Clear Sky Science · en
Soil dynamics and ecotoxicity of zinc extracted from black mass derived from discarded batteries
Turning Old Batteries into Plant Food
Every year, mountains of used batteries are thrown away, adding to the growing problem of electronic waste. Yet inside those batteries are metals that plants actually need to grow. This study asks a simple but powerful question: can we safely turn part of that battery waste into a fertilizer that feeds crops with zinc, an essential micronutrient, without polluting soils and water?
Why Zinc and Battery Waste Matter
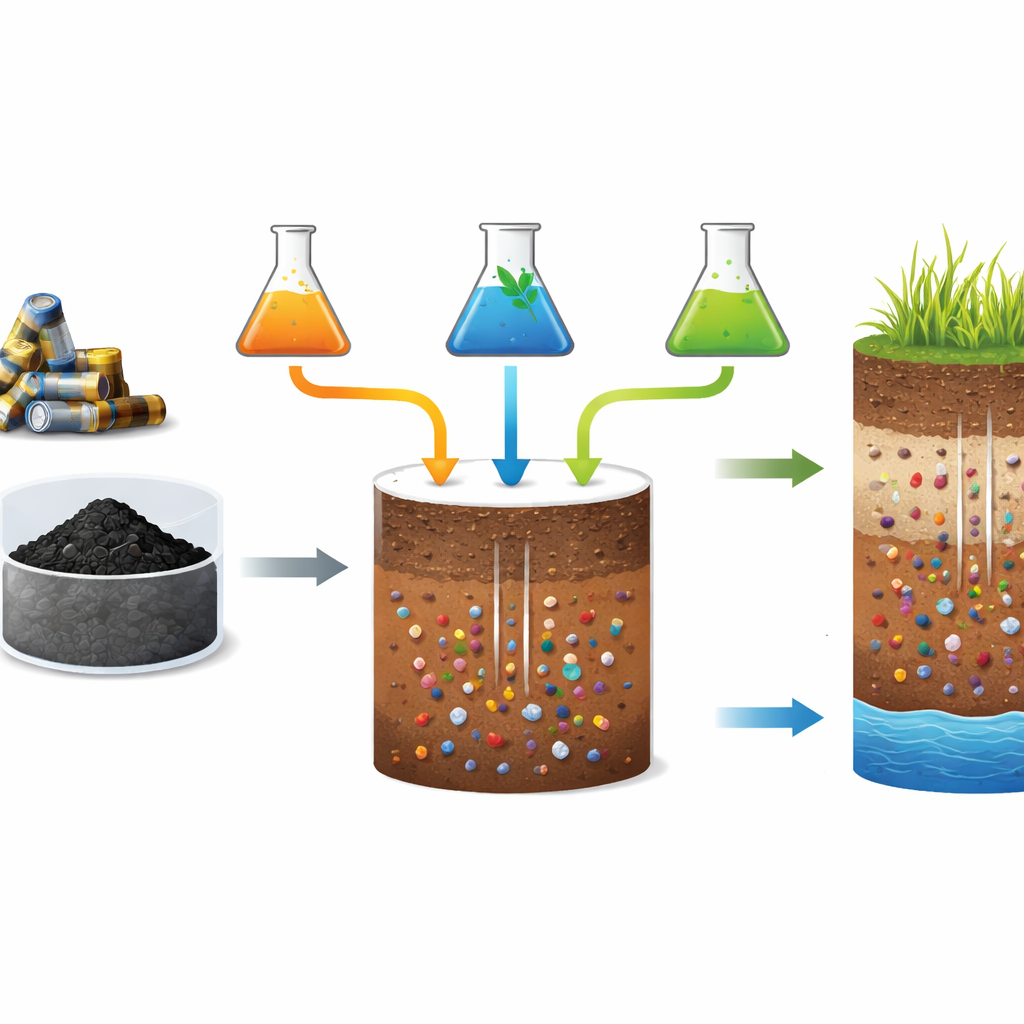
Zinc helps plants build proteins, control growth hormones, form chlorophyll, and defend themselves against stress. Many farm soils around the world do not naturally provide enough zinc, so farmers add it as a fertilizer. At the same time, discarded electronics, including common household batteries, pile up in landfills or are handled in unsafe recycling sites, releasing harmful metals into the environment. The dark powder left after processing spent zinc batteries—known as black mass—contains large amounts of zinc along with other metals. Recovering zinc from this black mass could turn a waste problem into a resource, fitting neatly into circular economy strategies that aim to reuse materials instead of throwing them away.
How Researchers Tested Recycled Zinc
The scientists extracted zinc from black mass using different liquids: two “gentler” organic options based on glycine (an amino acid) and citric acid (a common organic acid), and two stronger, more conventional sulfuric acid solutions, plus a standard commercial zinc sulfate fertilizer for comparison. They then added equal amounts of zinc from each source to columns packed with two typical agricultural soils from Spain—one acidic and sandy, the other alkaline and rich in calcium. Over time, they pumped a salt solution through the columns to mimic rainwater moving through soil, collected the water that came out, and measured how much zinc had been washed away. Afterward, they sliced the columns in half to see how much zinc remained in the upper and lower soil layers, and how much of that zinc stayed in a form that plants can use.
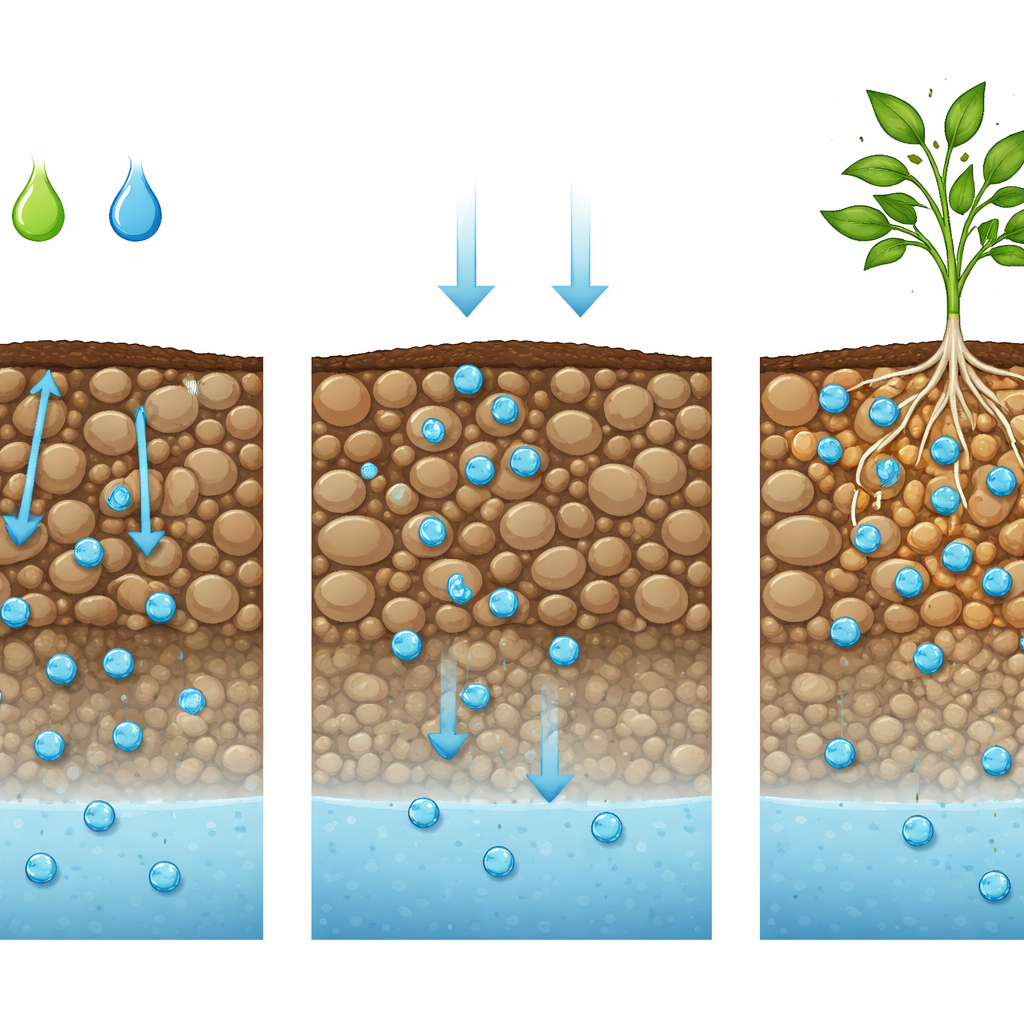
What Happened to Zinc in Different Soils
In the acidic, sandy soil, almost all of the zinc from every source eventually washed through the columns, but the speed and pattern of release differed. Zinc from strong acids and from commercial zinc sulfate came out quickly in a sharp pulse, leaving very little behind. In contrast, zinc tied up with glycine or citric acid leaked out more gradually and left slightly more plant-available zinc in the soil, especially in the upper layer. In the alkaline, calcium-rich soil, the situation was reversed. Here, zinc from the strong acid and commercial sources barely moved and tended to get locked up in forms that plants cannot easily access, especially near the surface. Glycine-based zinc, on the other hand, remained very mobile and washed almost entirely through the column, while citric-acid zinc showed an in-between behavior—some leaching, but also a portion left in a usable form near the top.
Checking for Effects on Young Plants
To understand how these zinc solutions might affect crops directly, the researchers carried out a simple seed test using tomatoes. They placed seeds on moist filter paper and watered them with different zinc concentrations from each source, from very low to quite high. At low levels, all of the zinc solutions actually stimulated germination and early seedling growth, acting as beneficial micronutrient treatments. As the dose increased, however, this benefit turned into harm. Medium to high concentrations, especially from the glycine and strong-acid extracts, shortened roots and stems and sharply reduced a combined germination-and-growth score known as the germination index. At the highest tested levels, some treatments essentially stopped seeds from establishing healthy seedlings.
What This Means for Farmers and the Environment
The study shows that zinc recovered from discarded batteries can behave much like conventional fertilizers, but its impact depends strongly on both the soil type and the chemical form of the zinc. In acidic soils, inorganic forms and strong-acid extracts risk flushing zinc downward toward groundwater, while organic complexes leak out more slowly and leave a small but useful reserve for plants. In alkaline soils, traditional zinc sources tend to get trapped and become unavailable, whereas glycine-based zinc can move too freely and be lost. For young plants, low doses from all sources can be helpful, but higher doses quickly become toxic. In practical terms, black-mass-derived zinc could become a valuable recycled fertilizer that supports sustainable agriculture—provided it is carefully formulated and applied at well-controlled rates, and its long-term environmental effects are tested in real fields before widespread use.
Citation: Almendros, P., Gascó, G., Ortiz, R. et al. Soil dynamics and ecotoxicity of zinc extracted from black mass derived from discarded batteries. Sci Rep 16, 14302 (2026). https://doi.org/10.1038/s41598-026-44947-x
Keywords: e-waste recycling, zinc fertilizer, soil leaching, battery waste, sustainable agriculture