inerting
Keeping airplane fuel tanks safe
When we board an airliner, we rarely think about the fuel tanks hidden inside the wings and fuselage. Yet those tanks hold large volumes of flammable liquid right beneath our feet. Since a tragic mid‑air explosion in 1996, engineers have worked hard to keep fuel tanks from ever reaching a dangerous mix of fuel vapor and oxygen. This study explores a promising way to make tanks safer by stripping oxygen out of the fuel itself using streams of tiny bubbles of nitrogen‑enriched air.
How bubbles can protect a fuel tank
Modern passenger jets already use special systems to pump low‑oxygen air into the empty space above the fuel, called the ullage, to prevent flames from starting. But as an aircraft climbs and the outside pressure drops, dissolved air inside the fuel can suddenly come out of solution as bubbles. That extra oxygen can undo the protection and briefly raise the fire risk. The technique studied here, called fuel scrubbing inerting, tackles the problem at its source. Instead of treating only the space above the fuel, nitrogen‑enriched air is injected from the bottom of the tank as a cloud of bubbles. As these bubbles rise, oxygen moves from the fuel into the bubbles, which then burst into the ullage carrying the removed oxygen away through a vent. 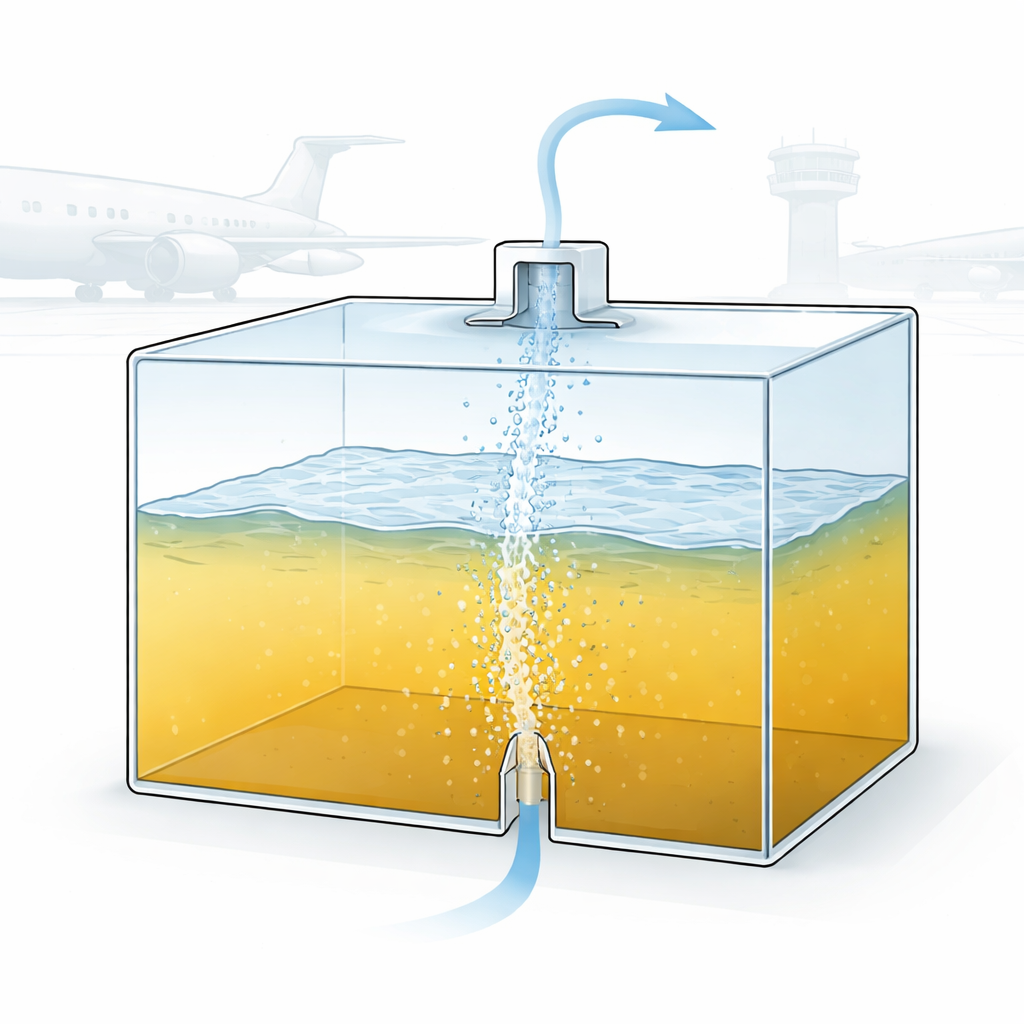
Building a virtual fuel tank
Because jet fuel is opaque and actual tanks are complex, watching what really happens inside is extremely difficult. The researchers therefore built a detailed computer model of a simplified tank filled with RP‑5 aviation fuel, a heavy, viscous fuel used in some aircraft. In their virtual tank, the liquid fuel and gas bubbles were treated as two intermingling fluids whose motion and exchange of oxygen and nitrogen could be calculated in three dimensions and over time. They used established turbulence and mass‑transfer models and fed in temperature‑dependent properties of RP‑5 fuel, such as density, viscosity, and how easily gases dissolve in it. To make sure the simulations were realistic, they constructed an experimental rig with controlled nitrogen‑enriched air, sensors for oxygen in both fuel and ullage, and cameras to measure actual bubble sizes. The agreement between experiment and simulation was close, with only a few percent difference, giving confidence that the virtual tank captured the key physics.
Why smaller bubbles matter most
With the model validated, the team explored how three knobs—bubble size, oxygen level in the nitrogen‑rich gas, and gas temperature—affect how quickly oxygen can be stripped from the fuel. The clearest message came from bubble size. When the average bubble diameter was reduced from 2.5 millimeters to 1.0 millimeter, the overall oxygen transfer rate per unit volume increased by almost a factor of four. The reason is simple geometry: many small bubbles provide far more surface area than a few large ones, giving oxygen more interface across which to jump from liquid to gas. The study showed that even though larger bubbles travel faster through the fuel, their lower surface area makes them much less effective at cleaning out dissolved oxygen. In practical terms, this means that devices that break the incoming gas into fine microbubbles are critical to making scrubbing systems compact and efficient.
Balancing purity and temperature of the gas
The amount of oxygen left in the nitrogen‑enriched air is another powerful lever. Gas with less oxygen creates a stronger mismatch between the fuel and the bubbles, driving oxygen to leave the fuel more quickly. In the simulations, increasing the oxygen content of the scrubbing gas from 3 to 9 percent nearly doubled the time needed to bring the ullage down to a safe oxygen level, even though the basic flow patterns did not change. However, achieving very low‑oxygen gas requires more complex and heavier onboard equipment, so aircraft designers must trade purification performance against cost and weight. Temperature turned out to be trickier. Warmer gas boosts how fast oxygen molecules can move, which should improve cleaning. But for RP‑5 fuel, higher temperatures also allow the fuel to hold more dissolved gas at equilibrium. The model showed that this thermodynamic effect wins out: hotter scrubbing gas slightly speeds the early stages of oxygen removal but ultimately leaves the fuel with a higher final oxygen content, a kind of “pseudo‑inerting” that looks effective at first but falls short in the long run. 
What this means for future aircraft
Overall, the study concludes that effective use of nitrogen‑enriched air bubbles can significantly improve the safety of aircraft fuel tanks by removing dissolved oxygen before it has a chance to appear suddenly as bubbles during flight. The most important design priority is to create and maintain very small bubbles, which maximize the contact area between gas and fuel and dramatically raise the rate at which oxygen can be stripped out. Using gas with lower oxygen content further shortens the time needed to reach safe tank conditions, while careful thermal management is essential to avoid the hidden downside of high temperatures that let the fuel re‑absorb more oxygen. These insights provide a scientific foundation for future “green inerting” systems that may draw nitrogen‑rich exhaust from onboard fuel cells, helping aircraft become both safer and more energy‑efficient.
Citation: Li, C., Liu, S., Xu, L. et al. Study on the deoxygenation characteristics of RP-5 aviation fuel via nitrogen-enriched air scrubbing inerting. Sci Rep 16, 14313 (2026). https://doi.org/10.1038/s41598-026-45269-8
Keywords: aircraft fuel tank safety, nitrogen-enriched air scrubbing, jet fuel deoxygenation, microbubble mass transfer, green aviation