Clear Sky Science · en
Advanced simulation and numerical evaluation of pharmaceutical solubility estimation under supercritical processing using artificial intelligence computations
Why this matters for future medicines
Many modern drugs do not dissolve well in water, which can limit how much medicine actually reaches the body. Drug makers are turning to greener processing methods that use compressed carbon dioxide to improve how drugs dissolve. This study shows how artificial intelligence can help predict, on a computer, how a diabetes drug behaves in such a system so that costly trial and error in the lab can be reduced.
A cleaner way to make hard-to-dissolve drugs
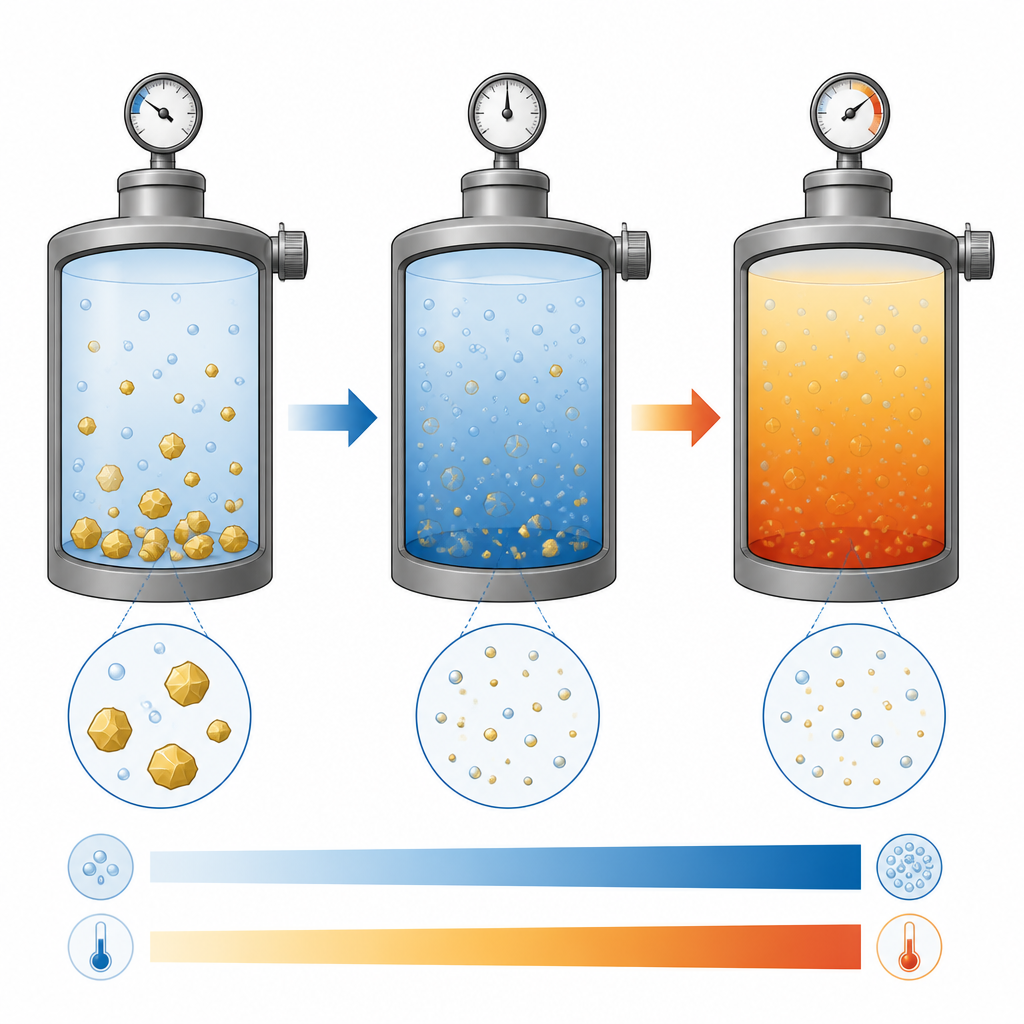
Pharmaceutical companies are interested in supercritical carbon dioxide, a special state of CO₂ where it behaves like both a gas and a liquid. In this form it can gently turn drugs into tiny particles that dissolve better, without relying on toxic organic solvents. One key challenge is knowing in advance how much of a given drug will dissolve in this unusual fluid at different temperatures and pressures. Traditional thermodynamic formulas can do this, but they require detailed measurements for each new drug and are not easy to reuse across many compounds.
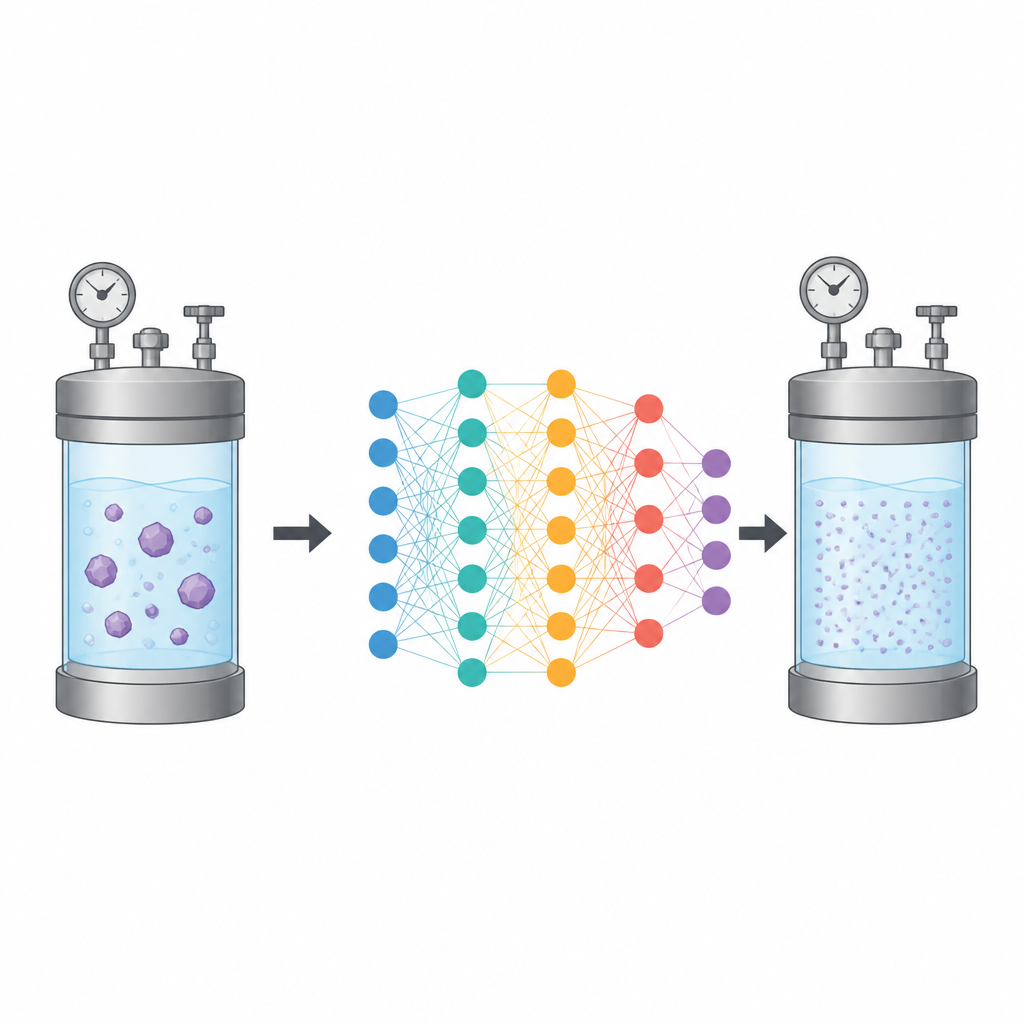
Letting computers learn from data
The researchers focused on glibenclamide, a poorly water-soluble medicine used to treat diabetes. They gathered existing experimental measurements of how much of this drug dissolves in supercritical CO₂ under many combinations of temperature and pressure, along with how dense the CO₂ is under those conditions. Using this dataset, they trained three types of artificial neural network models to learn the link between the operating conditions (temperature and pressure) and two outputs that matter for design: the density of the CO₂ and the solubility of glibenclamide.
Smarter tuning with a nature-inspired tool
To get the best performance from these neural networks, the team used a search method called the Dragonfly Algorithm, inspired by how dragonflies move in groups while hunting and migrating. Instead of testing every possible setting of the models by brute force, this algorithm guides the search toward promising combinations of internal parameters. It balances exploring new options with refining good ones, which is especially useful when many settings interact in complex ways.
What the models learned about pressure and heat
After training, all three models were able to closely match the measured solubility and density values, with a relatively simple network known as a multilayer perceptron giving the most accurate predictions. The models confirmed that raising pressure and temperature generally increases how much glibenclamide can dissolve in the supercritical CO₂, while higher temperature tends to lower the fluid’s density. By building smooth surfaces and contour maps from the predictions, the authors could visualize how these variables interact, revealing zones where small changes in pressure or temperature have strong effects on solubility or where the trend reverses at low pressure.
Beyond one drug and toward greener design
To see whether their approach was tied only to glibenclamide, the authors tested the best-performing model on solubility data for five other drugs with different structures. The model again predicted solubility in supercritical CO₂ with high accuracy, suggesting that it captures general patterns rather than memorizing one case. This means it could become a practical tool for screening and designing supercritical processes for new medicines using mainly existing data and limited extra experiments.
What this means for patients and the planet
In simple terms, this work shows that computers can learn to predict how a drug will dissolve in a cleaner CO₂-based process just from past measurements. That makes it easier for scientists to find operating conditions that give better solubility without excessive trial and error or waste. While the study does not directly test how these changes affect patients, it lays the groundwork for making difficult drugs more usable and for doing so with fewer harmful solvents, helping both medicine development and environmental sustainability.
Citation: Albariqi, A.H., Alzhrani, R.M., Alghamdi, M.A. et al. Advanced simulation and numerical evaluation of pharmaceutical solubility estimation under supercritical processing using artificial intelligence computations. Sci Rep 16, 15099 (2026). https://doi.org/10.1038/s41598-026-45225-6
Keywords: drug solubility, supercritical CO2, machine learning, pharmaceutical processing, glibenclamide