Clear Sky Science · en
Eco-optimized RP-HPLC method for chiral tazobactam and piperacillin drugs: integration with Box–Behnken design and multi-sustainability tools
Cleaner tests for lifesaving antibiotics
Hospitals rely on powerful antibiotics like piperacillin and tazobactam to treat life‑threatening infections, especially in vulnerable patients such as those receiving liver transplants. Yet checking that these drugs are made correctly and stay stable usually depends on lab tests that use large volumes of harsh solvents. This study introduces a laboratory method that can measure both drugs at once while sharply cutting the environmental burden of the analysis itself—showing how medical quality control and greener chemistry can go hand in hand.
Why these drugs matter
Piperacillin and tazobactam are given together to combat serious infections caused by bacteria that resist many other treatments. In transplant patients, where infections are common and kidneys are already under strain, getting the dose right is crucial: too little can let infections rage, too much can damage the kidneys. Drug manufacturers and hospital labs therefore need precise, reliable tests to confirm how much of each drug is present in injectable products and to monitor how the mixture behaves over time.
A greener way to separate and measure
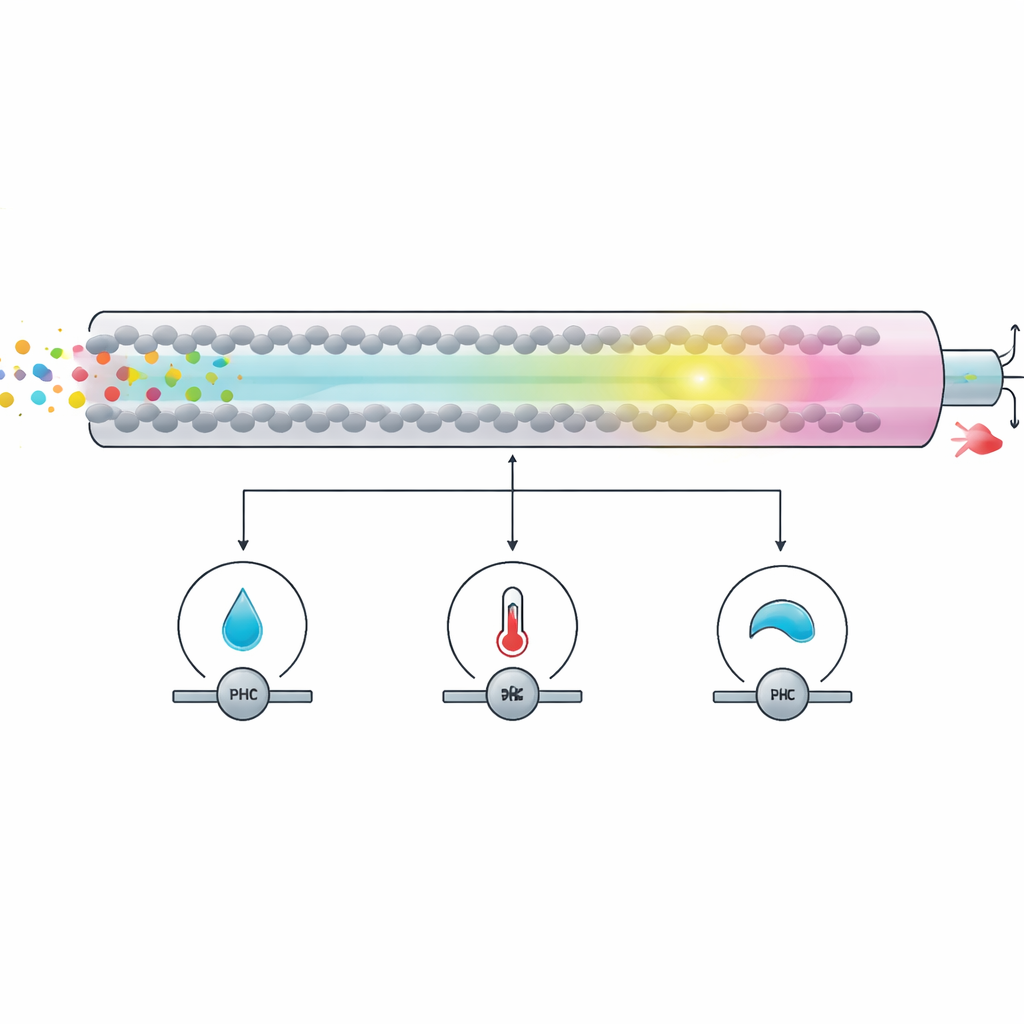
The authors focused on improving a workhorse lab technique called high‑performance liquid chromatography (HPLC), which separates a drug mixture as it flows through a packed column, allowing each ingredient to be measured. Traditional versions of this method often rely on solvents such as acetonitrile or methanol, which are effective but more toxic and resource‑intensive. In the new approach, the liquid mixture that carries the sample is based mainly on ethanol and water, with a small amount of an additive to help the two drugs travel differently through the column. By carefully choosing a slightly acidic pH and a modest column temperature, the team achieved clear separation of the two drugs in under eight minutes while keeping solvent choices more environmentally benign.
Smart optimization instead of trial and error
Rather than tuning the method by guesswork, the researchers used a statistical planning tool called Box–Behnken design. This approach treats key settings—pH, flow rate, and column temperature—as variables in a structured grid of experiments, allowing the software to map how each one affects separation quality and analysis time. From just 15 runs, the team identified conditions that gave sharp, well‑resolved peaks for both drugs and highly consistent results. They then tested the method according to international guidelines, showing that it is linear (accurate across a wide concentration range), precise (repeatable from run to run and between analysts), and robust (little affected by small day‑to‑day changes in settings).
Measuring how green the method really is
To move beyond simple solvent substitution, the study applied several independent “greenness” and sustainability scores that examine energy use, waste volume, toxicity of chemicals, and overall carbon footprint. Tools such as CaFRI, AGREE, GAPI, MoGAPI, the Analytical Eco‑Scale, and an index of solvent volume intensity all rated the new method as strongly eco‑friendly. For example, it earned an Eco‑Scale score of 87 out of 100, considered outstanding, and a MoGAPI score of 85 out of 100, with no high‑risk red zones. Compared with previously published methods and the official pharmacopoeia procedure, the new setup uses less hazardous solvents, generates less waste per analysis, and still delivers equal or better analytical performance.
What this means for patients and the planet
In practical terms, the method offers pharmaceutical companies and hospital laboratories a fast, dependable way to check the quality of piperacillin–tazobactam injections while shrinking the environmental footprint of routine testing. Although the authors note that further work is needed to fully prove how the method handles breakdown products during long‑term stability studies, their results show that advanced statistics and sustainability metrics can be woven into everyday analytical chemistry. As more drug assays are redesigned along these lines, it becomes easier for healthcare systems to protect both patients from infection and the environment from unnecessary chemical and energy use.
Citation: Al‐Wasidi, A.S., Sanari, J.A., Alminderej, F.M. et al. Eco-optimized RP-HPLC method for chiral tazobactam and piperacillin drugs: integration with Box–Behnken design and multi-sustainability tools. Sci Rep 16, 11668 (2026). https://doi.org/10.1038/s41598-026-44942-2
Keywords: green analytical chemistry, antibiotic quality control, HPLC method development, sustainable pharmaceuticals, piperacillin tazobactam