Clear Sky Science · en
Disentangling solvent effects on optical response via p-π and sp² lone-pair contributions in non-aromatic fused systems
Shaping Light with Smart Liquids
Light is at the heart of high‑speed internet, advanced sensors, and laser-based technologies. This study explores how we might build the next generation of “smart” molecules that can bend, double, and switch light on demand—and how simply changing the surrounding liquid can dramatically boost their performance. By understanding how solvents tug on electrons inside specially designed organic molecules, the authors show a path to cheaper, tunable materials for photonics and optoelectronics.
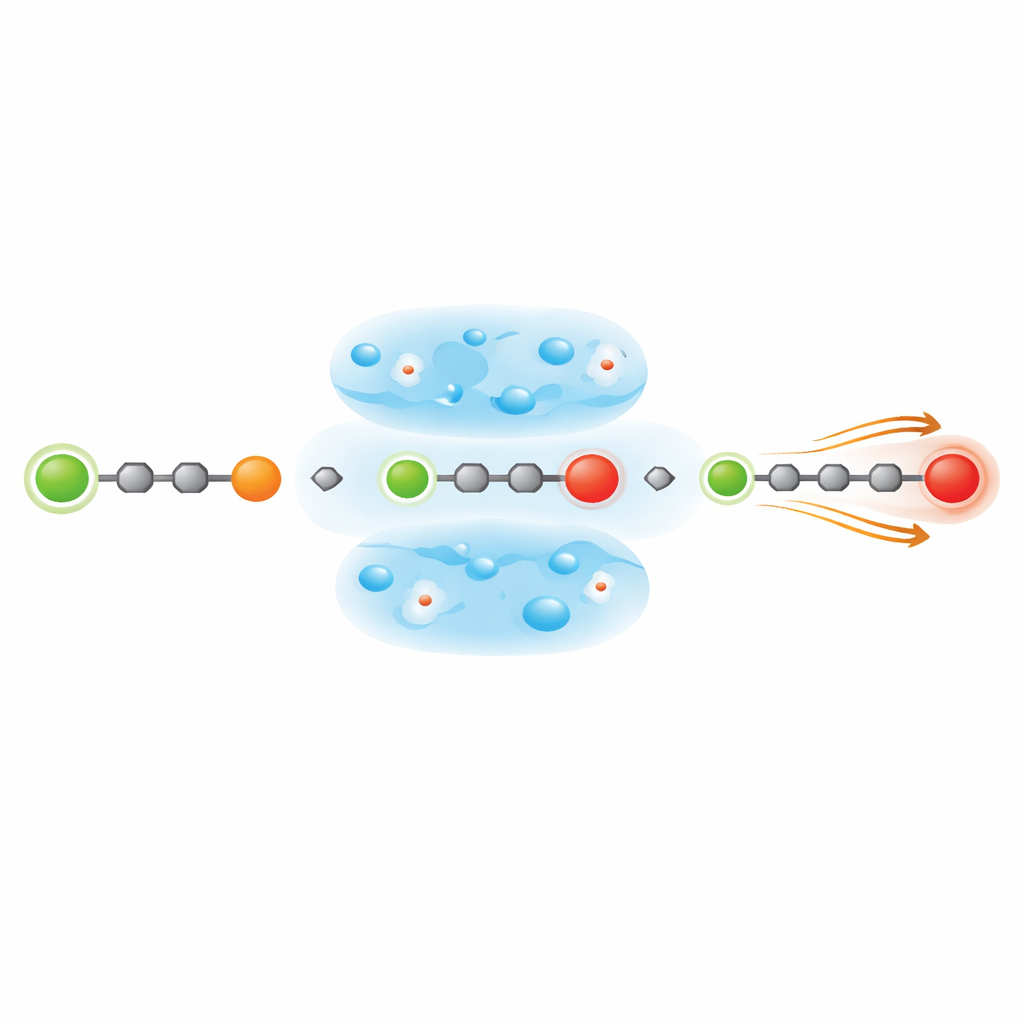
Building Push–Pull Molecular Wires
The researchers focused on a family of elongated organic molecules that act like tiny push–pull wires. One end of each molecule strongly donates electrons, while the opposite end strongly pulls them in. Between these ends lies a fused ring framework based on a non‑aromatic scaffold called 6H‑heptacene. Into this backbone they inserted one atom from a trio of “chalcogens”—oxygen, sulfur, or selenium—to see how the heavier or lighter atom alters the way electrons can shift. They then placed different electron-hungry groups at one end (such as forms related to nitro, cyano, or aldehyde units) and a strong electron‑giving group at the other, creating nine distinct push–pull designs in three related series.
How Structure Controls Electron Flow
Using modern quantum‑chemistry tools, the team first optimized the shapes of all molecules and examined their key electronic features. They analyzed the highest occupied and lowest unoccupied molecular orbitals—the frontier levels that govern how easily electrons can move. The energy gap between these levels shrank from about 4.2 electron volts in the parent framework down to just 1.95 electron volts in the best‑performing design, labelled IM3. In these push–pull systems, electrons in the donor region can flow across the central bridge toward the accepting end when an electric field or light is applied. Additional analyses of aromaticity and electron delocalization showed that oxygen-based variants, especially IM3, encourage strong internal polarization, while carefully arranged stacked structures of the molecules further stabilize the flowing electrons.
Color Shifts and Strong Light Response
These structural tweaks have clear consequences for how the molecules interact with light. Calculations of ultraviolet–visible absorption revealed that all compounds absorb in the near‑ultraviolet region, with the main transitions corresponding to electrons leaping across the donor–acceptor bridge. As the push–pull character strengthens and the chalcogen becomes heavier, absorption shifts toward longer wavelengths, signaling more extended electron delocalization. The key performance measure here is “hyperpolarizability,” which describes how strongly a molecule’s electronic cloud distorts in response to an electric field—a hallmark of materials useful for frequency doubling and fast optical switching. While the basic skeleton hardly responds at all, some tailored derivatives show hyperpolarizability values thousands of times larger than simple benchmark molecules, with IM3 standing out as exceptional.
Liquids as Invisible Tuning Knobs
A central message of the work is that the surrounding liquid environment can act as a powerful, invisible tuning knob. The authors compared three solvents: highly polar water, moderately polar ethanol, and non‑polar benzene. In polar solvents, the separation of charge within the push–pull molecules is more strongly stabilized, making it easier for electrons to shift from donor to acceptor. As a result, the key hyperpolarizability component along the molecular axis soars—reaching extremely large values for IM3 in water—while remaining substantial for sulfur‑ and selenium‑containing counterparts IM6 and IM9. At the same time, heavy atoms like selenium enhance how easily the electron cloud can be deformed, even if the overall charge separation is slightly weaker than in oxygen-based designs.
Promise for Future Light‑Based Devices
In simple terms, this study shows that by combining a push–pull molecular design with the right central atom and the right solvent, one can greatly magnify how a material responds to light and electric fields. Oxygen‑bridged IM3, in particular, offers a rare mix of strong internal charge transfer, efficient stacking in the solid state, and outstanding nonlinear optical response, while IM6 and IM9 provide complementary performance. These findings suggest practical routes to tailor organic molecules for use in optical modulators, signal processors, and other photonic components, where selecting both the molecular structure and the operating solvent could unlock powerful and tunable light control.
Citation: Ibrahim, M., Yousuf, A., Qureshi, M.Z. et al. Disentangling solvent effects on optical response via p-π and sp² lone-pair contributions in non-aromatic fused systems. Sci Rep 16, 10935 (2026). https://doi.org/10.1038/s41598-026-44846-1
Keywords: nonlinear optics, push–pull molecules, solvent effects, organic photonics, charge transfer