Clear Sky Science · en
Interplay between gelation and glass formation in silica nanoparticle colloids
From Everyday Fluids to Hidden Solids
From paints and cosmetic creams to mudflows and medical gels, many familiar materials are actually tiny particles drifting in a liquid. This study looks inside one such system, a suspension of nanometer-scale silica spheres known as Ludox, to explain why the same “liquid” can behave like water under some conditions, like jelly under others, and finally like brittle glass. By following how the particles move, stick and crowd together, the authors build a bridge between what happens at the nanoscale and the way these materials flow, crack or solidify in our hands and in the Earth.
Why Tiny Particles Matter
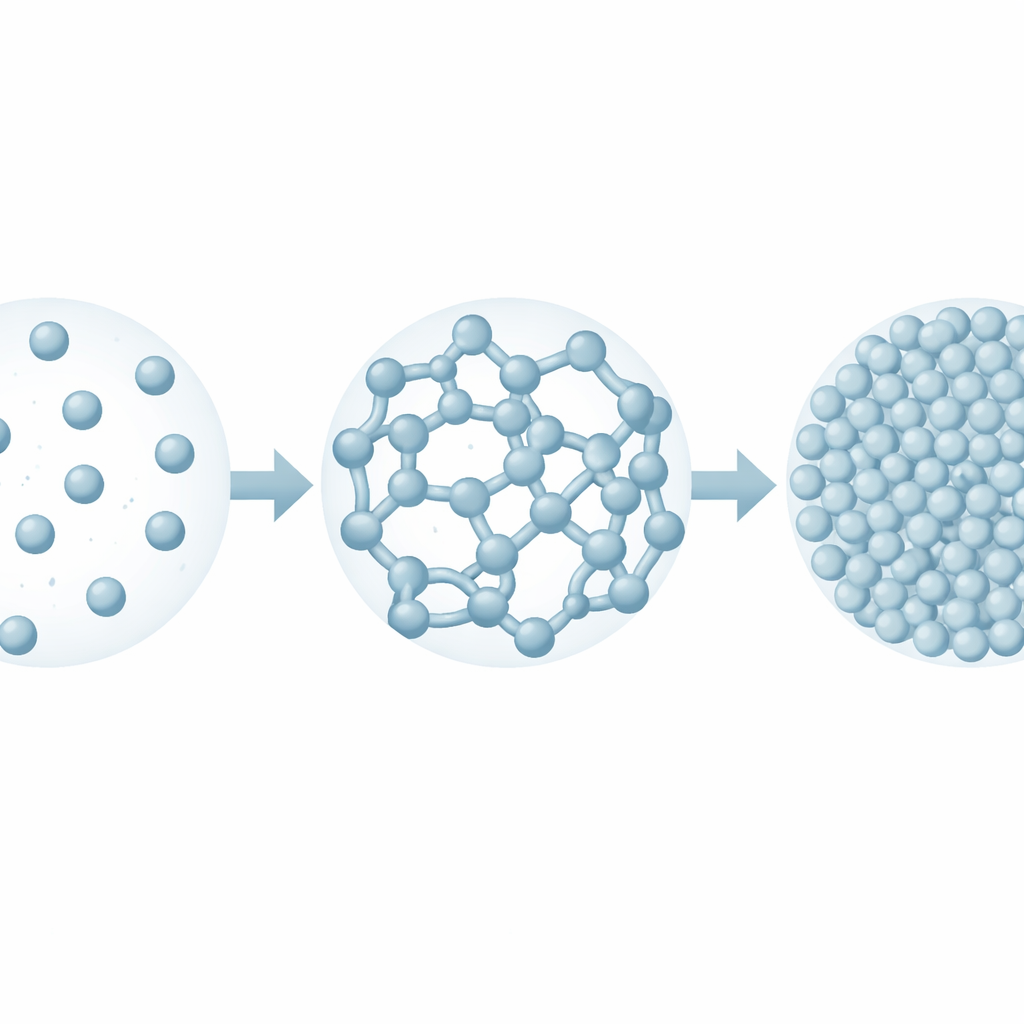
Colloidal dispersions are mixtures where solid particles, often only billionths of a meter across, are suspended in a fluid. Because these particles are so small, thermal jostling from surrounding molecules constantly pushes them around. At the same time, forces between the particles – a tug from weak attraction and a push from electric repulsion – decide whether they stay apart, form loose clusters, or lock into rigid frameworks. In Ludox, the silica particles carry an electric charge that keeps them from clumping, but as the suspension is concentrated during processes such as drying, the surrounding salt level rises and screens this repulsion. The central question of the paper is how this gradual change in the balance of forces turns a flowing liquid into a gel and then into a glass-like solid.
Watching a Network Take Shape
The researchers use detailed computer simulations to mimic Ludox suspensions over a wide range of particle concentrations. They track how the particles arrange themselves in space and how easily they can wander over long times. At relatively low concentrations, particles form loose, elongated clusters that do not span the whole sample; the material still flows like a thick liquid. As more particles are added and electric repulsion weakens, these clusters merge into a single connected network that threads through the entire volume. At the same time, the empty spaces between particles, or cavities, become fewer and smaller, and the number of contacts per particle rises toward the level required for mechanical stability. This marks the onset of a true gel: a solid-like network held together by many tiny, reversible bonds.
Slowing Motion and Approaching Glass
Beyond this gel point, increasing concentration further has a dramatic effect on motion. The team measures how fast particles diffuse by following their average squared displacement over time. They find that as soon as a system-spanning network forms, particle motion slows sharply. At still higher concentrations, diffusion almost grinds to a halt. Maps of individual particle mobilities reveal patches of slower and faster regions, a hallmark of “dynamical heterogeneity” known from glass-forming liquids. Statistical measures confirm that the distribution of particle displacements becomes strongly non-Gaussian, and a characteristic relaxation time, describing how long it takes structures to rearrange, shoots up by several orders of magnitude. Together, these signs indicate a continuous progression from a soft gel to an arrested, glass-like solid as crowding and connectivity increase.
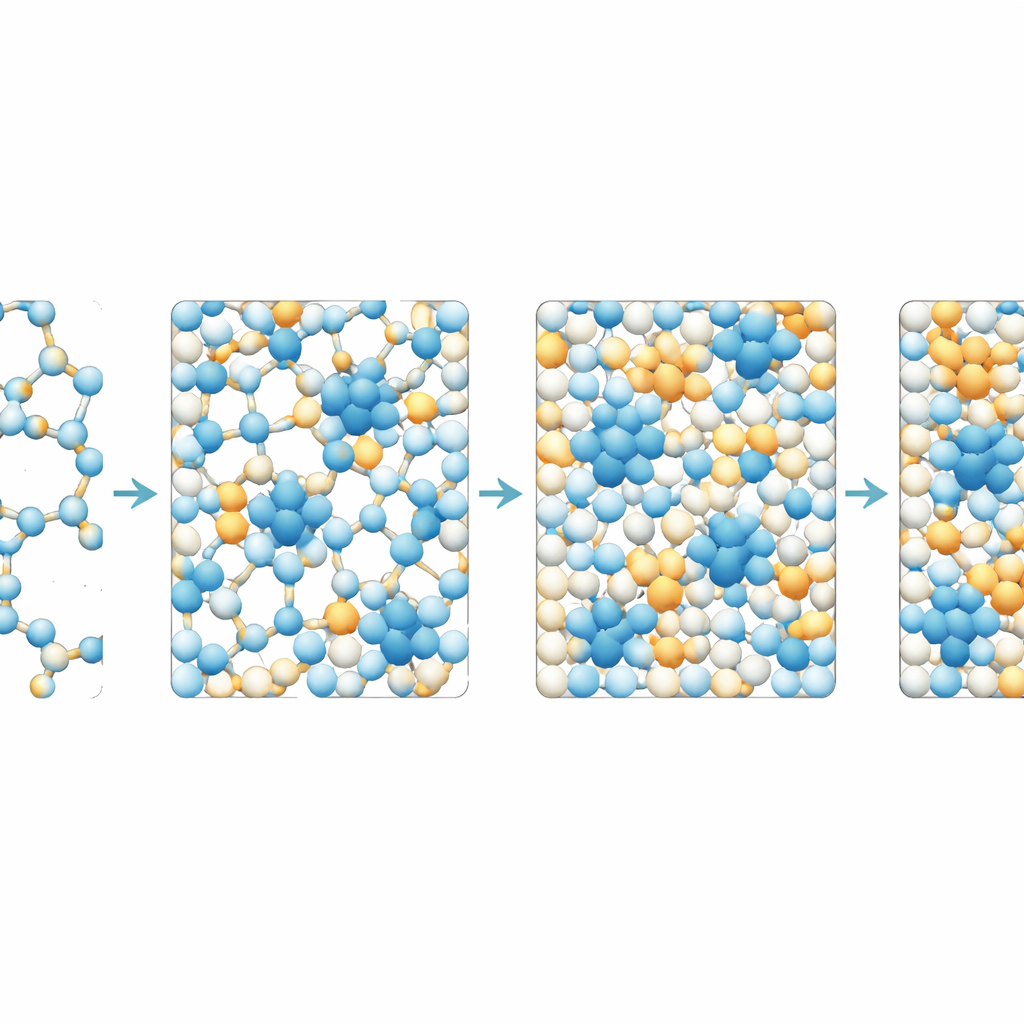
A Simple Rule for When Gels Form
To give experimenters a practical handle, the authors condense the complex interplay of particle concentration and electrostatic repulsion into a single dimensionless parameter that compares the strength of electric repulsion to thermal energy, scaled by how densely particles are packed. When they plot the simulated diffusion data against this combined parameter, results from many different conditions collapse onto a single curve. This reveals a clear threshold value: above it, particles remain dispersed and the material behaves like a liquid; below it, a percolating network forms and the system becomes a gel. The same scaling fails once the system enters the glassy regime, where crowding-driven heterogeneity dominates the dynamics, but it remains very effective across the liquid and gel states.
From Microscopic Forces to Useful Materials
In simple terms, the study shows that by tuning how strongly silica nanoparticles repel each other electrically, and how tightly they are packed, one can steer a material smoothly from fluid to gel to glass. The formation of a continuous particle network signals gelation, while the near disappearance of free space and the emergence of sluggish, uneven motion signal glass-like arrest. The proposed scaling rule turns these insights into a quantitative guide, helping researchers and engineers design colloidal products that flow when needed, set when desired, and resist cracking under stress, all by controlling conditions that are accessible in the lab and in industry.
Citation: Gerardi, G., Alba-Simionesco, C., Pépin, M. et al. Interplay between gelation and glass formation in silica nanoparticle colloids. Sci Rep 16, 10964 (2026). https://doi.org/10.1038/s41598-026-45258-x
Keywords: colloidal gels, silica nanoparticles, glass transition, rheology, electrostatic interactions