Clear Sky Science · en
Carbon sequestration potential of electrokinetically treated fine-grained expansive soils
Why treating troublesome soils matters
Across dry and semi-dry regions, certain clays swell when wet and shrink when dry, cracking roads and stressing building foundations. At the same time, engineers are searching for ways to lock away carbon dioxide in solid form. This study brings these two concerns together by testing whether a gentle electrical treatment can both tame unstable clay soils and store small amounts of carbon inside them as new minerals.
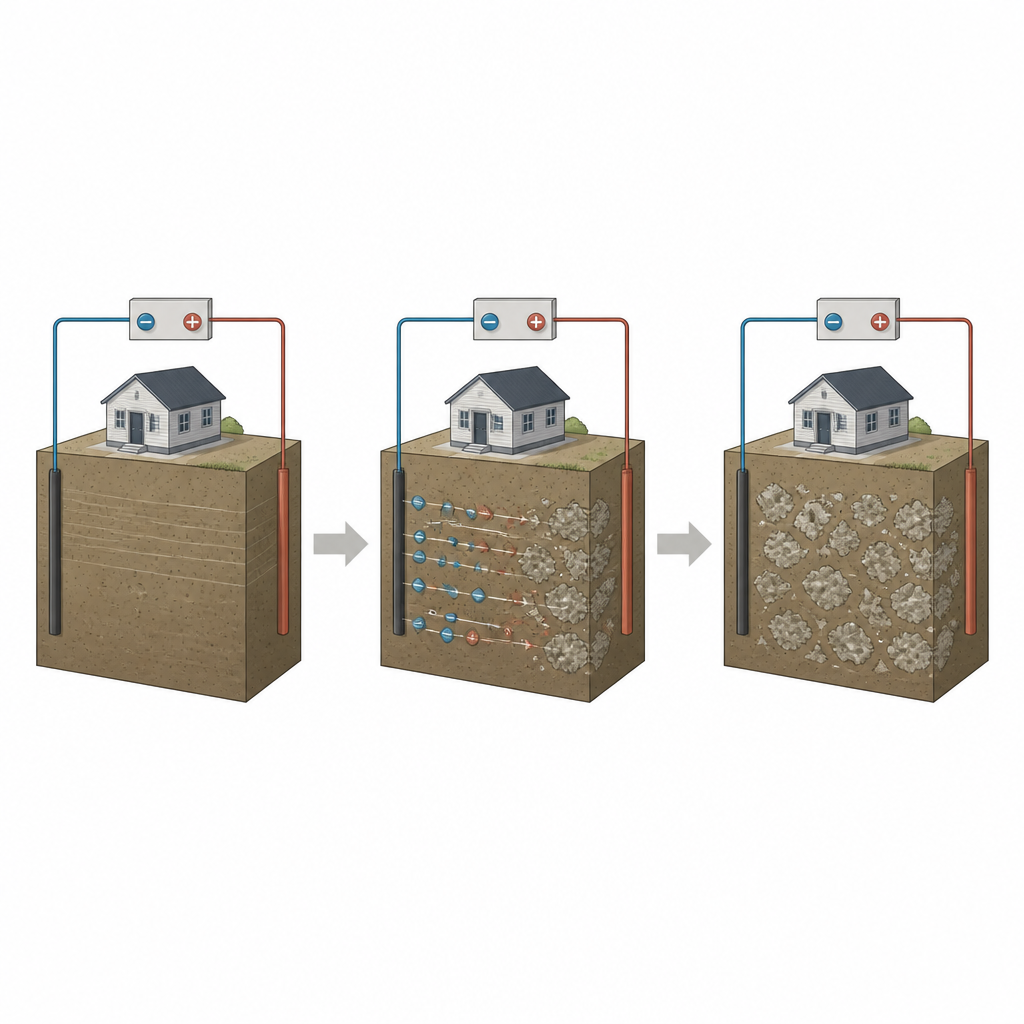
Turning electricity into underground change
The research focuses on a method called electrokinetic treatment, in which a low direct current is applied across a block of fine-grained expansive soil. Metal plates act as electrodes at opposite sides of the soil, and salty solutions rich in calcium and carbonate ions are placed next to them. When the current flows, charged particles move through the tiny water-filled pores in the clay. This controlled movement encourages calcium and carbonate to meet and solidify as calcium carbonate, a stable mineral that can store carbon dioxide within the soil matrix.
Designing a careful laboratory test bed
To explore this process in detail, the author collected intact blocks of highly plastic clay from a coastal site in North Cyprus, where seasonal wetting and drying cause serious ground movement. In the lab, each block was placed in a clear plastic tank between two electrolyte chambers containing calcium chloride and sodium carbonate solutions. A modest voltage was applied for 28 days while common water-quality measures such as acidity level, salt content, total dissolved solids, electrical conductivity, and resistivity were recorded several times per day inside the soil. By carefully weighing the solid material that formed and using basic chemical ratios, the team inferred how much calcium carbonate had precipitated and how much carbon dioxide it could represent.
From measurements to a predictive map
Rather than looking at each measurement in isolation, the study used a structured statistical approach known as response surface methodology to run 48 different combinations of conditions. This allowed the author to build an equation that links the routine readings to the amount of carbon locked in minerals. Three factors stood out as especially important: total dissolved solids, how easily the pore water conducts electricity, and its electrical resistivity. The model also revealed that some pairs of factors work together in non-obvious ways. For example, medium salt levels and slightly alkaline conditions favor mineral formation, and the best results occur when conductivity is high while resistivity is low, indicating a well-connected pathway for ions to travel and react.

How much carbon can this soil really store
Using a calibrated constant that ties measured conductivity directly to inferred carbon content, the model predicts that, under the best laboratory conditions, the treated soil can immobilize about 2 grams of carbon dioxide for every kilogram of dry soil, or roughly 2 kilograms per tonne. This capacity is modest on its own, but it is achieved while simultaneously improving the stiffness and stability of a problematic foundation soil. The framework turns simple field-friendly measurements into a quick way to estimate how much carbon might be stored during treatment, at least for this particular clay and electrolyte pair.
What this means for future building sites
The work shows that low-voltage electrical treatment can promote the formation of carbon-bearing minerals within expansive clays, offering a path to slightly lower-carbon ground improvement. However, the author is careful to stress that these results come from a single soil type and a controlled setup, and that the stored carbon was inferred rather than directly identified under a microscope. Before engineers rely on this technique at full scale, pilot projects must confirm the mineral phases formed, test how they hold up under weather changes, and weigh the energy used against the carbon stored. Even so, the study provides a clear starting map for tuning electrokinetic treatments so that they stabilize the ground while quietly locking away some carbon.
Citation: Abiodun, A.A. Carbon sequestration potential of electrokinetically treated fine-grained expansive soils. Sci Rep 16, 15068 (2026). https://doi.org/10.1038/s41598-026-44896-5
Keywords: electrokinetic treatment, expansive clay, carbon sequestration, calcium carbonate, soil stabilization